Taffix® Nasal Powder Spray Forms an Effective Barrier Against Infectious New Variants of SARS-Cov-2 (Alpha, Beta and Delta)
Article Information
Michal Mandelboim1, 2, Ella Mendelson1, 2, Yaron Drori1, 2, Nofar Atari1, Tair Lapidot3*, Dalia Megiddo3*, Micha Gladnikoff4
1Central Virology Laboratory, Ministry of Health, Chaim Sheba Medical Center, Ramat Gan, Israel
2School of Public Health, Sackler Faculty of Medicine, Tel-Aviv University, Tel-Aviv, Israel
3Nasus Pharma, Tel Aviv, Israel
4Smart Assays Biotechnologies Ltd., Nes Ziona, Israel
*Corresponding author(s): Tair Lapidot, Nasus Pharma, Tel Aviv, Israel
Dalia Megiddo, Nasus Pharma, Tel Aviv, Israel
Received: 25 August 2021; Accepted: 06 September 2021; Published: 11 October 2021
Citation: Michal Mandelboim, Ella Mendelson, Yaron Drori, Nofar Atari, Tair Lapidot, Dalia Megiddo, Micha Gladnikoff. Taffix® Nasal Powder Spray Forms an Effective Barrier Against Infectious New Variants of SARS-Cov-2 (Alpha, Beta and Delta). Archives of Clinical and Biomedical Research 5 (2021): 794-802.
Share at FacebookAbstract
While vaccination efforts against SARS-CoV-2 around the world are ongoing, new highly infectious virus variants continue to evolve. The protection provided by the available vaccines against some of the new variants is weaker. Additional preventive measures will therefore be needed to protect the population until effective vaccinations are widely available. TaffiX® is an anti-viral nasal powder spray comprised of low-pH hypromellose, which forms a protective mechanical barrier that prevents viruses from engaging with nasal cells. The current study aimed to test the protective effect of TaffiX® against SARS-CoV-2 Alpha (B.1.1.7; hCoV-19/Israel/CVL-46879-ngs/ 2020), Beta, (B.1.351; hCoV-19/Israel/CVL-2557-ngs/2020) and Delta (B.1.617.2; hCoV-19/Israel/ VVL-12806/2021), three highly infectious and pathogenic SARS-CoV-2 strains.
A nylon filter was treated with Taffix® gel, after which SARS-CoV-2 Alpha, Beta or Delta was seeded. After a 10-min incubation, the downstream side of each filter was washed, and the rinse was collected and placed over Vero-E6 cells. After 5 days of incubation, viral RNA was extracted and subjected to SARS-CoV-2 RT-PCR analysis. Taffix® fully blocked passage of all three tested SARS-CoV-2 variants, as demonstrated by a 100% reduction of recoverable viral RNA from Vero-E6 cells, as compared to cells treated with rinse from filters not treated with Taffix(R). These results support its use as an effective barrier against new variants of SARS-CoV-2 in conjunction with other protective measures.Keywords
CoVID-19; SARS-CoV-2; Variant B.1.1.7; Variant B.1.351; Variant B.1.617.2; Variant Alpha; Variant Beta; Variant Delta; Taffix; Nasal Spray
CoVID-19 articles; SARS-CoV-2 articles; Variant B.1.1.7 articles; Variant B.1.351 articles; Variant B.1.617.2 articles; Variant Alpha articles; Variant Beta articles; Variant Delta articles; Taffix; Nasal Spray articles
Article Details
1. Introduction
While vaccination efforts against SARS-CoV-2 around the world are ongoing, new highly infectious virus variants continue to evolve. The protection conferred by the available vaccines against some of the new variants is weaker [1], and experts are concerned that newer as well as yet undescribed variants of this RNA virus will eventually prove resistant to the current vaccines [2, 3]. Additional preventive measures will therefore be needed to protect the population until effective vaccinations become available. It is now well established that the nasal epithelium, specifically goblet and cilia cells, is the primary portal of entry of SARS-CoV-2 into the body, and protection at the nasal cell level could therefore be meaningful in reducing the risk of infection [4-7]. Taffix® is an anti-viral nasal powder spray comprised of low-pH hypromellose that upon insufflation into the nose, creates a thin gel layer over the nasal mucosa, forming a protective mechanical barrier, stable for at least 5 hours, that prevents viruses from engaging with nasal cells [8]. The acidity of the gel enhances the barrier by inactivating the viruses trapped within it. Taffix® is fully biocompatible according to the requirements of the current ISO 10993-1, ISO 14971, and FDA Biocompatibility Guidance (reports available upon request). It is commercially available in many countries across Europe, Asia, America and Africa. In a preclinical study testing its barrier performance when applied to a nylon filter, Taffix® effectively prevented passage of SARS-CoV-2 Hong Kong/VM20001061/2020 through the filter, as measured by plaque assay and viral RNA assessment of the flow-through [8].
In addition, in a prospective two-week user survey completed by 243 individuals participating in a two-day mass-gathering event in a highly endemic community, none of the 81 individuals who regularly applied Taffix® nasal spray during the event and the subsequent two weeks reported on SARS-CoV-2 infection [9]. In contrast, of the 18 individuals who tested SARS-CoV-2-positive in the subsequent two weeks, 16 (98%) did not use Taffix® and the remaining two individuals reported using it once or twice in the study period. No side effects were reported. The present in vitro study aimed to assess the protective effect of Taffix® against new pathogenic, highly infectious SARS-CoV-2 variants [10, 11], i.e., the Alpha (B.1.1.7; hCoV-19/Israel/CVL-46879-ngs/2020), Beta (B.1.351; hCoV-19/Israel/CVL-2557-ngs/2020) and Delta (B.1.617.2; hCoV-19/Israel/VVL-12806/2021) variants.
2. Materials and Methods
2.1 Materials
Alpha virus stock was isolated from patients in Israel on 20 December 2020. Beta virus stock was isolated from patients in Israel on 15 January 2021. Delta virus stock was isolated from patients in Israel on 20 April 2021. Cell strainers (40 µm Cat# SO SCS402) were obtained from AlexRed, cell culture medium (MEM-EAGLE) was obtained from Biological Industries Israel Beit Haemek Ltd., Vero-E6 cells were obtained from HTCC and TaffiX® was supplied by Nasus Pharma Ltd.
2.2 Virus propagation
Vero-E6 cells were grown at 37 °C, 5% CO2 in MEM-Eagle Earle's salts Base containing 50 IU/ml penicillin and 50 µg/ ml streptomycin (Biological Industries, Israel) and 10% heat-inactivated fetal calf serum (FCS) (Biological Industries, Israel). Confluent Vero-E6 cells were incubated for 1 h at 33°C with 300 µl of the nasopharyngeal samples stored in Copan Universal Transport Medium (UTM-RT, Italy), after which, 5 ml MEM-EAGLE medium supplemented with 2% FC were added. The infected cells were cultured for 3-10 days. When a cytopathic effect (structural changes in host cells) was observed, 300µl of the supernatant was added to T-75 flasks seeded with 10x106Vero-E6 cells, to which 20 ml 2% FCS MEM-EAGLE were immediately added. Samples were incubated at 33°C for 3-5 days, in order to reach higher viral loads. When a cytopathic effect was microscopically observed, supernatants were aliquoted and stored at -80°C.
2.3 Taffix® assay
Sterile water (150 ml) was placed over sterile 40 mm cell strainers placed on a 50 ml tube. Taffix® (20 mg) was then added and mixed with the water with a pipette tip until a homogeneous layer formed across the cell strainer surface. Filters were then incubated at room temperature for 10 min. Control filters were incubated with 150 ml sterile water only. Thereafter, 10 µl of SARS-CoV-2 Alpha, Beta or Delta variants were seeded on the filters at a concentration CT 20 (approximately ~600,000 copies/ml) for Alpha and Beta, and CT 21 (approximately ~300,000 copies/ml) for Delta, which is parallel to the clinical concentrations found in nasal swabs of patients diagnosed positive for SARS-CoV-2 (COVID-19).
After a 10-min incubation at room temperature, the downstream side of each filter was washed with 500 µl cell culture medium, which was subsequently collected and diluted 1:10 with culture medium. A sample was taken from the rinse solution of the Delta variant for determination of viral count using qRT-PCR. Then, 50 µl of the diluted medium was placed into 24-well plates containing Vero-E6 cells (2x105 cells/well, in 2% fetal calf serum). After 5 days of incubation at 33°C (5% CO2), a 200 µl sample from each well was taken for viral RNA extraction followed by qRT-PCR analysis. Each treatment was repeated in duplicates for Alpha and Beta variants and in triplicates for the Delta variant.
2.4 Quantitative reverse transcriptase PCR
Nucleic acids were extracted using the MagLead extractor (PSS, Japan), according to the manufacturer’s instructions. Reverse transcription polymerase chain reactions (RT-PCRs) using primers corresponding to the SARS-CoV-2 envelope (E) gene, were performed as previously described [10]. Quantitative RT-PCR (qRT-PCR) was performed in 25 μl SensiFast reaction mix (Bioline, USA ) using TaqMan Chemistry on a CFX-96 instrument.
3. Results
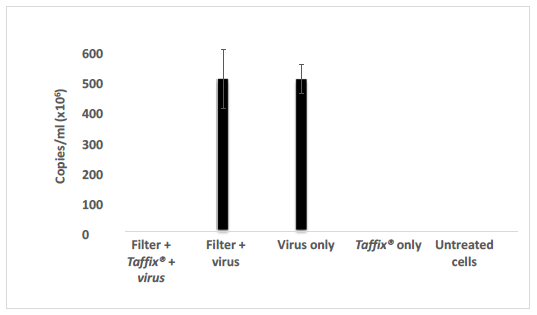
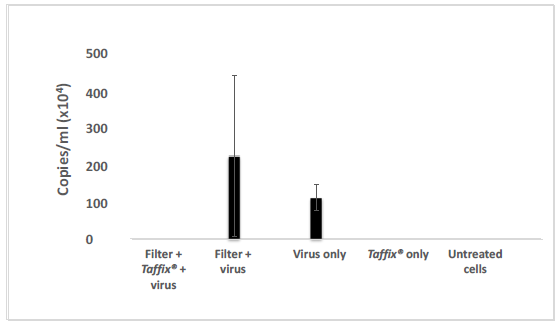
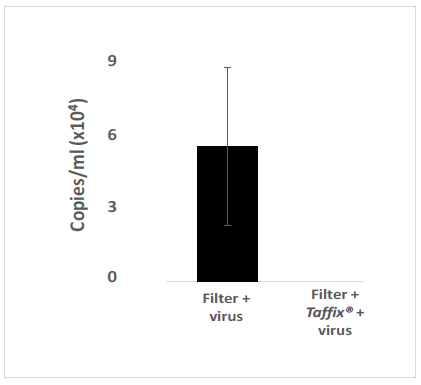
The Taffix® layer applied to cell strainers fully (100%) protected Vero-E6 cells from infection by the Alpha, Beta and Delta SARS-CoV-2 variants. Initial concentrations of approximately 300,000 to 600,000 copies/ml, which is equivalent to a clinical PCR result of CT21 to CT20, typically found in COVID-19 positive nasal swabs, were applied to the filter, with or without the Taffix® layer. No infection (zero) was diagnosed in Vero-E6 cells incubated with the solution used to rinse the downstream side of the Taffix®-coated filter. In contrast, Vero-E6 cells treated with medium used to rinse filters without Taffix®, showed very high Alpha (2,236,550 copies/ml; Figure 1), Beta (506,578,602 copies/ml (Figure 2) and Delta (54,583 copies/ml (Figure 3) virus copy numbers. The number of Delta viral RNA copies in the rinse solution eluted from Taffix®-treated filters was found to be 22.3-fold lower than in unprotected filters (60 copies/ml vs. 1338 copies/ml, respectively).
Figure 1: Taffix® prevented infection of Vero-E6 cells by the Alpha B.1.1.7 SARS-CoV-2 variant. Vero-E6 cells were incubated with inoculum of SARS-CoV-2 variant Alpha B.1.1.7 at a concentration of ~600,000 copies/ml (which is parallel to a clinical PCR results of CT20), which had been passed through an untreated cell strainer filter or through one pretreated with Taffix®. Total RNA was extracted from the growth medium 5 days later for qPCR analysis. Viral copies were calculated from qPCR CT values. For each test, a positive sRNA control and negative control were used. The results are average of duplicates.
Figure 2: Taffix prevented infection of Vero-E6 cells by the Beta B.1.351 SARS-CoV-2 variant. Vero-E6 cells were grown and incubated with inoculum of SARS-CoV-2 variant Beta B.1.351 at a concentration of ~600,000 copies/ml (which is parallel to a clinical PCR result of CT20), which had been passed through an untreated cell strainer filter or through one pretreated with Taffix®. Total RNA was extracted from the growth medium 5 days later for qPCR analysis. Viral copies were calculated from qPCR CT values. For each test, a positive sRNA control and negative control were used. The results are average of duplicates.
Figure 3: Taffix prevented infection of Vero-E6 cells by the Delta B.1.617.2 SARS-CoV-2 variant. Vero-E6 cells were grown and incubated with inoculum of SARS-CoV-2 variant Delta B.1.617.2 at a concentration of ~300,000 copies/ml (which is parallel to a clinical PCR result of CT21), which had been passed through an untreated cell strainer filter or through one pretreated with Taffix®. Total RNA was extracted from the growth medium 5 days later for qPCR analysis. Viral copies were calculated from qPCR CT values. For each test, a positive sRNA control and negative control were used. The results are average of triplicates.
4. Discussion
In a prior in vitro study, Taffix® nasal spray powder formed a protective barrier limiting entry of SARS-COV-2 Hong Kong/VM20001061/2020 and preventing >99.9% of viruses from infecting Vero-E6 cells [8]. The protective effect of Taffix® in vitro was further verified by a real-life survey conducted in Israel during September 2020, following a “superspreader” event, which found a 78% lower rate of COVID-19 cases among Taffix® users as compared to those who did not use it at all or who failed to use it as recommended [9]. According to the American Centers of Disease Control and Prevention (CDC) [12] and the United Kingdom (UK) government publication [13], a variant called B.1.1.7 with a large number of mutations was detected in the Fall of 2020. This variant spreads more easily and quickly than other variants. Recent data from the UK reported that this variant may be associated with an increased risk of death compared to other variant viruses [14]. It has since been detected in many countries around the world. This variant was first detected in the US at the end of December 2020 and is now the dominant variant [15].
In South Africa, the B.1.351 variant emerged independently of B.1.1.7. Originally detected in early October 2020, B.1.351 shares some mutations with B.1.1.7. Cases caused by this variant were reported in the US at the end of January 2021 [16]. As of March 2021, the B.1.1.7 variant was the most abundant virus found in clinical samples of COVID-19 patients around the world, and the B.1.351 variant was the second most abundant [17]. The Delta B.1.617.2 variant, dominant in the powerful 2021 surge in COVID-19 cases in India, is currently rapidly spreading across the globe, threatening to overwhelm healthcare systems and negatively impact already challenged economies. While the data are still scarce, preliminary evidence questions the effectiveness of commercial vaccines in protecting against Delta B.1.617.2 transmission and associated morbidity.
Taffix® is used worldwide as an additional layer of protection to help reduce the risk of contracting viral diseases caused by viruses whose first portal of entry to the body passes through the nasal cavity, such as influenza viruses, rhinoviruses and coronaviruses. Its efficacy against the newly emerging SARS-CoV-2 variants is therefore clinically relevant. This study showed that Taffix® effectively blocks passage of the new variants under in vitro conditions [18, 19]. The concept of nonspecific protection against upper respiratory infectious viruses was first described by Hull [20] et al., who emphasized the potential of creating a hostile microenvironment in the nasal cavity to inhibit upper respiratory infectious pathogens. Indeed, many upper respiratory viruses are sensitive to low pH, including rhinoviruses [21, 22], influenza viruses [23], respiratory syncytial virus (RSV) [24], and coronavirus [25]. Hull et al. demonstrated in a randomized, double blind, placebo-controlled clinical study that irrigation of the nasal mucosa with an acidic hydrogel spray reduced the severity and duration of common cold symptoms [21]Error! Bookmark not defined.. Sungnak et al. [5] found that SARS-CoV-2 entry factors are highly expressed in goblet and ciliated nasal epithelial cells, together with innate immune genes. These factors render the nose a likely coronavirus entry point.
Taffix® low-pH (pH 3.5) gel forms a protective layer within 1 minute from spray and the acidic film is maintained on the nasal tissue for about 5 hours [8]. The protective layer mechanically prevents virus particles from reaching the nasal mucosa and creates hostile environment to pathogens due to the acidic pH. The natural pH of the human nasal mucosa is approximately 5.5-6.5 [24], which is a favorable environment for most viral pathogens including the SARS-COV-2 virus [24].Thus, a protective device acting within a lower pH range is clinically advantageous. Taffix® is non-toxic, biocompatible, and can be easily used in addition to other protective measures, such as a face mask. It may also provide protection which is easy and comfortable to apply in situations where use of a face mask is not practical, for example during dining or sports activities.
5. Conclusions
In conclusion, under in vitro conditions, Taffix® formed a highly effective protective barrier against SARS-COV-2 variants. These results are consistent with prior findings demonstrating the high in vitro efficacy of Taffix® gel in preventing viruses from reaching and infecting cells, and support the use of the nasal spray as an effective barrier against new variants of SARS-CoV-2. Since the development and distribution of new vaccines against novel variants is a long and complex process, additional protective measures are required. Taffix® may provide a simple and safe means of reducing infections and spread of COVID-19.
Author Contributions
Conceptualization, Tair Lapidot and Dalia Megiddo; methodology, Micha Gladnikoff, Michal Mandelboim, and Yaron Drori; validation and experimentation, Nofar Atari; writing—original draft preparation Tair Lapidot and Dalia Megiddo; writing—review and editing, Michal Mandelboim, Ella Mendelson; supervision, Ella Mendelson and Tair Lapidot. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Nasus Pharma Ltd.
Data Availability Statement
The data presented in this manuscript are available on request from the corresponding author.
Acknowledgments
We thank Dr. Yehudit Posen for assisting with the scientific writing and editing.
Conflicts of Interest
The authors declare no conflict of interest. The funders had a role in the writing of the manuscript, and in the decision to publish the results.
References
- Abdool Karim SS, de Oliveira T. New SARS-CoV-2 Variants - Clinical, Public Health, and Vaccine Implications. N Engl J Med (2021).
- Rubin R. COVID-19 Vaccines vs Variants—Determining How Much Immunity Is Enough. JAMA 325 (2021): 1241-1243.
- New Sars-Cov-2 variants: How-can-vaccines-be-adapted? (2021).
- Wenling Wang, Yanli Xu, Ruqin Gao, Roujian Lu, Kai Han, Guizhen Wu, et al. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA (2020).
- Waradon Sungnak, Ni Huang, Christophe Bécavin, Marijn Berg, Rachel Queen, Monika Litvinukova, et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat Med 26 (2020): 681-687.
- Yixuan J Hou, Kenichi Okuda, Caitlin E Edwards, David R Martinez, Takanori Asakura, Kenneth H Dinnon, et al. SARS-CoV-2 Reverse Genetics Reveals a Variable Infection Gradient in the Respiratory Tract Cell. Cell 182 (2020): 429.e14-446.e14.
- Oreste Gallo, Luca Giovanni Locatello, Alessio Mazzoni, Luca Novelli, Francesco Annunziato. The central role of the nasal microenvironment in the transmission, modulation, and clinical progression of SARS-CoV-2 infection. Mucosal Immunol (2020).
- Barbara J Mann, Brett Moreau G, Tair Lapidot, Dalia Megiddo. TaffiX® Nasal Powder forms an Effective Barrier against SARS-CoV-2. Biomedical Journal of Scientific & Technical Research 3 (2021): 25843-25845.
- Klang Shmuel, Megiddo Dalia, Lapidot Tair, Naparstek Yaakov. Low pH Hypro-mellose (Taffix) nasal powder spray could reduce SARS-CoV-2 infection rate post mass-gathering event at a highly endemic community: an observational prospective open label user survey. Expert Review of Anti-infective Therapy (2021): 1-6.
- Summer E Galloway, Prabasaj Paul, Duncan R MacCannell, Michael A Johansson, John T Brooks, Adam MacNeil, et al. Emergence of SARS-CoV-2 B.1.1.7 Lineage — United States, December 29, 2020–January 12, 2021. MMWR Morb Mortal Wkly Rep 70 (2021): 95-99.
- Mulenga Mwenda, Ngonda Saasa, Nyambe Sinyange, George Busby, Peter J Chipimo, Jason Hendry, et al. Detection of B.1.351 SARS-CoV-2 Variant Strain — Zambia, December 2020. MMWR Morb Mortal Wkly Rep 70 (2021): 280-282.
- PHE investigating a novel variant of COVID-19 (2020).
- Challen R, Brooks-Pollock E, Read J M, Dyson L, Tsaneva-Atanasova K, Danon L, et al. Risk of mortality in patients infected with SARS-CoV-2 variant of concern 202012/1: matched cohort study BMJ 372 (2021): 579.
- Denise Chow. U.K. coronavirus variant is now the dominant strain in the U.S., CDC says (2021).
- Houriiyah Tegally, Eduan Wilkinson, Marta Giovanetti, Arash Iranzadeh, Vagner Fonseca, Jennifer Giandhari, et al. Emergence and rapid spread of a new severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2) lineage with multiple spike mutations in South Africa. medrxiv (2021).
- Variants and Genomic Surveillance for SARS-CoV-2. CDC (2021).
- Hull D, Rennie P, Noronha A, Poore C, Harrington N, Fearnley V, et al. Effects of creating a non-specific, virus-hostile environment in the nasopharynx on symptoms and duration of common cold. Acta otorhinolaryngologica italica 2 (2007): 73.
- Wall EC, Wu M, Harvey R, Kell G, Warchal S, Sawyer C, et al. Neutralising antibody activity against SARS-CoV-2 VOCs B.1.617.2 and B.1.351 by BNT162b2 vaccination. Lancet 397 (2021): 2331-2333.
- Planas D, Veyer D, Baidaliuk A, Staropoli I, Guivel-Benhassine F, Rajah MM, et al. Reduced sensitivity of infectious SARS-CoV-2 variant B.1.617.2 to monoclonal antibodies and sera from convalescent and vaccinated individuals. bioRxiv (2021).
- Gern JE, Mosser AG, Swenson CE, Rennie PJ, England RJ, Shaffer J, et al. Inhibition of Rhinovirus Replication In Vitro and In Vivo by Acid-Buffered Saline, J Infect Dis 195 (2007): 1137-1143.
- Hughes JH, Thomas DC, Hamparian VV. Acid lability of rhinovirus type 14 : effect of pH, time and temperature. Proc Soc Exp Biol Med 144 (1973): 555-560.
- Paul Rennie, Philip Bowtell, David Hull, Duane Charbonneau, Robert Lambkin-Williams, John Oxford. Low pH gel intranasal sprays inactivate influenza viruses in vitro and protect ferrets against influenza infection. Respir Res 38 (2007).
- Ausar SF, Rexroad J, Frolov VG, Look JL, Konar N, Middaugh CR. Analysis of the thermal and pH stability of human respiratory syncytial virus. Mol Pharm 2 (2005): 491-499.
- Lamarre A, Pierre J. Talbot Effect of pH and temperature on the infectivity of human coronavirus 229E; Canadian Journal of Microbiology 35 (1989): 972-974.
- England RJA, Homer JJ, Knight LC, Ell SR. Nasal pH measurement: a reliable and repeatable parameter. Clinical Otolaryngology & Allied Sciences 24 (1999): 67-68.
- Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu DK, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill 25 (2020): 2000045.