Ticagrelor and Bradyarrythmias
Article Information
Ranjan Modi1*, Rajiv Mehrotra2, Diwakar Kumar3
1Associate consultant, Cardiology, Indraprastha Apollo Hospital, New Delhi, India
2Senior Consultant , Cardiology, Indraprastha Apollo Hospital , New Delhi , India
3Senior Registrar, Cardiology, Indraprastha Apollo Hospital , New Delhi , India
*Corresponding Author: Ranjan Modi, Associate consultant, Cardiology, Indraprastha Apollo Hospital, New Delhi, India
Received: 15 May 2021; Accepted: 24 May 2021; Published: 01 June 2021
Citation: Ranjan Modi, Rajiv Mehrotra, Diwakar Kumar. Ticagrelor and Bradyarrythmias. Cardiology and Cardiovascular Medicine 5 (2021): 321-325.
Share at FacebookKeywords
Ticagrelor; Bradyarrythmias
Article Details
1. Introduction
Ticagrelor, a oral direct-acting and reversible P2Y12-adenosine diphosphate receptor antagonist, is recommended as a first line antithrombotic agent in patients with acute coronary syndromes [1].
Unlike clopidogrel and prasugrel which require hepatic activation, ticagrelor, and its metabolite are biologically active following gastrointestinal absorption, have a more rapid onset of action and a higher degree of platelet inhibition [1]. The PLATO (Platelet Inhibition and Patient Outcomes) trial first demonstrated superiority of ticagrelor over clopidogrel when used in conjunction with aspirin, in reducing rates of cardiovascular death and recurrent myocardial infarction in patients with ACS [2]. However, ticagrelor is associated with a greater risk of minor and major bleeding (including intracranial haemorrhage), bradyarrhythmias, and dyspnoea, which are major causes of medication discontinuation [2].
The superiority of ticagrelor over other P2Y12 antagonists is thought to be mediated in part by pleiotropic properties associated with an increased concentration of adenosine, including cardioprotection, anticoagulant effects, and anti-inflammatory properties [2]. However, these pleiotropic properties can also be responsible for major adverse effects, including electrophysiological consequences. Herein, we describe the case of a patient with severe cyclical sinus bradycardia and atrioventricular (AV) block related to ticagrelor.
2. Case Presentation
A 72 -year-old male was admitted with history of chest discomfort and positive troponin. The patient was diagnosed as unstable angina. The patient had cardiac risk factors including coronary artery disease-double vessel disease in 2018 on medical treatment, hypertension, bronchial asthma, old cerebrovascular accident. He had no history of hypercholesterolaemia, elevated body mass index, or impaired glucose tolerance. His medical history was significant for psoriasis and obstructive sleep apnoea.
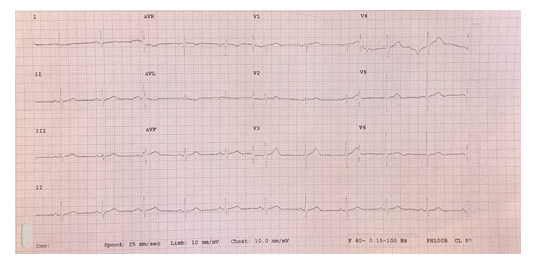
His admission electrocardiogram (ECG) showed sinus rhythm, normal axis, narrow QRS (80 ms) with no ischaemic change or conduction defects (Figure 1). Echocardiography showed normal biventricular size and systolic function, with normal valves. Radial approach coronary angiography was performed which revealed double vessel disease, the culprit vessel was shown to be the left circumflex artery (non-dominant) with a proximal 95% stenosis followed by total occlusion in mid portion. The patient was explained the need for revascularization, consent taken and then loaded with oral 180 mg of ticagrelor.
A 2.5 X 15 Xience xpedition everolimus eluting stent was deployed in LCX resulting in restoration of TIMI 3 flow (Figure 2). Medical management was elected for the patient’s residual noncritical disease in right coronary artery. The procedure was successful and uneventful. The patient was stable and shifted to the coronary care unit. Three to four hours post procedure, whilst convalescing in the coronary care unit, the patient began complaining of severe dyspnoea with no clinical signs of left ventricular failure. The patient had bradycardia episode associated with syncope.
The patient underwent emergency right femoral venous temporary pacing wire insertion. Ticagrelor was considered as the probable cause for the pauses, for that reason no additional ticagrelor was given after the loading dose and clopidogrel was commenced in place. Holter monitoring revealed minimum heart rate of 52 / min with no bradycardia episodes. No further bradyarrhythmias were detected in the subsequent 72h , the patient underwent a check angiography which revealed patent stents with TIMI 3 flow thus the un-utilized temporary pacing wire was removed. The patient was later discharged with adequate medical therapy. Betablocker therapy was not prescribed in view of preceding events. A extended holter monitoring was placed for 5 -7 days which revealed no further arrythmic episodes. No cardiorespiratory symptoms were raised during regular follow-up.
3. Discussion
Bradycardia related to ticagrelor was first described in a phase IIb dose-ranging study, where a post hoc analysis of cardiac arrhythmias revealed an unexpected increased incidence of predominantly asymptomatic ventricular pauses [3]. These findings were corroborated by the prospective PLATO (Platelet Inhibition and Patient Outcomes) trial [4]. Among 2908 patients with 7-day electrocardiographic monitoring at the time of randomization and again at 1 month, ventricular pauses > 3 seconds occurred more frequently in patients receiving ticagrelor compared to clopidogrel during the first week (5.8% vs 3.6%). Most pauses were asymptomatic, of sinoatrial origin, and nocturnal. However, no significant difference was observed in recorded bradyarrhythmias at 1 month and there were no apparent clinical bradycardic events during follow-up [4].
Nonetheless, the study concluded that ticagrelor’s bradyarrhythmic potential was transient and not clinically significant beyond the acute initiation phase with no difference in rates of syncope or need for pacemaker insertion at follow-up [3]. Since then, there have been emerging case reports of ticagrelor-associated high-degree heart block, requiring drug discontinuation and in some cases, pacemaker insertion.
The exact mechanism of ticagrelor-induced brady-arrhythmia is unclear, although inhibition of adenosine reuptake is proposed as likely. Extracellular adenosine has a half-life of several seconds due to rapid cellular uptake through sodium independent equilibrative nucleoside transporters (ENTs) and sodium dependent concentrative nucleoside transporters (CNTs). ENTs are ubiquitous, present on erythrocytes as well as the liver, heart, spleen, kidneys, lungs, intestines, and brain, while CNTs are found primarily in the liver, kidneys, and small intestine [5]. Ticagrelor shares structural similarity with adenosine, binding to ENT1 receptors on erythrocytes, and competitively inhibiting cellular adenosine uptake. Three adenosine receptor subtypes (A1, A2a, and A3) have cardiac expression with agonism resulting in bradycardia, coronary vasodilatation, and activation of multifaceted cardioprotective mechanisms, respectively [1, 6].
The earliest coronary response to myocardial ischaemia of any aetiology is vasodilatation; mediated by A2a receptors on vascular smooth muscle cells and adenosine release from the ischaemic myocardium [6]. In the setting of ACS treated with ticagrelor, extracellular adenosine concentrations are amplified both by the ischaemic milieu with myocardial adenosine release and blunted cellular reuptake. This leads to enhanced agonism of adenosine A1 receptors, highly expressed in cardiac conduction tissue, causing negative chronotropy via suppression of sinoatrial node automaticity and negative dromotropy via impulse conduction delay at the atrioventricular node.
We report this case of severe sinus respiratory arrhythmia after ticagrelor loading. Cheyne-Stokes respiration frequently occurs in patients with congestive heart failure [7] and has been associated with exaggerated respiratory heart rate variations [8]. While our patient had no evidence of heart failure, few reports in literature suggest that ticagrelor may itself induce central sleep apnea and Cheyne-Stokes respiration [9]. Proposed mechanisms include antagonism of microglial P2Y12 receptors and effects on pulmonary C fibers, either as a result of increased adenosine tissue levels or because of putative P2Y12 receptors on pulmonary C fibers [9].
4. Conclusion
This report demonstrates that, although rare, ticagrelor can induce significant bradyarrhythmias. Physicians and Interventional Cardiologists should, therefore, be aware of this reversible cause of sinus node dysfunction in order to manage patients appropriately and avoid unnecessary pacemakers.
References
- Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 361 (2009): 1045-1057.
- Kubisa MJ, Jezewski MP, Gasecka A, Siller-Matula JM, Postula M. Ticagrelor - toward more efficient platelet inhibition and beyond. Ther Clin Risk Manag 14 (2018): 129-140.
- Cannon CP, Husted S, Harrington RA, Scirica BM, Emanuelsson H, et al. Safety, tolerability, and initial efficacy of AZD6140, the first reversible oral adenosine diphosphate receptor antagonist, compared with clopidogrel, in patients with non-ST-segment elevation acute coronary syndrome: primary results of the DISPERSE-2 trial. J Am Coll Cardiol 50 (2007): 1844-1851.
- Scirica BM, Cannon CP, Emanuelsson H, et al. The incidence of bradyarrhythmias and clinical bradyarrhythmic events in patients with acute coronary syndromes treated with ticagrelor or clopidogrel in the PLATO (Platelet Inhibition and Patient Outcomes) trial: results of the continuous electrocardiographic assessment substudy. J Am Coll Cardiol 57 (2011): 1908-1916.
- Van Giezen JJ, Sidaway J, Glaves P, Kirk I, Bjorkman JA. Ticagrelor inhibits adenosine uptake in vitro and enhances adenosine-mediated hyperemia responses in a canine model. J Cardiovasc Pharmacol Ther 17 (2012): 164-172.
- Wittfeldt A, Emanuelsson H, Brandrup-Wognsen G, van Giezen JJ, et al. Ticagrelor enhances adenosine-induced coronary vasodilatory responses in humans. J Am Coll Cardiol 61 (2013): 723-727.
- Javaheri S, Parker TJ, Liming JD, Corbett WS, Nishiyama H, et al. Sleep apnea in 81 ambulatory male patients with stable heart failure. Types and their prevalences, consequences, and presentations. Circulation 97 (1998): 2154-2159.
- Leung RS, Floras JS, Bradley TD. Respiratory modulation of the autonomic nervous system during Cheyne-Stokes respiration. Can J Physiol Pharmacol 84 (2006): 61-66.
- Revol B, Jullian-Desayes I, Tamisier R, Puel V, Mallaret M, et al. Ticagrelor and central sleep apnea. J Am Coll Cardiol 71 (2018): 2378-2379.
