The Correlation between Slow Coronary Flow and Galectin-3
Article Information
Mehmet Ali Kobat1*, Mehmet Kalayci2
1Department of Cardiology, Faculty of Medicine, Firat University, Elazig, Turkey
2Department of Biochemistry, Health Sciences University, Fethi Sekin City Hospital, Elazig, Turkey
*Corresponding author: Mehmet Ali Kobat, Department of Cardiology, Elazig Education and Research Hospital, Elazig, Turkey
Received: 06 July 2021; Accepted: 19 July 2021; Published: 02 August 2021
Citation: Mehmet Ali Kobat, Mehmet Kalayci. The Correlation between Slow Coronary Flow and Galectin-3. Cardiology and Cardiovascular Medicine 5 (2021): 423-434.
Share at FacebookAbstract
Background: It was investigated whether Galectin-3 is one of the contributing factors to slow coronary flow (SCF). In the cases with SCF angiography shows lagged opacification of epicardial coronary arteries. Research has reported that several determinants of SCF are inflammation and endothelial dysfunction. It is a new finding that Galactin-3 also causes intravascular inflammation followed by lipid endocytosis, macrophage activation, cellular proliferation, monocyte chemotaxis, and cell adhesion, successively. Thus, the cases enrolled in this study were selected based on their angiographical negative results with obstructive coronary artery disease.
Method: The study group consists of 94 patients with SCF, and the control group of 92 with normal coronary flow (NCF). The total (n=186) were the cases who were examined for diagnosis of coronary artery disease based on coronary angiography records and atherosclerotic lesion was not detected. SCF rates were identified using TIMI frame count (TFC).
Results: The groups had similar demographic characteristics at the baseline. The study group had significantly higher values (2.4 ± 0.6 mg/dL and 16.6 ± 0,7 ng/mL) than the control group (1.1 ± 0.2 mg/dL and 9.2 ± 0,6 ng/mL) in terms of average serum high sensitivity C-reactive protein (hsCRP) (p=.005) and Galectin-3 (p=.003), respectively. For diagnostic purpose at a 95% confidence interval, the plasma Galectin-3 levels were moderate to distinguish the SCF with the NCF patients (0.7220.722
Conclusion: It was determined that serum Galectin-3 levels were significantly higher in the SCF patients. It may be too early to reach a conclusion related to the underlying pathological process of SCF. Thus, the present study could stimulate further studies on coronary vasculature to discover more active roles of Galectin-3 in the SCF p
Keywords
Slow coronary flow; Coronary arteries; Coronary vasculature; Inflammation
Slow coronary flow articles; Coronary arteries articles; Coronary vasculature articles; Inflammation articles
Article Details
1. Introduction
Coronary arteries could be opacified in the inner layer of pericardium in a time-lag, despite lack of obstruction from a coronary disease, which is the slow coronary flow (SCF) phenomenon observed on angiographic images [1]. The prevalence of SCF is low in the angiographic reports by about 1% of the cases suspected of coronary artery disease (CAD) [2]. Originated in 1972 [1] SCF has been not much studied with a limited number of reports, and hence the current evidence is poor about clinical implications and clear pathophysiological processes.
On the other hand, several mechanisms have been suggested including small vessel disease, diffuse atherosclerosis, microvascular vasomotor dysfunction, and inflammation as well as endothelial dysfunction [3-6].
Another proposition is that occlusive disease of the small coronary arteries, a type of acute atherosclerosis, could be a development factor of SCF [7]. In fact, the risk of transient myocardial hypoperfusion is higher with SCF among the patients with angina problem and without coronary artery disease; in addition, these patients are significantly under the threat of CAD because of limited prognosis [8].
The critical processes to deal with acute and chronic inflammation can be regulated centrally by Galectin-3. This regulator is enable the body to make a response to acute inflammation including chemoattraction of monocytes/macrophages [9], neutrophil clearance [10], opsonization of apoptotic neutrophils [11], and mast cell degranulation [12]. Chronic inflammation is typically accompanying with fibrosis, loss of tissue structure, and subsequent organ failure, which lays heavy burden on global healthcare systems.
With the activation of Galectin-3, inflammatory agents are secreted to aggravate inflammation by fibroblasts, the key cells to initiate and perpetuate tissue fibrogenesis [13]. Endothelial cells (ECs) are vital to maintain vascular homeostasis for responses to many stimuli.
Chronic inflammation paves the way for variations in normal endothelial function due to longer activation of ECs by inflammatory stimuli and thus for endothelial dysfunction developing atherosclerosis, the most prevalent factor of cardiovascular diseases [14, 15]. Research have demonstrated that endothelial cells are injured with oxidized low-density lipoprotein (ox-LDL) modifying proinflammatory gene expression [16, 17].
The overall evidence has showed that Galectin-3 induces inflammation to increase the ox-LDL-mediated endothelial damage [18, 19]. Focal inflammation by Ox-LDL leads to endothelial dysfunction and then greater expression of atherogenic signaling molecules that deteriorate the monocyte adhesion to the arterial endothelium and the monocyte penetration into the intima.
The current study might be the first research in terms of its design in the literature. The study results support that Galectin-3 is a key factor of inflammation and endothelial dysfunction and thus development of vascular diseases. Consequently, it can not be rejected that Galectin-3 may contribute to SCF phenomenon.
2. Methods
2.1 The study design
Our study design was arranged in an observational and cross-sectional structure.
2.2 Study population
The study group consisted of 94 with SCF (slow coronary flow), and the control of 92 with NCF (normal coronary flow), and both totaled 186 cases. All of the cases were selected from the outpatient ones undergoing the coronary angiography with suspicion of CAD from May 2018 to March 2020 who presented in our services. Their normal coronary arteries varied by coronary flow rates and were clean from atherosclerotic lesions. The individuals had a story of chest pain or indicative signs of angina with application of his/her myocardial perfusion or treadmill test. It was confirmed that all the cases indicated cardiac syndrome-X: (1) They experienced angina caused by single exercises and however it was not so abnormal as that of CAD; (2) In the cases of randomly occurring or elicited angina, they indicate coronary flow anomalies or myocardial ischemia; (3) Their angiographic reports showed normal coronary arteries; (4) There was no sign of any typical cardiac disease.
All the patients underwent cardiac catheterization following physical examination and medical history acquisition by a cardiologist. The collected data are retained in the coronary angiography laboratory at the hospital. The disease history including the peripheral vascular or coronary-based was identified as cause of exclusion, as well as ectatic coronary arteries, non-ischemic dilated cardiomyopathy, renal or hepatic dysfunctions, chronic inflammation or infection, hematological dysfunctions, and any malignancies.
It was ensured that none of the cases in this study took any sort of vasoactive prescription drug. The current study was in compliance with the Helsinki Declaration. Before the start, all the patients signed the informed consent forms.
2.3 Biochemical analysis
Preprandial blood glucose tests were conducted after a half-day fasting. Serum samples were centrifuged at 4000 × g for 10 minutes to decompose and frozen at -80°C before the next treatment. Complete blood count was estimated to identify differentials using the blood samples from the cases at admission.
The measurements were made by an automatized analyzer for high sensitivity C-reactive protein (hs-CRP), total cholesterol, triglycerides, creatinine, and low (LDL) and high (HDL) density lipoprotein cholesterol levels. ELISA was a useful tool to determine the values of Galectin-3.
2.4 Diagnostic analysis of the coronary arteries for SCF based on angiography
In the Judkins method the cases underwent the coronary angiography through the femoral artery way. The Ultravist 370 iopromide (Schering AG, Berlin, Germany) was administered to each patient to achieve contrasting. The cardiologists make SCF assessments for all the cases based on angiographical reports. TFC measured the individual’s coronary flow rate [20]. For objectivity, the coronary flow rate was estimated by two independent clinicians who were blind to the patient’s clinical records. The characteristics of TFC is the requirement of the amount of cine frames (taken at 30 frame/s) and hence the contrast can be achieved at the level of the normal distal coronary landmarks for the arteries of left circumflex (LCX), left anterior descending (LAD), and right coronary arteries (RCA).
The distal bifurcation for the LAD, usually represented by the ‘pitchfork’or ‘whale’s tail’, the distal bifurcation of the segment with the longest total distance for the LCX, and the first branch of the posterolateral artery for the RCA were already specified as a distal landmark. For typical coronary arteries the standard means are 36.2 ± 2.6, 22.2 ± 4.1 and 20.4 ± 3 frames respectively. The longest TFC for the LAD yields mostly the highest value among the coronary arteries; it is necessary to divide it by 1.7. Its corrected value is estimated as 21.1 ± 1.5 frames. In our study, significance level for diagnosis was defined as more than 2 s.d. of the reported range for each artery. To calculate the mean value the addition of TFC values for the three coronary arteries was divided by 3. Quantitative coronary angiography (QCA) produces the vessel lengths and ostial diameters of LCX, LAD, and RCA.
2.5 Statistics
Statistical outcomes were obtained using SPSS software (version 20.0 for Windows, Inc., Chicago, Illinois, USA). Descriptive statistics were arranged as mean and standard deviation for continuous variables and percentage for categorical ones. Chi-square test was used for the analysis of the obtained data. Normal distribution was determined by Kolmogorov-Smirnov test. If normally distributed, univariate analysis was modeled with two sample T test, and if not, with Mann Whitney U test. The independent risk factors of SCF were identified through logistic regression analysis. Two-tail tests were used at a 95% significance level (p<.05; 0.025 from the right and 0.025 from the left).
3. Results
3.1 Clinical characteristics
Table 1 presents the key clinical parameters from all the cases. The parameters of gender, age, presence of hypertension, history of smoking, and presence of diabetes yields no significant inter-group difference (p>.05) as well as preprandial blood glucose levels and lipid parameters (p>.05).
|
Variables |
SCF (n=94) |
NCF (n=92) |
*p |
|
Age, years |
52 ± 10 |
50 ± 13 |
0.74 |
|
Male gender, n |
48 (%51) |
46 (%50) |
0.88 |
|
BMI, kg/m2 |
30 ± 8 |
27 ± 7 |
0.72 |
|
Hypertension, n |
28 (%30) |
24 (%26) |
0.52 |
|
Diabetes mellitus, n |
32 (%34) |
28 (%30) |
0.54 |
|
Hyperlipidemia, n |
20 (%21) |
17 (%18) |
0.60 |
|
Cigarette smoking, n |
38 (%40) |
34 (%37) |
0.68 |
|
Family history of CAD, n |
18 (%19) |
16 (%17) |
0.78 |
|
Fasting glucose, mg/dL |
114 ± 32 |
101 ± 17 |
0.72 |
|
Total cholesterol, mg/dL |
201 ± 41 |
193 ± 17 |
0.76 |
|
Triglycerides, mg/dL |
152 ± 72 |
131 ± 25 |
0.48 |
|
HDL cholesterol, mg/dL |
38 ± 9 |
41 ± 11 |
0.74 |
|
LDL cholesterol, mg/dL |
113 ± 22 |
124 ± 33 |
0.80 |
|
hsCRP, mg/dL |
2.4 ± 0.6 |
1.1± 0.2 |
0.005 |
|
Galectin-3, ng/mL |
16.6 ± 0.7 |
9.2 ± 0.6 |
0.003 |
|
TIMI frame count measurements |
|||
|
LAD |
62 ± 19 |
31 ± 9 |
<0.001 |
|
LAD(corrected) |
37 ± 21 |
19 ± 6 |
<0.001 |
|
LCx |
31 ± 17 |
29 ± 4 |
<0.001 |
|
RCA |
43 ± 21 |
21 ± 7 |
<0.001 |
|
The length of epicardial coronary arteries |
|||
|
LAD,mm |
172 ± 19 |
172 ± 21 |
0.78 |
|
LCx,mm |
130 ± 27 |
127 ± 33 |
0.74 |
|
RCA,mm |
187 ± 31 |
170 ± 22 |
0.62 |
|
Diameters of coronary arteries |
|||
|
LAD,mm |
3.98 ± 0.68 |
3.59 ± 0.53 |
0.026 |
|
LCx,mm |
3.64 ± 0.69 |
3.22 ± 0.74 |
0.044 |
|
RCA,mm |
3.78 ± 0.83 |
3.06 ± 0.68 |
0.003 |
|
Coronary flow velocities |
|||
|
LAD,mm/s |
104.8 ± 47.7 |
180.6 ± 57.0 |
<0.001 |
|
LCx,mm/s |
149.7 ± 61.3 |
202.7 ± 61.5 |
<0.001 |
|
RCA,mm/s |
167.9 ± 74.6 |
279.4 ± 76.8 |
<0.001 |
Data are presented as mean ± SD, median (inter quartile range) and as number (per-centage).*Student’s t-test, Mann-Whitney U test and Chi-square test; BMI- body mass index, HDL-high density lipoprotein, LAD-left anterior descending artery, LCx-left circumflex artery, LDL-low-density lipoprotein, NCF-normal coronary flow, RCA-right coronary artery, SCF-slow coronary flow, hsCRP: high sensitivity C-reactive protein
Table 1: Demographic and clinical characteristics of study participants.
|
Variables |
Slow coronary flow (Dependent variable) |
Variables |
|
OR ( 95%CI) |
*p |
|
|
Galectin-3, ng/mL |
1.044 (1.006-1.084) |
0.023 |
|
Mean coronary diameter |
7.412 (1.996-29.60) |
0.003 |
|
hsCRP |
1.55 (1.00-2.39) |
0.038 |
*Logistic regression analysis; OR: Odds ratio, hsCRP: high sensitivity C reactive protein.
Table 2: The independent variables with slow coronary flow phenomenon.
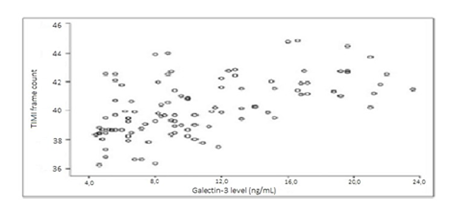
3.2 TFC
As expected, the study group had significantly higher TFC values for all the three arteries of interest (p< .001, r = 0.478 to p > .05, r = -0.382) (see Figure 1). Furthermore, the SCF patients’ hsCRP values were proportionally associated with mean TFC values (p < .001, r = 0.646 to p >.05, r = -0.181; p <.001, r = 0.592 for all].
3.3 Correlations between Galectin-3 and hsCRP with SCF
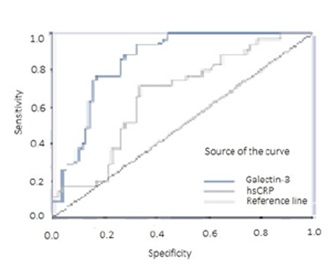
The mean values of serum hsCRP and Galectin-3 were significantly higher in the SCF patients compared to the control group (p=0.005, 2.4 ± 0.6 to 1.1 ± 0.2 mg/dL for hsCRP; 16.6 ± 0,7 to p=.003 9.2 ± 0,6 ng/mL, for Galectin-3). Logistic regression model estimated the SCF condition solidly from mean coronary diameter (OR:7.412, CI: 1.996-29.60, p=.003) and weakly from Galectin-3 (OR: 1.044, CI: 1.006-1.084, p=.023) and hsCRP (OR: 1.55, CI: 1.00-2.39, p=.038 ) (see Table 2). A receiver operating characteristic (ROC) curve helped to determine the sensitivity and specificity of hsCRP and Galectin-3 for diagnosis of SCF.
As shown in Figure 2, plasma Galectin-3 was a significant precursor of SCF (AUC = 0.823, CI: 0.722–0.896, p< .001) and a key determinant compared to plasma hsCRP (AUC = 0.587, CI: 0.533–0.632, p= .010).
4. Discussion
In this study, the SCF cases had significantly higher values of Galectin-3 than those with NCF, as well as hsCRP, which is a marker of inflammation. Furthermore the SCF rates were strongly associated with systemic inflammation. To the best of our knowledge, our study is unprecedented in showing the correlation of Galectin-3 with SCF.
Inflammation mediates an atherosclerotic SCF condition. Any type of mediator cells escalate proinflammatory signals and keep them high, resulting in the development of SCF. It has been reported that several cardiac problems are mostly derived from inflammation, as well as a variety of different clinical conditions of CAD. More research has revealed that chronic inflammation have great part in cardiovascular conditions [21, 22]. Furthermore, it has been reported that endothelial cells may activate to imitate and sustain atherosclerotic diseases in addition to inflammation [23-25].
Most of epidemiological research has studied Galectin-3 since it is a novel biomarker of cardiovascular diseases. Several studies [26, 27] have revealed that the quantity of plasma Galectin-3 concentration could be an efficient biomarker of atherosclerotic diseases. However, the epidemiological studies on Galectin-3 and atherosclerosis have focused heavily on heart failure and CAD in recent years. This independent risk factor could develop a cardiovascular disease. In a 10-year cohort study of 7968 participants it was showed that the cases with high levels of Galectin-3 was inclined to cardiovascular disease [28]. For older patients (mean age: 69 years) with low Galectin-3 the cardiovascular risk was remarkably low based on the follow-up controls for 2.7 years [29].
Madrigal-Matute et al. suggested that there was a positive relationship between Galectin-3 plasma level and carotid intimamedia thickness as Galectin-3 level escalated in the carotid atherosclerosis patients [30]. Pusuroglu et al. and Ozturk et al. reported that Galectin-3 increased with more severe carotid atherosclerosis in such groups having obstructive sleep apnea syndrome or Type II diabetes mellitus [31, 32]. In recent cohort studies it has been showed with univariate analysis that serum Galectin-3 levels had a positive association with carotid intima-media thickness which is the marker of subclinical atherosclerosis, in relatively well-controlled rheumatoid arthritis (RA) patients with long-standing disease and low levels of systemic inflammation and concluded that Galectin-3 could be potentially used as a biomarker for subclinical cardiovascular disease in these patients [33]. There is further evidence that Galectin-3 is one of the key independent precursors for coronary atherosclerosis [34].
Much research revealed that Galectin-3 has potential as a strong biomarker for coronary heart diseases excluding heart failure. In a study of Falcone, among 125 coronary artery disease patients with unstable angina and stable angina, Galectin-3 level significantly increased in the study group with unstable angina and further was higher in the cases with three pieces of CAD compared to one or two pieces [35]. There is another report that it is a risk factor for unstable angina [36]. Further research suggested that higher circulating level of Galectin-3 is a potential, free precursor of the combined 30- day major adverse clinical outcome in the cases with STsegment elevation myocardial infarction (MI) and who underwent primary percutaneous coronary operation [37].
An investigation of a community cohort of incident MI patients proposed that higher Galectin-3 is associated with higher risk of mortality and heart failure from the adjusted data in age, sex, comorbidities, and troponin and that Galectin-3 levels could be usefol for risk evaluation of post-MI period [38]. In addition, Szadkowska et al. showed that Galectin-3 (particularly 18.1 ng/mL) was an independent risk factor also for reinfarction in MI patients after the surgery [39]. In a prospective study including 782 coronary patients, the low levels of Galectin-3 were more prognostic than the high levels [40]. Furthermore, when both Galectin-3 and carotid intima-media thickness are detected the clinician could simply assess the CAD [41].
Our study showed the correlation of TFC with hsCRP. The reason could be why hsCRP is an inflammatory marker for development of atherosclerosis and the related coronary flow patterns [42]. In angiogenic microvessels from unhealthy tissues the contribuiton of CRP has been clarified in a report [43]. Also based on the literature review above, the present study indicates that inflammatory dysfunctions might be caused by dysregulated endothelial cells, which refers to the SCF. Thus, hypothesis was identified that inflammation did not stop by looking at higher Galectin-3 levels in SCF patients. At last the hypothesis was confirmed and the plasma Galectin-3 and other inflammatory marker, hsCRP were significantly related to each other. An further analysis of the ROC curve also discovered that Galectin-3 levels can be independent estimator of SCF. Moreover, we concluded here that hsCRP and Galectin-3 indicate comparative outcomes for diagnosis of SCF.
5. Limitations of the Study
We believe that the present study would make significant contributions to the literature even though there existed some limitations during the study. The first is of sample size. We consider that a greater number of patients could strengthen the statistical analysis and the results of the study. Additionally, it should be noted that our study design is observational so that we could not identify a simple method to determine the association of Galectin-3 upregulation and SCF. Further, the study group was not subjected to IVUS and thus the results are not clear from the suspicion of whether ‘isolated’ SCF was combined with non-obstructive CAD.
This may be a handicap to capture atherosclerotic variations in the coronary arteries, whereas such an independence is not only seen in the sample of study but also in the population of typical patients with SCF; namely, these patients undergo frequent controls including angiographic SCF tests. Lastly, the control group consisted of the cases having sort of cardiac risks including cardiac syndrome-X though their angiography reports indicated normal coronary arteries. We may have omitted variations of both groups that may also be statistically significant. Except for all these limitations, we have drawn up critical observations on the relationship of Galectin-3 with SCF.
References
- Tambe AA, Demany MA, Zimmerman HA, et al. Angina pectoris and slow flow velocity of dye in coronary arteries--a new angiographic finding. Am Heart J 84 (1972): 66-71.
- Goel PK, Gupta SK, Agarwal, et al. Slow coronary flow: a distinct angiographic subgroup in syndrome X. Angiology 52 (2001): 507-514.
- Bolayir A, Cigdem B, Gokce SF, et al. The Effect of Eosinopenia on Mortality in Patients with Intracerebral Hemorrhage. Journal of Stroke and Cerebrovascular Disease 10 (2017): 2248-2255.
- Bolayir A, Gokce SF, Cigdem B, et al. Monocytes/high-density lipoprotein ratio predicts the mortality in ischemic stroke. Neurol Neurochir Pol (2017).
- Sezgin AT, Sigirci A, Barutcu I, et al. Vascular endothelial function in patients with slow coronary flow. Coron Artery Dis 14 (2003): 155-161.
- Riza Erbay A, Turhan H, Yasar AS, et al. Elevated level of plasma homocysteine in patients with slow coronary flow. Int J Cardiol 102 (2005): 419-423.
- Mosseri M, Yarom R, Gotsman MS, et al. Histologic evidence for small-vessel coronary artery disease in patients with angina pectoris and patent large coronary arteries. Circulation 74 (1986): 964-972.
- Fragasso G, Chierchia SL, Arioli F, et al. Coronary slow-flow causing transient myocardial hypoperfusion in patients with cardiac syndrome X: long-term clinical and functional prognosis. Int J Cardiol 137 (2009): 137-144.
- Subhash VV, Ling SSM, Ho B. Extracellular galectin-3 counteracts adhesion and exhibits chemoattraction in Helicobacter pylori-infected gastric cancer cells, Microbiology 162 (2016): 1360-1366.
- Wright RD, Souza PR, Flak MB, Thedchanamoorthy P, Norling LV, et al. Galectin-3-null mice display defective neutrophil clearance during acute inflammation, Journal of Leukocyte Biology 101 (2017): 717-726.
- Karlsson A, Christenson K, Matlak M, et al. Galectin-3 functions as an opsonin and enhances the macrophage clearance of apoptotic neutrophils, Glycobiology 19 (2009): 16-20.
- Bambouskova M, Polakovicova I, Halova I, et al. New regulatory roles of galectin-3 in high-affinity IgE receptor signaling, Molecular and Cellular Biology 36 (2016): 1366- 1382.
- Filer A, Bik M, Parsonage GN, et al. Galectin 3 induces a distinctive pattern of cytokine and chemokine production in rheumatoid synovial fibroblasts via selective signaling pathways, Arthritis and Rheumatism 60 (2009): 1604-1614.
- Davignon J, Ganz P. Role of endothelial dysfunction in atherosclerosis, Circulation 109 (2004): III27-III32.
- Tabas I, García-Cardeña G, Owens GK. Recent insights into the cellular biology of atherosclerosis, Journal of Cell Biology 209 (2015): 13-22.
- Li JB, Wang HY, Yao Y. et al. Overexpression of microRNA-138 alleviates human coronary artery endothelial cell injury and inflammatory response by inhibiting the PI3K/Akt/eNOS pathway, Journal of Cellular and Molecular Medicine 21 (2017): 1482-1491.
- Zhang S, Guo C, Chen Z, Zhang P, Li J, et al. Vitexin alleviates ox-LDL-mediated endothelial injury by inducing autophagy via AMPK signaling activation, Molecular Immunology 85 (2017): 214-221.
- Chen X, Lin J, Hu T, et al. Galectin-3 exacerbates ox-LDLmediated endothelial injury by inducing inflammation via integrin β1-RhoA-JNK signaling activation, Journal of Cellular Physiology 234 (2019): 10990-11000.
- Ou HC, Chou WC, Hung CH, et al. Galectin-3 aggravates ox-LDL-induced endothelial dysfunction through LOX-1 mediated signaling pathway, Environmental Toxicology 34 (2019): 825-835.
- Gibson CM, Cannon CP, Daley WL, et al. TIMI frame count: a quantitative method of assessing coronary artery flow. Circulation 93 (1996): 879-888.
- Sullivan GW, Sarembock IJ, Linden J. The role of inflammation in vascular disease. J Leukoc Biol 67 (2000): 591-602.
- Gokce M, Kaplan S, Tekelioglu Y, et al. Platelet function disorder in patients with coronary slow flow. Clin Cardiol 28 (2005): 145-148.
- Camsari A, Pekdemir H, Cicek D, et al. Endothelin-1 and nitric oxide concentrations and their response to exercise in patients with slow coronary flow. Circ J 67 (2003): 1022-1028.
- Yildiz A, Gur M, Yilmaz R, et al. Association of paraoxonase activity and coronary blood flow. Atherosclerosis 197 (2008): 257-263.
- Enli Y, Turk M, Akbay R, et al. Oxidative stres parameters in patients with slow coronary flow. Adv Ther 25 (2008): 37-44.
- Salvagno GL, Pavan C. Prognostic biomarkers in acute coronary syndrome, Annals of Translational Medicine 4 (2016): 258.
- Casanegra AI, Stoner JA, Tafur AJ, Pereira HA, Rathbun SW, et al. Differences in galectin-3, a biomarker of fibrosis, between participants with peripheral artery disease and participants with normal ankle-brachial index, Vascular Medicine 21 (2016): 437-444.
- de Boer RA, van Veldhuisen DJ, Gansevoort RT, et al. The fibrosis marker galectin-3 and outcome in the general population, Journal of Internal Medicine 272 (2012): 55-64.
- Mortensen MB, Fuster V, Muntendam P, et al. Negative risk markers for cardiovascular events in the elderly, Journal of the American College of Cardiology 74 (2019): 1-11.
- Madrigal-Matute J, Lindholt JS, Fernandez-Garcia CE, et al. Galectin-3, a biomarker linking oxidative stress and inflammation with the clinical outcomes of patients with atherothrombosis, Journal of the American Heart Association 3 (2014): e000785.
- Pusuroglu H, Somuncu U, Bolat I, et al. Galectin-3 is associated with coronary plaque burden and obstructive sleep apnoea syndrome severity, Kardiologia Polska 75 (2017): 351-359.
- Ozturk D, Celik O, Satilmis S, et al. Association between serum galectin-3 levels and coronary atherosclerosis and plaque burden/structure in patients with type 2 diabetes mellitus, Coronary Artery Disease 26 (2015): 396-401.
- Anyfanti P, Gkaliagkousi E, Gavriilaki E, et al. Association of galectin-3 with markers of myocardial function, atherosclerosis, and vascular fibrosis in patients with rheumatoid arthritis, Clinical Cardiology 42 (2019): 62-68.
- Aksan G, Gedikli O, Keskin K, et al. Is galectin-3 a biomarker, a player-or both-in the presence of coronary atherosclerosis, Journal of Investigative Medicine 64 (2016): 764-770.
- Falcone C, Lucibello S, Mazzucchelli I, et al. Galectin-3 plasma levels and coronary artery disease: a new possible biomarker of acute coronary syndrome, International Journal of Immunopathology and Pharmacology 24 (2011): 905-913.
- Higueras J, Martin-Ventura JL, Blanco-Colio L, et al. Impact of plasma pro-B-type natriuretic peptide aminoterminal and galectin-3 levels on the predictive capacity of the LIPID clinical risk Scale in stable coronary disease, Clínica e Investigación en Arteriosclerosis 27 (2015): 57-63.
- Tsai TH, Sung PH, Chang LT, et al. Value and level of galectin-3 in acute myocardial infarction patients undergoing primary percutaneous coronary intervention, Journal of Atherosclerosis and Thrombosis 19 (2012): 1073-1082.
- Asleh R, Enriquez-Sarano M, Jaffe AS, et al. Galectin3 levels and outcomes after myocardial infarction: a population-based study, Journal of the American College of Cardiology 73 (2019): 2286-2295.
- Szadkowska I, Wlazel RN, Migala M, et al. The association between galectin-3 and occurrence of reinfarction early after first myocardial infarction treated invasively, Biomarkers 18 (2013): 655-659.
- Maiolino G, Rossitto G, Pedon L, et al. Galectin-3 predicts long-term cardiovascular death in high-risk patients with coronary artery disease, Arteriosclerosis, Thrombosis, and Vascular Biology 35 (2015): 725-732.
- Lisowska A, Knapp M, Tycinska A, et al. Predictive value of Galectin-3 for the occurrence of coronary artery disease and prognosis after myocardial infarction and its association with carotid IMT values in these patients: a mid-term prospective cohort study, Atherosclerosis 246 (2016): 309- 317.
- Pekdemir H, Cin VG, Çiçek D, et al. Slow coronary flow may be a sign of diffuse atherosclerosis: contribution of FFR and IVUS. Acta Cardiol 59 (2004): 127-133.
- Slevin M, Krupinski J. A role of C-reactive protein in the regulation of angiogenesis, endothelial cell inflammation and thrombus formation in cardiovascular disease. Histol Histopathol 24 (2009): 1473- 1478.