The Role of Electrical Cardiometry in Paediatrics and Neonatal Anaesthesia and Intensive Care: A Narrative Review
Article Information
Khaled A Yassen1*, Zahra Al Ghadeer1, Fatimah AlHejji1, Fatimah Alothman1, Huda Alethan1, Danah AlAli1, Lamis AlJamaan1, Dur I Shahwar1, Lien Reyin2, Yasser Nassef3
1Department of Anaesthesia Unit / Surgery, College of Medicine, King Faisal University, Al-Ahsa, Hofuf City, Saudi Arabia
2Department of Pediatrics, Chang Gung Memorial Hospital Linkou, Taoyuan, Taiwan
3Institute of Medicine, Chung Shan Medical University, Taiwan
*Corresponding Author: Khaled A Yassen, Department of Anaesthesia Unit / Surgery, College of Medicine, King Faisal University, Al-Ahsa, Hofuf City, Saudi Arabia
Received: 05 April 2022; Accepted: 12 April 2022; Published: 13 April 2022
Citation: Khaled A Yassen, Zahra Al Ghadeer, Fatimah AlHejji, Fatimah Alothman, Huda Alethan, Danah AlAli, Lamis AlJamaan, Dur I Shahwar, Lien Reyin, Yasser Nassef. The Role of Electrical Cardiometry in Paediatrics and Neonatal Anaesthesia and Intensive Care: A Narrative Review. Anesthesia and Critical care 4 (2022): 73-83.
Share at FacebookAbstract
Invasive hemodynamic monitoring could be challenging among pediatrics, particularly infants and new-borns. This current narrative review aims to discuss and present the published literature that addresses the optimal use and validity of the electrical cardiometry (EC) device among pediatrics. This is a non-invasive continuous cardiac output and hemodynamic monitor that measures the electrical transthoracic bioimpedance. Literature search focused on PubMed (MEDLINE), Saudi Digital library (SDL), and Google Scholar database between 2010 and 2022. English language studies were included only. The retrieved data were peer-reviewed manuscripts, including print publications and case presentations. This review provided evidence that EC monitoring among the pediatrics age group was able to track CO changes over the time (Trend). However, EC absolute values for CO were interchangeable with the CO measurements derived from the thermodilution technique in some studies and were not interchangeable in some others. The variation in clinical conditions and surgical procedures affected the performance EC CO. In specific clinical scenarios, EC CO trend changes proved to be of extreme help during transportations and in operating rooms during PDA ligation. The EC technology is easy to use and is an addition for future developments in monitoring, particularly for the pediatric age group. More studies are recommended to improve further the precision of the absolute CO measurements and to study the impact of the EC on short- and long-term clinical outcomes.
Keywords
Cardiac output, Impedance cardiography, Hemodynamic monitoring, Paediatrics, Electrical cardiometry
Cardiac output articles; Impedance cardiography articles; Hemodynamic monitoring articles; Paediatrics articles; Electrical cardiometry articles
Cardiac output articles Cardiac output Research articles Cardiac output review articles Cardiac output PubMed articles Cardiac output PubMed Central articles Cardiac output 2023 articles Cardiac output 2024 articles Cardiac output Scopus articles Cardiac output impact factor journals Cardiac output Scopus journals Cardiac output PubMed journals Cardiac output medical journals Cardiac output free journals Cardiac output best journals Cardiac output top journals Cardiac output free medical journals Cardiac output famous journals Cardiac output Google Scholar indexed journals Impedance cardiography articles Impedance cardiography Research articles Impedance cardiography review articles Impedance cardiography PubMed articles Impedance cardiography PubMed Central articles Impedance cardiography 2023 articles Impedance cardiography 2024 articles Impedance cardiography Scopus articles Impedance cardiography impact factor journals Impedance cardiography Scopus journals Impedance cardiography PubMed journals Impedance cardiography medical journals Impedance cardiography free journals Impedance cardiography best journals Impedance cardiography top journals Impedance cardiography free medical journals Impedance cardiography famous journals Impedance cardiography Google Scholar indexed journals Hemodynamic monitoring articles Hemodynamic monitoring Research articles Hemodynamic monitoring review articles Hemodynamic monitoring PubMed articles Hemodynamic monitoring PubMed Central articles Hemodynamic monitoring 2023 articles Hemodynamic monitoring 2024 articles Hemodynamic monitoring Scopus articles Hemodynamic monitoring impact factor journals Hemodynamic monitoring Scopus journals Hemodynamic monitoring PubMed journals Hemodynamic monitoring medical journals Hemodynamic monitoring free journals Hemodynamic monitoring best journals Hemodynamic monitoring top journals Hemodynamic monitoring free medical journals Hemodynamic monitoring famous journals Hemodynamic monitoring Google Scholar indexed journals Paediatrics articles Paediatrics Research articles Paediatrics review articles Paediatrics PubMed articles Paediatrics PubMed Central articles Paediatrics 2023 articles Paediatrics 2024 articles Paediatrics Scopus articles Paediatrics impact factor journals Paediatrics Scopus journals Paediatrics PubMed journals Paediatrics medical journals Paediatrics free journals Paediatrics best journals Paediatrics top journals Paediatrics free medical journals Paediatrics famous journals Paediatrics Google Scholar indexed journals Electrical cardiometry articles Electrical cardiometry Research articles Electrical cardiometry review articles Electrical cardiometry PubMed articles Electrical cardiometry PubMed Central articles Electrical cardiometry 2023 articles Electrical cardiometry 2024 articles Electrical cardiometry Scopus articles Electrical cardiometry impact factor journals Electrical cardiometry Scopus journals Electrical cardiometry PubMed journals Electrical cardiometry medical journals Electrical cardiometry free journals Electrical cardiometry best journals Electrical cardiometry top journals Electrical cardiometry free medical journals Electrical cardiometry famous journals Electrical cardiometry Google Scholar indexed journals echocardiography articles echocardiography Research articles echocardiography review articles echocardiography PubMed articles echocardiography PubMed Central articles echocardiography 2023 articles echocardiography 2024 articles echocardiography Scopus articles echocardiography impact factor journals echocardiography Scopus journals echocardiography PubMed journals echocardiography medical journals echocardiography free journals echocardiography best journals echocardiography top journals echocardiography free medical journals echocardiography famous journals echocardiography Google Scholar indexed journals stroke volume articles stroke volume Research articles stroke volume review articles stroke volume PubMed articles stroke volume PubMed Central articles stroke volume 2023 articles stroke volume 2024 articles stroke volume Scopus articles stroke volume impact factor journals stroke volume Scopus journals stroke volume PubMed journals stroke volume medical journals stroke volume free journals stroke volume best journals stroke volume top journals stroke volume free medical journals stroke volume famous journals stroke volume Google Scholar indexed journals red blood cells articles red blood cells Research articles red blood cells review articles red blood cells PubMed articles red blood cells PubMed Central articles red blood cells 2023 articles red blood cells 2024 articles red blood cells Scopus articles red blood cells impact factor journals red blood cells Scopus journals red blood cells PubMed journals red blood cells medical journals red blood cells free journals red blood cells best journals red blood cells top journals red blood cells free medical journals red blood cells famous journals red blood cells Google Scholar indexed journals intensive care unit articles intensive care unit Research articles intensive care unit review articles intensive care unit PubMed articles intensive care unit PubMed Central articles intensive care unit 2023 articles intensive care unit 2024 articles intensive care unit Scopus articles intensive care unit impact factor journals intensive care unit Scopus journals intensive care unit PubMed journals intensive care unit medical journals intensive care unit free journals intensive care unit best journals intensive care unit top journals intensive care unit free medical journals intensive care unit famous journals intensive care unit Google Scholar indexed journals
Article Details
1. Introduction
In adults, the thermodilution technique (Fick Principle) is recognized as the gold standard technique for cardiac output (CO) measurements. However, unfortunately, this method is of limited use among pediatrics during acute clinical conditions due to its invasiveness and associated risk of morbidity [1]. Current practice adopted by several physicians is to measure the CO in neonatal units with bedside echocardiography (Echo). However, the Echo examination is not suitable for continuous monitoring and requires training. This indicates the need for other alternatives to continuously monitor the CO in this age group [2,3]. Electrical Cardiometry (EC) is a non-invasive method designed for continuous cardiac output (CO) measurement and fluid intake guidance.
The EC determines an estimate of the stroke volume (SV) by measuring the thoracic electrical bio impedance (TEB) with relation to the cardiac cycle. The method of EC is based on the fact that the conductivity of the blood in the aorta changes during the cardiac cycle. The orientation of red blood cells (erythrocytes) in the aorta prior and shortly after aortic valve opening affect the conductivity and hence the TEB. Prior to opening of aortic valve, the red blood cells (RBCs) assume a random orientation and hence an electrical current passing through the aorta will face low conductivity as a result of this random position of the RBCs. Very shortly after aortic valve opening, the pulsatile blood flow forces the red blood cells to align in parallel with the blood flow. Now the same electrical current passes the red blood cells more straight forward with a higher conductivity. The alignment of RBCs in the aorta contributes to the significant change in the impedance soon after aortic valve opening (Figures 1,2). EC relies on the change in impedance of the aortic blood to calculate the CO and other hemodynamic parameters. EC estimates stroke volume (SV) and CO based on body mass and peak aortic acceleration of blood flow, and a measurement of flow time [4,5]. EC was evaluated by several researchers among children and neonates as a continuous CO monitor [6,7]. EC is portable, and this offers a considerable advantage over invasive monitors, particularly during transfer [2,8]. This current narrative review aims to search for published literatures that address the optimal use and validity of EC as a non-invasive continuous cardiac output among pediatric age groups. The limitations, lessons learned, and areas for future research and improvements will be discussed.
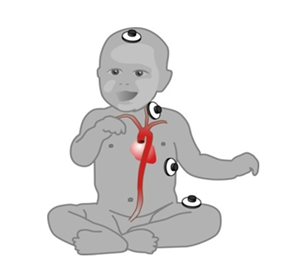
Figure 1: EC electrode array for measuring Thoracic Electric Bio impedance (TEB)
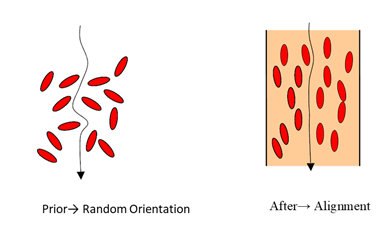
Figure 2: Orientation of red blood cells (RBCs) in aora prior and after aortic valve opening.
2. Methods
This narrative review was conducted following approval by the local research and ethics committee of King Fahad Hospital (IRB KFHH No. (H-05-HS-065), Ministry of Health, Hofuf City, Saudi Arabia. Research proposal approval number is RCA NO: 02-E-2022.
2.1 Literature Search
Relevant abstracts and published studies were retrieved from the database in specific PubMed (MEDLINE), Saudi Digital library (SDL), and Google Scholar between July 2010 and January 2021. Keywords included: cardiac output, electrical cardiometry, bioimpedance, pediatric, hemodynamic monitoring, anesthesia, critical care, and surgery. There is no restriction on a specific study design. Observational studies, randomized controlled trials, pilot, retrospective studies, case reports were included. English language studies confined to humans were only included. The studies reference list was searched for additional papers.
2.2 Population Selection
Pediatrics monitored by electrical cardiometry (EC, ICON, Cardiotronic, Osypka Medical, Inc., La Jolla CA, USA) in intensive care units (ICU), operating theatres, and during transportation. Patients with respiratory support were included and those > 18 years excluded.
2.3 Data extraction
The following data will be extracted: the name of the first author, publication year, type of study, sample size, type of surgery, and type of invasive or non-invasive devices. The effect of cardiac output (CO) and stroke volume variation (SVV) parameters of EC on hemodynamic monitoring and guided fluid therapy were identified for discussion.
3. Results
Results of this search provided evidence that EC was able to track CO values with time and to follow the trend changes in CO. However, more studies are still required to improve the precision of the EC absolute CO values compared to gold standard methods. The review looked into different studies comparing the EC with other CO monitors and covering different practical applications of the device, as studied by various research groups among pediatrics. The results and conclusions vary widely because of the heterogeneity of these published studies. The comparisons of the EC with different devices at different clinical and surgical conditions could be responsible for these variations in percentage errors and accuracy of the EC absolute CO values between various studies. However, there is growing evidence to support the benefits of EC as a non-invasive continuous cardiac output trend monitor among pediatrics, particularly in intensive care units, operating theatres, and during transportation. EC was able to guide intravenous fluids administration without the need for an invasive approach. More studies are still needed with EC at various clinical conditions to improve the accuracy of the absolute CO measurements.
4. Discussion
Cote et al. in 2015 provided evidence that supports the EC role as a continuous noninvasive CO device that can monitor rapid trend changes among pediatrics. They build their suggestion from the analysis of 402 pediatrics patients’ anesthesia records [9] Norozi et al. [5] and Narula et al. [6] studies also presented acceptable agreements between the CO of EC and that derived by the Fick principle (Gold standard) among pediatrics suffering from various tructural heart diseases. Mansfield et al. in a recent systematic review and meta-analysis also reported no significant difference between the CO derived by EC and that by other devices (except in neonatal stroke volume studies) [10]. However, Mansfield and his colleagues blamed the wide range in the percentage error they noted in their analysis on the heterogeneity of the study designs included and the difference in technology.
EC CO accuracy
Various studies looked into the accuracy of the absolute EC CO and reported conflicting results. Sanders et al., in a recent meta-analysis (2020), reported a wide range of bias with EC CO, although their pooled bias was close to zero [11]. One of the limitations in their meta-analysis was including both adults and pediatrics together in the analysis. In 2015, Suehiro et al. performed a systematic review and meta-analysis that focused on the pediatric population and looked into the precision of several minimally-invasive CO devices, including EC [12]. Their inter-study heterogeneity was significant because of the inclusion of different devices. However EC CO had the slightest bias and the lowest percentage of error. The variation in clinical conditions and surgical procedures significantly affected the CO monitors' performance included in Suehiro et al. meta-analysis. Cardiac surgery had the lowest bias and mean percentage error (MPE). Three years later (2018), Altamirano-Diaz et al. demonstrated that the CO measurements by EC in pediatric patients undergoing coarctation of aorta repair were equivalent to those by the transoesophageal echocardiography (TEE) provided that no increase in left ventricular output (LVO) is present. The increase in LVO impacts the accuracy of TEE CO and hence its agreement with the EC CO [2]. In another study group among pediatrics suffering from sepsis and septic shock, the evaluation of CO by arterial waveform analysis yielded the worst findings. The bias was negatively influenced by the low systemic vascular resistance (SVR) associated with sepsis. This was not the case with EC. EC provided real-time guidance and improved outcomes, as stated by Rao et al. in their observational pilot study [13]. The exact clinical circumstance should be defined during the validation of any device. EC performance could be suitable for certain clinical conditions, as in sepsis, while unsuitable for others [14,15]. A study by Torigoe et al. [16] in 2015 among low birth weight preterm infants with patent ductus arteriosus (PDA) provided evidence that EC was interchangeable with echocardiography (Echo) during mechanical ventilation. Boet et al. [17] in 2016 also noted that the CO of EC was in good correlation with Echo CO in hemodynamically stable preterm infants. However an overestimation was noted at high CO values. In 2017, Hsu et al. [18] reported a similar finding among preterm infants with PDA. They noted that the negative bias and the error percentage were more prominent with high CO values (>280 ml/kg/min). High-frequency ventilation (HFV) was also found to affect the EC CO. In a prospective observational study by Song et al. (40 preterm neonates), a decline in the agreement between EC CO and Echo CO was observed with HFV [19]. This could be attributable to several factors, one of them is the effect of chest wall vibrations on CO readings that associated HFV, and another is the similarity between the frequency utilized by EC and that by the high-frequency ventilator (12 Hz). Another possibility is that the increase in intrathoracic pressure caused by HFV can increase the distance from the electrodes to the descending aorta and hence diminish the bioimpedance signals. Results from Noori et al. study indicate that smaller and sicker infants have more reliable CO readings, as they tend to move less, and hence the artifacts are reduced [20].
For obese children, Simone et al. found no evidence of a male-to-female contribution to CO levels. The only statistical significance was in the preschool age but not in school-aged children. It is essential to normalize the CO to the ideal BSA for each age [21]. Boet et al [17] results indicate that SV and CO increase with higher birth weight and gestational age, and Grollmuss et al. [22] found a significant linear correlation between SV and birth weight.
Lotfy et al. conducted a randomized controlled trial among 42 infants undergoing surgical hepatoportoenterostomy (Kassi procedure). The EC CO was persistently higher than that measured by trans esophageal Doppler (TED CO) despite excellent reliability [23]. This reliability was the same within the Grollmuss et al. study, where the SV of EC and Doppler-TTE were also found to be interchangeable among newborns following cardiac switch surgery [22].
EC role in specific medical situations
The accuracy of EC as a monitor for CO varies from one clinical scenario to another. The pro and cons, and limitations will be discussed in the following specific clinical applications.
During emergency transportation
Boet et al. prospective cohort study noted that SV and CO trend monitoring was possible with EC without interference during neonatal transportation and with reliability compared to heart ultrasound [24]. This is an advantage as it allows for the continuous monitoring during the transfer of critically ill pediatric patient to specialized centres. However, it is essential to take into consideration the tendency for the SV and CO of EC to be higher than other devices as reported by Boet et al. [23] versus (vs.) ultrasound, Grollmuss et al. vs. Doppler-TTE [22] and Lotfy et al. vs.TED [23].
During patent ductus arteriosus ligation (PDA)
Lien et al. succeeded in monitoring with EC the significant reduction in SV (>73%) and the increase in SVR immediately following PDA ligation. It was the decrease in SV rather than the increase in HR that contributed to the deterioration in CO. The study was performed among very low birth weight (≤1500 g) infants [25]. The SVR increase after ligation was significant with infants suffering from larger PDAs. The smaller the infant, the greater the likelihood of cardiovascular impact. EC also noted the return of the CO to 92 and 94% of baseline respectively within 24 and 48 h. Hsu KH et al. study had proved that Infants with PDA had higher baseline CO by EC compared to those without PDA [26]. Hsu KH et al. also demonstrated that EC and Echo have a wide but clinically acceptable agreement in measuring CO in preterm infants with hemodynamically significant PDA [18]. The utilization of non-invasive cardiac output monitors duet al ring PDA ligation was also highlighted by several research teams but with different monitors. Noori et al. [27] and McNamara et al. [28] with serial echocardiographic analysis, presented similar results to that by EC in Lien et al. study. Another study with pulsed doppler technique by Linder et al. [29] demonstrated a decrease in the SV among preterm infants with very low birth weight in two days following PDA ligation. Rowland et al. also noted by echocardiography that the newborn myocardial performance was sensitive to afterload due to the immaturity of their hearts [30].
During cardiac arrhythmias and depression
Infants are less able to augment their cardiac output by increasing stroke volume as a result of their low myocardium compared to adults [31]. To maintain cardiac output, infants are classically described as heart rate dependent and can have a higher cardiac output in relation to their BSA than adults. Green et al. demonstrated that sevoflurane administration to healthy infants could lead to a nodal rhythm [32]. Dewhirst et al. also showed that following anesthesia induction, tachycardia developed in 50% of the pediatric patients in their study despite no significant changes in blood pressure [33]. Vanderhoek et al. reported that a 6-year-old boy developed supraventricular tachycardia (SVT) during an upper endoscopy while under general anesthesia [34]. EC was applied in this case to measure the effect of SVT on cardiac index and SV before and after adenosine administration. Following administration of adenosine, the CI fell by 41%, while SV decreased by 9%. This highlights the relationship between CO and HR in children and the possible future role of EC in arrhythmia monitoring and guided management. HR rhythmic changes and their effects on CO among infants can be easily monitored by EC. In another specific and rare clinical condition named Pompe disease. Liu et al. were capable of detecting the hemodynamic trend changes that associated this disease more reliably with EC than with pulse contour analysis [35]. In this rare case, an enzyme replacement therapy is usually used to reduce the severe organ dysfunction that associates with the disease. EC CO monitoring proved to be of importance in detecting the moderate cardiac depression associated with anesthesia induction.
Hemodynamics’ normal range for Neonates
Hsu et al study was one of the first studies from the Asia Pacific region to create reference normal range for neonates and premature of different age groups and weight [5].
EC Limitations
In Kadafi et al. recent study (2021), EC was unable to assess fluid responsiveness and preload adequately. They studied the EC SVV cut-off values compared to the ultrasonic CO monitor in shock children on mechanical ventilation [36,37]. Unfortunately, the sensitivity and specificity of EC were only 58% and 74%, respectively, while the optimal cut-off point of SVV was 16.5%. In 2016 Hsu et al. suggested that the hemodynamic reference ranges of EC can only be of help as an alarm, when CO falls outside the reference value for neonates in NICU [5]. Hsu et al. added that the hemodynamic reference of EC is affected by the neonates’ maturity, age, and body size. The weight and BSA had a positive correlation with CO, while the HR and SVR had a negative correlation with CO. SVV is also affected by the diameter of the blood vessels. The larger the neonate, the less the SVV and the more is the CO. Hence the cardiac index is better as it includes the BSA. CI is recommended for cardiac performance assessment among neonates to reduce maturity differences.
5. Conclusion
This review provided evidence that EC can track CO changes with time and warn against significant hemodynamic changes, but more in the future are still required to improve the precision of the absolute EC CO measurements as presented in several studies. The variation in clinical conditions and the type of the surgical procedure affected the performance of the EC CO. In specific clinical scenarios, the EC CO was found to be of extreme help as during transportations of neonates and infants, in operating rooms during PDA ligation, and in NICU.
Potential conflict of interest
Yasser Nassef is a memeber of the advisory board for Osypka medical GmbH.
All other authors reported no potential conflict of interest relevant to this article.
No funding was allocated to the research.
IRB number
Approved by the local research and ethics committee of King Fahad Hospital (IRB KFHH No. (H-05-HS-065), Ministry of Health, Hofuf City, Saudi Arabia. Research proposal approval number is RCA NO: 02-E-2022.
Clinical trial registration number
Not applicable.
References
- Jeremy S, Hertzig, Jacqueline M, et al. Schenkma: Principles of Invasive Monitoring, In Fuhrman B, Zimmerman J (eds): Pediatric Critical Care (Fourth Edition), Mosby 12 (2011): 234-245.
- Altamirano-Diaz L, Welisch E, Dempsey AA, et al. Non-invasive measurement of cardiac output in children with repaired coarctation of the aorta using electrical cardiometry compared to transthoracic Doppler echocardiography. Physiol Meas 39 (2018): 055003.
- Electrical Cardiometry Technology (EC), Osypka Medical, Cardiotronic (2021).
- Osypka M. An introduction to electrical cardiometry. Cardiotronic (2009).
- Norozi K, Beck C, Osthaus WA, et al. Electrical velocimetry for measuring cardiac output in children with congenital heart disease. Br J Anaesth 100 (2008): 88-94.
- Narula J, Chauhan S, Ramakrishnan S, et al. Electrical Cardiometry: A Reliable Solution to Cardiac Output Estimation in Children with Structural Heart Disease. J Cardiothorac Vasc Anesth 31 (2017): 912-917.
- Hsu KH, Wu TW, Wang YC, et al. Hemodynamic reference for neonates of different age and weight: a pilot study with electrical cardiometry. J Perinatol 36 (2016): 481-485.
- Grollmuss O, Gonzalez P. Non-invasive cardiac output measurement in low and very low birth weight infants: a method comparison. Front Pediatr 25 (2014): 16-25.
- Coté CJ, Sui J, Anderson TA, et al. Continuous noninvasive cardiac output in children: Is this the next generation of operating room monitors? Initial experience in 402 pediatric patients. Paediatr Anaesth 25 (2015): 150-159.
- Norozi K, Beck C, Osthaus WA, et al. Electrical velocimetry for measuring cardiac output in children with congenital heart disease. Br J Anaesth 100 (2008): 88-94.
- Mansfield RC, Kaza N, Charalambous A, et al. Cardiac output measurement in neonates and children using noninvasive electrical bioimpedance compared with standard methods: A Systematic Review and Meta-Analysis. Crit Care Med 50 (2022): 126-137.
- Sanders M, Servaas S, Slagt C. Accuracy and precision of non-invasive cardiac output monitoring by electrical cardiometry: a systematic review and meta-analysis. J Clin Monit Comput 34 (2020): 433-460.
- Suehiro K, Joosten A, Murphy LS, et al. Accuracy and precision of minimally-invasive cardiac output monitoring in children: a systematic review and meta-analysis. J Clin Monit Comput 30 (2016): 603-620.
- Rao SS, Lalitha AV, Reddy M, et al. Electrocardiometry for Hemodynamic Categorization and Assessment of Fluid Responsiveness in Pediatric Septic Shock: A Pilot Observational Study. Indian J Crit Care Med 25 (2021):185-192.
- Biais M, Mazocky E, Stecken L, et al. Impact of Systemic Vascular Resistance on the Accuracy of the Pulsioflex Device. Anesth Analg 124 (2017): 487-493.
- Slagt C, Helmi M, Malagon I, et al. Calibrated versus uncalibrated arterial pressure waveform analysis in monitoring cardiac output with transpulmonary thermodilution in patients with severe sepsis and septic shock: an observational study. Eur J Anaesthesiol 32 (2015): 5-12.
- Torigoe T, Sato S, Nagayama Y, et al. Influence of patent ductus arteriosus and ventilators on electrical velocimetry for measuring cardiac output in very-low/low birth weight infants. J Perinatol 35 (2015): 485-489.
- Boet A, Jourdain G, Demontoux S, et al. Stroke volume and cardiac output evaluation by electrical cardiometry: accuracy and reference nomograms in hemodynamically stable preterm neonates. J Perinatol 36 (2016): 748-752.
- Hsu KH, Wu TW, Wu IH, et al. Electrical Cardiometry to Monitor Cardiac Output in Preterm Infants with Patent Ductus Arteriosus: A Comparison with Echocardiography. Neonatology 112 (2017): 231-237.
- Song R, Rich W, Kim JH, et al. The use of electrical cardiometry for continuous cardiac output monitoring in preterm neonates: a validation study. Am J Perinatol (2014): 1105-1110.
- Noori S, Drabu B, Soleymani S, et al. Continuous non-invasive cardiac output measurements in the neonate by electrical velocimetry: a comparison with echocardiography. Arch Dis Child Fetal Neonatal Ed 97 (2012): F340-F343.
- De Simone G, Devereux RB, Daniels SR, et al. Stroke volume and cardiac output in normotensive children and adults. Assessment of relations with body size and impact of overweight. Circulation 95 (1997): 1837-43.
- Grollmuss O, Demontoux S, Capderou A, et al. Electrical velocimetry as a tool for measuring cardiac output in small infants after heart surgery. Intensive Care Med 38 (2012): 1032-1039.
- Lotfy M, Yassen K, El Sharkawy O, et al. Electrical cardiometry compared to transesophageal doppler for hemodynamics monitoring and fluid management in pediatrics undergoing Kasai operation. A randomized controlled trial. Pediatric Anesthesia and Critical Care Journal 6 (2018): 46-54.
- Boet A, Jourdain G, Demontoux S, et al. Basic Hemodynamic Monitoring Using Ultrasound or Electrical Cardiometry During Transportation of Neonates and Infants. Pediatr Crit Care Med 18 (2017): e488-e493.
- Lien R, Hsu KH, Chu JJ, et al. Hemodynamic alterations recorded by electrical cardiometry during ligation of ductus arteriosus in preterm infants. Eur J Pediatr 174 (2015): 543-550.
- Hsu KH, Wu TW, Wu IH, et al. Baseline cardiac output and its alterations during ibuprofen treatment for patent ductus arteriosus in preterm infants. BMC Pediatr 19 (2019): 179.
- Noori S, Friedlich P, Seri I, et al. Changes in myocardial function and hemodynamics after ligation of the ductus arteriosus in preterm infants. J Pediatr 150 (2007): 597-602.
- McNamara PJ, Stewart L, Shivananda SP, et al. Patent ductus arteriosus ligation is associated with impaired left ventricular systolic performance in premature infants weighing less than 1000 g. J Thorac Cardiovasc Surg 140 (2010): 150-157.
- Lindner W, Seidel M, Versmold HT, et al. Stroke volume and left ventricular output in preterm infants with patent ductus arteriosus. Pediatr Res 27 (1990): 278-281.
- Rowland DG, Gutgesell HP. Noninvasive assessment of myocardial contractility, preload, and afterload in healthy newborn infants. Am J Cardiol 75 (1995): 818-821.
- De Souza D, McDaniel GM, Baum VC. Cardiovascular physiology. In: Davis PJ, Cladis FP, Moyotoma EK, (eds). Smith's anesthesia for infants and children, (8th edtn), Philadelphia: Elsevier Mosby 12 (2011): 80-115.
- Green DH, Townsend P, Bagshaw O, et al. Nodal rhythm and bradycardia during inhalation induction with sevoflurane in infants: a comparison of incremental and high-concentration techniques. Br J Anaesth 85(2000): 368-370.
- Dewhirst E, Lancaster C, Tobias JD. Hemodynamic changes following the administration of propofol to facilitate endotracheal intubation during sevoflurane anesthesia. Int J Clin Exp Med 6 (2013): 26-29.
- Vanderhoek SM, Coté CJ. Measurement of cardiac index and stroke volume using electrical cardiometry before and after administration of adenosine in a 6-year-old patient with supraventricular tachycardia. J Clin Anesth 27 (2015): 682-684.
- Liu MC, Wang MT, Chen PK, et al. Case Report: Anesthetic management and electrical cardiometry as intensive hemodynamic monitoring during cheiloplasty in an infant with enzyme-replaced pompe disease and preserved preoperative cardiac function. Front Pediatr 9 (2021): 729-824.
- Kadafi KT, Latief A, Pudjiadi AH. Determining pediatric fluid responsiveness by stroke volume variation analysis using ICON® electrical cardiometry and ultrasonic cardiac output monitor: A cross-sectional study. Int J Crit Illn Inj Sci 10 (2020): 123-128.
