Skin Transparency Improvement Effects of Rice Vitamin E Crude Extract
Article Information
Yuho Yamauchi†1, Jingxiu Zhou†1, Shiori Nakagawa1, Yoshikazu Uchida2, Toshio Nakamura*1, Takuo Tsuno1
1Tsuno Rice Fine Chemicals Co., Ltd Ito-gun, Katsuragi-cho, Chonomachi 2283, Wakayama, Japan
2Department of Food Science & Nutrition, Convergence Program of Material Science for Medicine and Pharmaceutics, and The Korean Institute of Nutrition Hallym University, Chuncheon, Republic of Korea.
*Corresponding Author: Toshio Nakamura, Tsuno Rice Fine Chemicals Co., Ltd, Ito-gun, Katsuragi-cho, Chonomachi 2283, Wakayama, Japan.
Received: 01 March 2023; Accepted: 08 March 2023; Published: 25 April 2023
Citation: Yuho Yamauchi, Jingxiu Zhou, Shiori Nakagawa, Yoshikazu Uchida, Toshio Nakamura, Takuo Tsuno. Skin Transparency Improvement Effects of Rice Vitamin E Crude Extract. Journal of Food Science and Nutrition Research. 6 (2023): 31-39.
Share at FacebookAbstract
Background: Rice bran contains various active ingredients that have attracted attention as materials for use in health foods and cosmetics. Vitamin E has beneficial antioxidant effects and are used in cosmetics. We have developed a method for producing Rice Vitamin E crude extract (RVE) which contains bioactive ingredients, such as tocopherols, tocotrienols, and plant sterols from the by-product of rice bran oil extraction. In this study, we verified the effectiveness of rice bran ingredients by using cell culture assays and face application tests in humans.
Methods: The expression of cyclooxygenase-2 (COX-2) and endothelin-1 (EDN-1), both of which promote melanogenesis, was analyzed in HaCaT cells with or without RVE treatment. In the face application test, RVEcontaining cream and placebo cream were each applied to half of the face twice a day, and melanin index measurement and VISIA image analysis were performed.
Results: In cell assays, RVE reduced COX-2 and EDN-1 expression levels. In the application test, the melanin index on the RVE treated side significantly decreased over time from the start of the test, and after 12 weeks, it decreased significantly compared to that with the placebo. In addition, the analysis of the visual analog scale (VAS) questionnaire, which evaluates the participant’s perception of the skin condition, indicated improvements in texture and transparency brought on by RVE.
Conclusions: This study indicates that RVE increases skin transparency by reducing the amount of melanin through suppression of COX-2 and EDN-1 production and by improving texture and elasticity.
Keywords
Rice bran, Oryza sativa, Antioxidant, Tocopherols, Tocotrienols, Transparency
Rice bran articles; Oryza sativa articles; Antioxidant articles; Tocopherols articles; Tocotrienols articles; Transparency articles
Rice bran articles Rice bran Research articles Rice bran review articles Rice bran PubMed articles Rice bran PubMed Central articles Rice bran 2023 articles Rice bran 2024 articles Rice bran Scopus articles Rice bran impact factor journals Rice bran Scopus journals Rice bran PubMed journals Rice bran medical journals Rice bran free journals Rice bran best journals Rice bran top journals Rice bran free medical journals Rice bran famous journals Rice bran Google Scholar indexed journals Oryza sativa articles Oryza sativa Research articles Oryza sativa review articles Oryza sativa PubMed articles Oryza sativa PubMed Central articles Oryza sativa 2023 articles Oryza sativa 2024 articles Oryza sativa Scopus articles Oryza sativa impact factor journals Oryza sativa Scopus journals Oryza sativa PubMed journals Oryza sativa medical journals Oryza sativa free journals Oryza sativa best journals Oryza sativa top journals Oryza sativa free medical journals Oryza sativa famous journals Oryza sativa Google Scholar indexed journals Antioxidant articles Antioxidant Research articles Antioxidant review articles Antioxidant PubMed articles Antioxidant PubMed Central articles Antioxidant 2023 articles Antioxidant 2024 articles Antioxidant Scopus articles Antioxidant impact factor journals Antioxidant Scopus journals Antioxidant PubMed journals Antioxidant medical journals Antioxidant free journals Antioxidant best journals Antioxidant top journals Antioxidant free medical journals Antioxidant famous journals Antioxidant Google Scholar indexed journals Tocopherols articles Tocopherols Research articles Tocopherols review articles Tocopherols PubMed articles Tocopherols PubMed Central articles Tocopherols 2023 articles Tocopherols 2024 articles Tocopherols Scopus articles Tocopherols impact factor journals Tocopherols Scopus journals Tocopherols PubMed journals Tocopherols medical journals Tocopherols free journals Tocopherols best journals Tocopherols top journals Tocopherols free medical journals Tocopherols famous journals Tocopherols Google Scholar indexed journals Tocotrienols articles Tocotrienols Research articles Tocotrienols review articles Tocotrienols PubMed articles Tocotrienols PubMed Central articles Tocotrienols 2023 articles Tocotrienols 2024 articles Tocotrienols Scopus articles Tocotrienols impact factor journals Tocotrienols Scopus journals Tocotrienols PubMed journals Tocotrienols medical journals Tocotrienols free journals Tocotrienols best journals Tocotrienols top journals Tocotrienols free medical journals Tocotrienols famous journals Tocotrienols Google Scholar indexed journals Transparency articles Transparency Research articles Transparency review articles Transparency PubMed articles Transparency PubMed Central articles Transparency 2023 articles Transparency 2024 articles Transparency Scopus articles Transparency impact factor journals Transparency Scopus journals Transparency PubMed journals Transparency medical journals Transparency free journals Transparency best journals Transparency top journals Transparency free medical journals Transparency famous journals Transparency Google Scholar indexed journals melanogenesis articles melanogenesis Research articles melanogenesis review articles melanogenesis PubMed articles melanogenesis PubMed Central articles melanogenesis 2023 articles melanogenesis 2024 articles melanogenesis Scopus articles melanogenesis impact factor journals melanogenesis Scopus journals melanogenesis PubMed journals melanogenesis medical journals melanogenesis free journals melanogenesis best journals melanogenesis top journals melanogenesis free medical journals melanogenesis famous journals melanogenesis Google Scholar indexed journals vitamin E articles vitamin E Research articles vitamin E review articles vitamin E PubMed articles vitamin E PubMed Central articles vitamin E 2023 articles vitamin E 2024 articles vitamin E Scopus articles vitamin E impact factor journals vitamin E Scopus journals vitamin E PubMed journals vitamin E medical journals vitamin E free journals vitamin E best journals vitamin E top journals vitamin E free medical journals vitamin E famous journals vitamin E Google Scholar indexed journals
Article Details
1. Introduction
Rice is cultivated in many countries worldwide, mainly in East and Southeast Asia. Rice bran, a non-edible component of rice, accounts for approximately 10% of its weight and is known to contain various functional ingredients [1]. Among the functional ingredients of rice bran are tocopherols and tocotrienols, which belong to the vitamin E group. Tocotrienols have approximately 50 times the antioxidant power of tocopherols but are found in small quantities in only certain foods. We have developed Rice Vitamin E crude extract (RVE), containing more than 3% each of tocopherols and tocotrienols, by removing fatty acids and concentrating unsaponifiable substances from the deodorized fraction, which is a by-product of the rice oil refining process. It is reported that tocotrienols or tocotrienol-rich fraction inhibit melanogenesis by modulating the expression of genes involved in the regulation of melanin synthesis and inhibiting tyrosinase activity in melanoma cells [2,3]. Previous studies have also shown that melanogenesis is promoted by cyclooxygenase-2 (COX-2) and endothelin-1 (EDN-1) [4,5]. In HaCaT cells exposed to UVA radiation, the application of vitamin E provided photoprotection [6,7]. In addition, it is reported that α-tocopherol is effective in diminishing the production of superoxide anion following UVA and UVB irradiation [8]. These reports indicated that vitamin E inhibits UV-induced melanogenesis and inflammation. RVE is rich in esters of phytosterols, triterpene alcohols, and fatty acids. Phytosterols are known to have various functionalities, such as antioxidant and anti-inflammatory effects in the field of cosmetics [9], however, complete understanding of the biological functions of the ester form of phytosterols is still lacking. We investigated whether RVE, a mixture of these rice bran-derived functional ingredients, is sufficiently effective as a cosmetic material, focusing on melanin-reduction, and spot- and pore-improving actions of antioxidants.
2. Material and Methods
2.1 Test samples
The RVE used in this study was provided by Tsuno Food Industry Co., Ltd. and was dissolved in dimethyl sulfoxide (DMSO) before being added to the medium.
2.2 Cell culture
HaCaT cells were cultured in Dulbecco’s modified Eagle medium (DMEM; Fuji Film Wako Pure Chemical Corporation, Osaka, Japan) containing 10% fetal bovine serum (FBS, Biosera, Nuaille, France) and 1% penicillin streptomycin (Fuji Film Wako Pure Chemical Corporation, Osaka, Japan). The cultured cells were suspended and removed on to a 24-well plate (WATSON, Tokyo, Japan) with 5.0 × 104 cells/well, and the cells were pre-cultured overnight at 37 °C under 5% CO2 atmosphere and then used for the examination.
2.3 mRNA expression levels of melanin-related factors
RVE was dissolved in DMEM containing 1% FBS at a final concentration of 0.01 or 0.005% (w/v). Pre-cultured cells were treated with the RVE media and cultured under 5% CO2 at 37 °C for 3 h. (n = 4) Total RNA was extracted from the cells using ISOGEN II (Nippon Gene, Tokyo, Japan). Total RNA was dissolved in Milli-Q water, and the RNA concentration was calculated using an Epoch microplate spectrophotometer (Agilent, Santa Clara, California, USA). Each RNA solution was adjusted to a concentration of 50 µg/µL. Reverse transcription was performed in a thermal cycler (Thermo Fisher Scientific, Waltham, MA, USA) using the Prime Script RT Reagent kit (Takara Bio, Shiga, Japan) according to the manufacturer’s instructions. The cDNA obtained was used as a template. Real-time PCR was performed using the StepOne system (Thermo Fisher Scientific, Waltham, MA, USA) with specific primers. Real-time PCR detected glyceraldehydes 3-phosphatedehydrogenase (GAPDH), cyclooxygenase-2 (COX-2), and endothelin-1(EDN-1) genes according to the method using Power Up SYBR Green Master Mix (Takara Bio, Shiga, Japan). The expression levels of the target genes were calculated by comparing the expression levels of the housekeeping gene GAPDH with the ΔΔCT method. Primer sequences for GAPDH and EDN-1 were based on reports by Adachi et al. (2015) [10] and Nakamura-Utsunomiya et al. (2020) [11], and COX-2 primers were designed using Primer BLAST (Table. 1).
|
Gene |
Sequence (5′→ 3′) |
|
GAPDH |
TGCACCACCAACTGCTTAGC |
|
(glyceraldehyde-3-phosphate dehydrogenase) |
TCTTCTGGGTGGCAGTGATG |
|
COX-2 |
AAGCAGGCTAATACTGATAGGAGAG |
|
(cyclooxygenase 2) |
GCCACTCAAGTGTTGCACATA |
|
EDN-1 |
ACTTCTGCCACCTGGACATC |
|
(endtoherin 1) |
GGCAAAAATTCCAGCACTTC |
Table 1: List of primers used for real-time PCR.
2.4 Clinical trial participants
This study was conducted in accordance with the Declaration of Helsinki and the Ethical Guidelines for Medical and Health Research Involving Human Subjects proposed by the Japan Ministry of Health, Labor, and Welfare. All participants provided written informed consent before the commencement of the study, which was approved by the Ethics Committees of Tsuno Food Industry Co., Ltd. (approval number: RD 21-003). In addition, we provided sufficient explanation to the test participants during the briefing session. The participants in this study met the registration/selection criteria and did not violate the exclusion criteria. We selected participants who were women in their 20s to 50s, confirmed in the questionnaire that they were healthy, did not have skin diseases, and did not correspond to statistical outliers in the measurement parameters.
2.5 Test cream product
The composition of the creams used in this study is listed in table 2. The RVE cream contained 1% RVE, and the placebo cream contained the same amount of water instead of RVE.
|
Component |
|
|
Placebo cream |
Water, triethylhexanoin, butylene glycol, stearyl alcohol, behenyl alcohol, polysorbate 20, |
|
glyceryl stearate, methylparaben, citric acid |
|
|
RVE cream |
placebo cream +1% RVE |
Table 2: RVE and placebo cream formulation
2.6 Clinical trial design
This clinical trial was a placebo-controlled, single-blind test. Application to half of the face continued for 12 weeks. Each cream was applied twice daily, in the morning and evening. The primary outcome was the melanin index, and the secondary outcomes were the VISIA spot and pore scores. The participants were equally divided into a group that applied a cream containing 1% RVE (RVE cream) on the right side and a group that applied a placebo cream on the right side. In addition, participants were allocated such that the initial measurements of the primary and secondary outcomes of the placebo-applied side and the RVE-applied side were even (Figure 1).
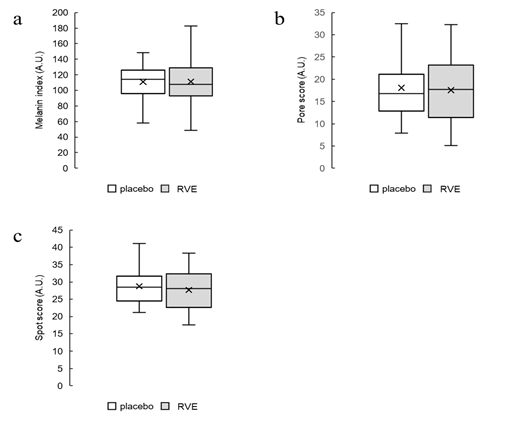
Figure 1: Box plot
Distribution of initial values of melanin index (a), spots score (b), and pore score (c) after allocation. The white bar shows the values of placebo treatment sides, and the gray bar shows the values of RVE treatment sides.
2.7 Measurements
Measurements were performed every four weeks after the start of the application (0th, 4th, 8th, and 12th weeks). Before the measurement, the participants washed their faces and acclimated for 20 min or more in a room with constant temperature and humidity (22 ± 2 °C, RH% = 50 ± 5). The cheeks of the participants were measured using a Mexameter MX18 (Courage + Khazaka, Cologne, Germany) for skin melanin index and a Cutometer DUAL MPA580 (Courage + Khazaka, Cologne, Germany) for skin viscoelasticity. In addition, spots and pores were analyzed using VISIA Evolution (Canfield Scientific, Parsippany, NJ, USA) for facial image analysis. The participants responded to a VAS questionnaire at the time of measurement. The degree of skin condition they felt was recorded on a 10 cm visual analog scale (0 cm = very bad, 10 cm = very good).
2.8 Statistical analyses
In this study, the values are shown as the mean ± standard error, and only the age of the participants is shown as the mean ± standard deviation. The measured values of the clinical test were set to 0 before application of the cream (0 week), and the degree of change after a certain period was determined. Comparisons between groups in the cell test and comparisons before and after application of the test cream in the clinical trial were analyzed using Dunnett's test; a risk rate of 5% or less was considered a significant difference. In addition, the clinical test results were compared between the placebo-treated side and RVE-treated side and analyzed using Student’s t-test. Cases with a risk rate ≤ 5% were found to have a significant difference. MEPHAS, a statistical analysis program for pharmaceutical data provided by the Center for Genetic Information Experiments at Osaka University, was used in this study.
3. Results
3.1 Expression levels of COX-2 and EDN-1 in cultured epidermal keratinocyte cells HaCaT
RT-qPCR was used to validate the effects of RVE on COX-2 and EDN-1 expression. COX-2 expression was significantly decreased in cells treated with 0.001% and 0.01% RVE compared with that in untreated cells (Figure 2a). A significant decrease in EDN-1 expression was also observed with the 0.001% and 0.01% RVE treatments (Figure 2b).
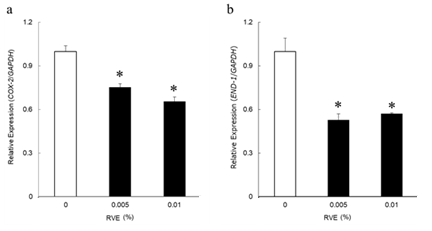
Figure 2: Related factors of melanin synthesis
The expression levels of COX-2 (a) and EDN-1(b) in HaCaT examined by real-time PCR. The expression level of each gene is given as a ratio to that of GAPDH, and the expression of RVE-untreated samples is denoted as 1. Data are presented as the mean ± SE. (n = 4) Data were analyzed by Dunnett's test. * ; p < 0.05 (vs. RVE-untreated samples)
3.2 Results of participant-grouping
The study design for topical application of RVE is presented in table 3. Twenty-five participants were assessed for eligibility, and three did not meet the inclusion criteria. Twenty-two participants were divided into two groups: one group applied RVE cream on the right side of the face, and the other group applied RVE cream on the left side. There was no significant difference between the initial values of the RVE-treated- and placebo-treated sides for any of the measurement parameters. The mean age of the participants at the start of the study was 37.5 ± 11.7 years. One participant dropped out during the test period, and the final analysis was conducted on 21 participants.
Table 3: Design of clinical trial
3.3 Melanin index
The melanin index measured by Mexameter MX18 showed a significant decrease from the initial value only at 8th week in the placebo group (p < 0.05) and in the RVE group at 4th, 8th, and 12th week (p < 0.05). The decrease from the initial measurement at 12th week was greater in the RVE group than that in the placebo group (p < 0.05) (Figure 3).
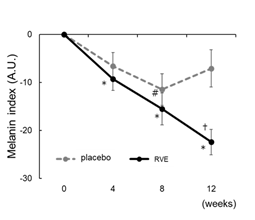
Figure 3:Improvement melanin index
Changes from the 0th week in melanin index. The dotted line shows placebo treated sides and the solid line shows the RVE treatment sides. Data are presented as the mean ± SE. Data were analyzed by Dunnett's test. * ; p < 0.05 (vs. 0th week RVE), # ; p < 0.05 (vs. 0th week placebo), † ; p < 0.05 (vs. placebo treated sides).
3.4 Results of VISIA analysis
Scores of the spots and pores were analyzed using VISIA evolution. There were no significant differences in the measurements between the initial and later stages and between the placebo and RVE sides (Figure 4).
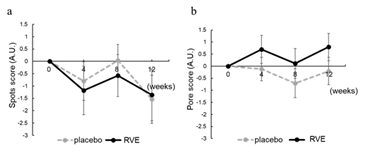
Figure 4: Result of Spots and Pore score
Changes from the 0th week in spots (a) and pore (b) score analyzed by VISIA. The dotted line shows placebo treated sides and the solid line shows the RVE treatment sides. Data are presented as the mean ± SE.
3.5 Elasticity
The R2 value, which is the gross elasticity, R5 value, which is the net elasticity, and R7 value, which is the recovery after deformation, measured by the Cutometer DUAL MPA580, were evaluated on the placebo and RVE sides. No significant differences were observed before and after application on either side (Figure 5). However, R5 and R7 values tended to increase from the initial values on the RVE side at weeks 4 and 8 compared to those on the placebo side (Figure 5b, 5c).
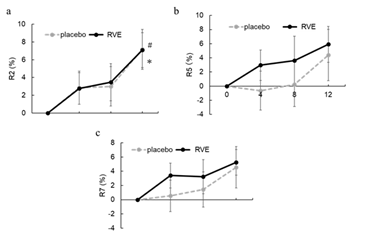
Figure 5: Result of skin elasticity
Changes from 0th week in the gross elasticity R2 (%) (a), net elasticity R5 (%) (b), and recovery after deformation R7 (%) (c). The dotted line shows placebo treated sides and the solid line shows the RVE treatment sides. Data are presented as the mean ± SE. * ; p < 0.05 (vs. 0th week RVE), # ; p < 0.05 (vs. 0th week placebo).
3.6 VAS questionnaire results
A VAS questionnaire was used to quantify and analyze the concerns regarding skin texture, elasticity, and transparency among the participants. The degree of improvement in skin texture tended to increase from the initial value on the RVE side to a greater extent than that in the placebo group at each stage (Figure 6a). Regarding elasticity, the amount of change from the initial value at 4th week was significantly higher in the RVE treated side than that in the placebo side (p < 0.05) (Figure 6b). Regarding transparency, only the RVE-treated side showed a significant score increase from the initial value at 8th week (p < 0.05) (Figure 6c).
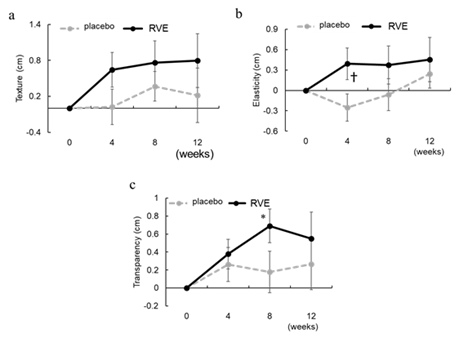
Figure 6: Result of VAS questionnaire
Changes from 0th week in the score of texture (a), elasticity (b), and transparency (c) by VAS questionnaire. The dotted line shows placebo treated sides and the solid line shows the RVE treatment sides. Data are presented as the mean ± SE. * ; p < 0.05 (vs. 0th week RVE), † ; p < 0.05 (vs. placebo treated sides).
4. Discussion
We examined the cosmetic effect of RVE on human skin by analyzing the gene expression of melanin production-promoting factors in cultured cells and the amount of melanin, spots, and pore scores in a half-face application test. In cultured cells, reduced expression levels of COX-2 and EDN-1, promoters of tyrosinase activity, was observed. We also found that RVE reduced the amount of melanin on human skin. There was no significant difference in the VISIA blemish score, but the decrease in the RVE treated side was greater than that in the placebo side at 4 and 8 weeks. RVE is thought to reduce melanin and spots because the vitamin E group in RVE contains tocotrienol, which is reported to have a strong antioxidant effect and removes reactive oxygen species generated by UV rays, thereby reducing COX-2 and EDN-1 synthesis.
The pore score, analyzed by VISIA, did not decrease due to RVE application. In this study, we included the pore score as a secondary outcome in anticipation of a reduction in pore darkening owing to the inhibition of lipid peroxide production by the antioxidant action of RVE. A once-daily application of a comprehensive anti-oxidant product containing vitamins C and E, among others, was reported to improve pores in men's skin as assessed by visual inspection by an expert [12]. On the other hand, in a study investigating the influence of astaxanthin-containing beverages, which have an anti-lipid peroxidation effect on the skin, positive effects were seen in skin texture and moisture content, but no effect was seen in pores analyzed by VISIA [13]. In the VISIA analysis, the score is calculated from the number and area of pores, and therefore, it is not possible to strictly evaluate changes in darkening due to oxidation of sebum in pores. Since the darkening of pores is also related to a decrease in skin transparency, the VAS questionnaire showed an improvement in transparency; therefore, we presumed that there was no effect on the conspicuity of pores.
Regarding skin elasticity, the R5 value, which is the net elasticity, and the R7 value, which is the recovery after deformation, tended to increase from the initial measurement values on the RVE side compared to those on the placebo side, although they were not statistically significant. The tendency to increase observed in R5 and R7 is consistent with the VAS questionnaire results, which showed a significantly greater change from the initial value in the 4th week in the RVE treated side than that in the placebo side. Similar to RVE, the phytosterol ester extract, which is rich in plant sterol esters, showed the effect of promoting type 3 collagen production in fibroblasts in our laboratory. In addition, since vitamin E has an antioxidant effect, it is thought to repair the structure of the dermis damaged by oxidative stress. These findings suggest that the plant sterol esters and vitamin E contained in RVE contribute to the improvement of elasticity.
In addition to the elasticity mentioned above, the VAS questionnaire showed that the application of RVE improved the texture and transparency. It has been reported that the fineness of texture and the amount of melanin are involved in the transparency of the skin [14]. Another study that systematically analyzed the interaction between clarity and skin physiology found that “red spot count” and “skin smoothness” and “yellowness or lightness in skin color” as key characteristics that dictate skin transparency [15]. In addition, it has been reported that the carbonylation of the stratum corneum protein is involved in the yellowing of the skin and the loss of transparency [16,17]. Carbonylated protein has been used as a measure of protein oxidation [18,19], and it should be considered that this might be improved by RVE containing vitamin E group with antioxidative effects. The transparency of the skin is considered to be a major factor in beautiful skin, so there should be a high demand for its improving effect.
In this study, RVE was shown to have melanin-reducing, elasticity-increasing, and texture- improving actions and has been found to be a valid cosmetic raw material with multiple effects of improving skin transparency.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
References
- Ghosh M. Review on recent trends in rice bran oil processing. J Am Oil Chem Soc 84 (2007): 315-324.
- Michihara A, Ogawa S, Kamizaki Y, et al. Effect of delta-tocotrienol on melanin content and enzymes for melanin synthesis in mouse melanoma cells. Biol Pharm Bull 33 (2010): 1471-1476.
- Makpol S, Jam FA, Rahim NA, et al. Comparable down-regulation of TYR, TYRP1 and TYRP2 genes and inhibition of melanogenesis by tyrostat, tocotrienol-rich fraction and tocopherol in human skin melanocytes improves skin pigmentation. Clin Ter 165 (2014): e39-e45.
- Eo S H, Kim S J. Resveratrol-mediated inhibition of cyclooxygenase-2 in melanocytes suppresses melanogenesis through extracellular signal-regulated kinase 1/2 and phosphoinositide 3-kinase/Akt signalling. European Journal of Pharmacology 860 (2019): 172586.
- Hachiya A, Kobayashi A, Ohuchi A, et al. The paracrine role of stem cell factor/c-kit signaling in the activation of human melanocytes in ultraviolet-B-induced pigmentation. J. Investig. Dermatol 116 (2001): 578-586.
- Delinasios GJ, Karbaschi M, Cooke MS, et al. Vitamin E inhibits the UVAI induction of “light” and “dark” cyclobutane pyrimidine dimers, and oxidatively generated DNA damage, in keratinocytes. Sci Rep 8 (2018): 423.
- Wu CM, Cheng YL, Dai YH, et al. alpha-Tocopherol protects keratinocytes against ultraviolet A irradiation by suppressing glutathione depletion, lipid peroxidation and reactive oxygen species generation. Biomed. Rep 2 (2014):419-423.
- Gao LX, Bian C, Wu Y, et al. Label-free electrochemical sensor to investigate the effect of tocopherol on generation of superoxide ions following UV irradiation. J Biol Eng 12 (2008): 17.
- Loizou S, Paraschos S, Mitakou S, et al. Chios mastic gum extract and isolated phytosterol tirucallol exhibit anti-inflammatory activity in human aortic endothelial cells. Exp Biol Med 234 (2009): 553–61.
- Adachi H, Murakami Y, Hotta N, et al. Investigation of the skin aging mechanism focusing on stratifin. J Soc Cosmet Chem Jpn 49 (2015): 211-217.
- Nakamura-Utsunomiya A, Tsumura M, Okada S, et al. Downregulation of endothelial nitric oxide synthase (eNOS) and endothelin-1 (ET-1) in a co-culture system with human stimulated X-linked CGD neutrophils. PLoS One 15 (2020): e0230665
- Sikora BC, Wortzman M, Nelson DB, et al. A pilot study evaluating the efficacy and tolerability of a comprehensive, hydrating topical antioxidant developed specifically for men. J Cosmet Dermatol 20 (2021): 2816-2823.
- Tsukahara H, Matsuyama A, Abe T, et al. Effects of intake of astaxanthin contained drink on skin condition. Jpn J Complementary Altern Med 13 (2016): 57-62.
- Masuda Y, Kunizawa N, Takahashi M. Methodology for evaluation of skin transparency and the efficacy of an essence that can improve skin transparency. J Soc Cosmet Chem 39 (2005): 201-208.
- Nakamura R, Itai E, Uehara S, et al. Development of Mathematical Model of Skin Translucency. J. Soc Cosmet. Chem. Jpn 54 (2020): 4.
- Masaki, H, Mizutani T, Ogawa N, et al. Carbonylated proteins contribute to the darkness around facial pores. J. Dermatol. Sci 89 (2008): 299-307.
- Ichiro Iwai, Tomohiro Kuwahara, Tetsuji Hirao. Decrease in the Skin Transparency Induced by Protein Carbonylation in the Stratum Corneum. J. Soc. Cosmet. Chem. Jpn 43 (2008): 1.
- CS Sander, H Chang, S Salzmann, et al. Thiele Photoaging is associated with protein oxidation in human skin in vivo J. Invest. Dermatol 118 (2022): 618-625.
- Laurent C, LeDuc A, Pottier I, et al. Dramatic Increase in Oxidative Stress in Carbon-Irradiated Normal Human Skin Fibroblasts. PLoS ONE 8 (2013): e85158.
