Sinking Skin Flap Syndrome: Cause of Secondary Neurological Deterioration
Article Information
Touab Rida1*, Rabii Andaloussi Mohamed1, Mohsani Mohamed2, Mounir Khalil1, Bensghir Mustapha1, Balkhi Hicham1
1Departement of Anesthesiology and Critical Care, Military Training Hospital Mohamed V, Rabat, Morocco
2Departement of Neurosurgery, Military Training Hospital Mohamed V, Rabat, Morocco
*Corresponding Author: Touab Rida, Nahda 1, Rabat, Morocco, Tel: +212649323552
Received: 01 February 2021; Accepted: 24 February 2021; Published: 26 February 2021
Citation: Touab Rida, Rabii Andaloussi Mohamed, Mohsani Mohamed, Mounir Khalil, Bensghir Mustapha, Balkhi Hicham. Sinking Skin Flap Syndrome: Cause of Secondary Neurological Deterioration. Anesthesia and Critical Care 3 (2021): 001-009.
Share at FacebookAbstract
Introduction: Sinking skin flap syndrome is a rare complication of craniectomy, which is performed as a treatment of severe intracranial hypertension.
Cases Reports: The first case is a 55 year old man. Admitted with Glascow score of 13/15, rapid neurological deterioration was noted with a GCS of 9/15, and then anisocoria. CT scan had objectified hemorrhagic contusions, subdural hematoma measured 11 mm and deviation of the median line. The patient was operated with evacuation of the subdural hematoma through a large decompressive craniectomy. In the second month, he presented a cranial deformation with a deepening of the cutaneous plane, with deterioration of the neurological status and a generalized convulsive crisis. The second case is that of a 32 year old man, admitted to the emergency room with 8/15 of GCS with anisocoria. CT scan was showing an 11 mm right subdural hematoma with a hemorrhagic contusion opposite, a 12 mm midline deviation and diffuse cerebral edema. The patient was operated with evacuation of the subdural hematoma through a large craniectomy. The neurological examination after the extubation showed a GCS of 14. Two days later, the patient presented a depression of the right scalp with an aspect of skin flap syndrome on CT scan without significant neurological deterioration.
Conclusion: The role of decompressive craniectomy in neurological improvement in still uncertain, and timing of cranioplasty is more debate: early with unclear neurologic status and preventing the skin flap syndrome or delete after final outcome.
Keywords
Craniectomy; Skin flap syndrome; Cranioplasty; Brain injury
Craniectomy articles; Skin flap syndrome articles; Cranioplasty articles; Brain injury articles
Craniectomy articles Craniectomy Research articles Craniectomy review articles Craniectomy PubMed articles Craniectomy PubMed Central articles Craniectomy 2023 articles Craniectomy 2024 articles Craniectomy Scopus articles Craniectomy impact factor journals Craniectomy Scopus journals Craniectomy PubMed journals Craniectomy medical journals Craniectomy free journals Craniectomy best journals Craniectomy top journals Craniectomy free medical journals Craniectomy famous journals Craniectomy Google Scholar indexed journals Skin flap syndrome articles Skin flap syndrome Research articles Skin flap syndrome review articles Skin flap syndrome PubMed articles Skin flap syndrome PubMed Central articles Skin flap syndrome 2023 articles Skin flap syndrome 2024 articles Skin flap syndrome Scopus articles Skin flap syndrome impact factor journals Skin flap syndrome Scopus journals Skin flap syndrome PubMed journals Skin flap syndrome medical journals Skin flap syndrome free journals Skin flap syndrome best journals Skin flap syndrome top journals Skin flap syndrome free medical journals Skin flap syndrome famous journals Skin flap syndrome Google Scholar indexed journals Cranioplasty articles Cranioplasty Research articles Cranioplasty review articles Cranioplasty PubMed articles Cranioplasty PubMed Central articles Cranioplasty 2023 articles Cranioplasty 2024 articles Cranioplasty Scopus articles Cranioplasty impact factor journals Cranioplasty Scopus journals Cranioplasty PubMed journals Cranioplasty medical journals Cranioplasty free journals Cranioplasty best journals Cranioplasty top journals Cranioplasty free medical journals Cranioplasty famous journals Cranioplasty Google Scholar indexed journals Brain injury articles Brain injury Research articles Brain injury review articles Brain injury PubMed articles Brain injury PubMed Central articles Brain injury 2023 articles Brain injury 2024 articles Brain injury Scopus articles Brain injury impact factor journals Brain injury Scopus journals Brain injury PubMed journals Brain injury medical journals Brain injury free journals Brain injury best journals Brain injury top journals Brain injury free medical journals Brain injury famous journals Brain injury Google Scholar indexed journals CT scan articles CT scan Research articles CT scan review articles CT scan PubMed articles CT scan PubMed Central articles CT scan 2023 articles CT scan 2024 articles CT scan Scopus articles CT scan impact factor journals CT scan Scopus journals CT scan PubMed journals CT scan medical journals CT scan free journals CT scan best journals CT scan top journals CT scan free medical journals CT scan famous journals CT scan Google Scholar indexed journals Neurological Deterioration articles Neurological Deterioration Research articles Neurological Deterioration review articles Neurological Deterioration PubMed articles Neurological Deterioration PubMed Central articles Neurological Deterioration 2023 articles Neurological Deterioration 2024 articles Neurological Deterioration Scopus articles Neurological Deterioration impact factor journals Neurological Deterioration Scopus journals Neurological Deterioration PubMed journals Neurological Deterioration medical journals Neurological Deterioration free journals Neurological Deterioration best journals Neurological Deterioration top journals Neurological Deterioration free medical journals Neurological Deterioration famous journals Neurological Deterioration Google Scholar indexed journals craniectomy articles craniectomy Research articles craniectomy review articles craniectomy PubMed articles craniectomy PubMed Central articles craniectomy 2023 articles craniectomy 2024 articles craniectomy Scopus articles craniectomy impact factor journals craniectomy Scopus journals craniectomy PubMed journals craniectomy medical journals craniectomy free journals craniectomy best journals craniectomy top journals craniectomy free medical journals craniectomy famous journals craniectomy Google Scholar indexed journals cranioplasty articles cranioplasty Research articles cranioplasty review articles cranioplasty PubMed articles cranioplasty PubMed Central articles cranioplasty 2023 articles cranioplasty 2024 articles cranioplasty Scopus articles cranioplasty impact factor journals cranioplasty Scopus journals cranioplasty PubMed journals cranioplasty medical journals cranioplasty free journals cranioplasty best journals cranioplasty top journals cranioplasty free medical journals cranioplasty famous journals cranioplasty Google Scholar indexed journals hemorrhagic stroke articles hemorrhagic stroke Research articles hemorrhagic stroke review articles hemorrhagic stroke PubMed articles hemorrhagic stroke PubMed Central articles hemorrhagic stroke 2023 articles hemorrhagic stroke 2024 articles hemorrhagic stroke Scopus articles hemorrhagic stroke impact factor journals hemorrhagic stroke Scopus journals hemorrhagic stroke PubMed journals hemorrhagic stroke medical journals hemorrhagic stroke free journals hemorrhagic stroke best journals hemorrhagic stroke top journals hemorrhagic stroke free medical journals hemorrhagic stroke famous journals hemorrhagic stroke Google Scholar indexed journals EEG articles EEG Research articles EEG review articles EEG PubMed articles EEG PubMed Central articles EEG 2023 articles EEG 2024 articles EEG Scopus articles EEG impact factor journals EEG Scopus journals EEG PubMed journals EEG medical journals EEG free journals EEG best journals EEG top journals EEG free medical journals EEG famous journals EEG Google Scholar indexed journals
Article Details
1. Introduction
Sinking skin flap syndrome is a rare complication of craniectomy, which is performed as a last resort in the treatment of severe intracranial hypertension secondary to a variety of hemorrhagic or ischemic, traumatic or spontaneous brain injuries. First described in 1939 by Grant and Norcross [1] as a set of subjective symptoms, and then redefined by Yamaura and Makino [2] as neurological deterioration with concave deformation and relaxation of the scalp skin occurring several weeks or months after a large craniectomy [3]. We describe two observations with the occurrence of skin flap syndrome in the aftermath of craniectomy performed in the management of severe head trauma, through which we raise the risk of late neurological deterioration after craniectomy, presenting the factors favoring and the rules of management of such rare complication, leaving open the debate on the timing of cranioplasty.
2. Case Reports
The first case, is a 55 year old man, with no notable pathological history, hit by a car with a fall and head trauma, transferred to the emergency room within two hours of trauma, with a Glascow score on admission of 13/15 including an eye opening rated at 3, motor response at 5 and verbal response at 5 while having a stable hemodynamic and respiratory status. Rapid neurological deterioration was noted with a GCS of 9/15 (eye opening 2, motor and verbal response 4 and 3 respectively) with no hemodynamic (mean blood pressure 80 mmHg, heart rate 105 beats per minute) or respiratory (SpO2 98% in ambient air) deterioration, capillary blood glucose 1.65 g/l and no signs of seizure, Hence his admission to the vital emergency room, where he was placed in a 30° head position under oxygen by a high-concentration face mask for preoxygenation, with continuous monitoring of his heart rate through an electrocardioscope, his SPO2 and his non- invasive blood pressure every five minutes. After rapid preparation, rapid sequence anesthetic induction with analgesic injection and without Sellick maneuver by fentanyl at a dose of 2 mcg/Kg, propofol at a dose of 2 mg/Kg and rocuronium at a dose of 1 mg/Kg. Intubation was performed successfully, without desaturation or use of ventilation likely to increase the risk of inhalation, with mean post-induction blood pressure at 75 mmHg. The patient was placed under continuous sedation with midazolam and fentanyl with norepinephrine introduction to increase the mean arterial pressure allowing cerebral perfusion, and under controlled ventilation mode with a tidal volume at 8 ml/Kg, a FiO2 at 0.4 allowing 98% SpO2, a PEEP at 5 cm H2O and a respiratory rate at 14 cycles per minute allowing a PaCO2 at 36 mmHg.
A full body CT scan showed lesions on the single intracranial stage not related to emergency surgery, including a 3mm subdural hematoma on the left side, and a right frontal contusion site. The patient was admitted to intensive care unit, maintaining continuous intravenous sedation, ventilation by the same parameters, norepinephrine, infusion of isotonic solutions, starting primary prophylaxis of convulsions by leviteracetam and prevention of gastric stress ulcer by a proton pump inhibitor, prevention of thromboembolic disease at this stage by compression stockings and pressure ulcers through a water mattress. Neurological monitoring was carried out by checking the state of the pupils that were in miosis, transcranial Doppler performed three times a day and at each need objectivizing diastolic velocities and a pulsality index at 32 and 1 respectively on the right and 28 and 1.10 respectively on the left, associated with a verification of the absence of secondary aggressions of systemic origin on an hourly basis. The blood pressure was continuously monitored to adjust the dose of norepinephrine with a MAP target of 90 mmHg.
At H14 of admission, the patient had exhibited an anisocoria, motivating the increase in MAP via an increase in the dose of norepinephrine, infusion of mannitol at a dose of 1g/Kg over 20 minutes, and mild hyperventilation, then the realization of an emergency CT scan which had objectified multiple foci of bi-hemispheric hemorrhagic contusions, an increase in the volume of the HSD which had become 11 mm, a deviation of the median line of 8 mm, collapsed lateral ventricles and diffuse cerebral edema. In view of this aspect, the patient was operated on emergency with evacuation of the subdural hematoma through a large left fronto-parieto-temporal craniectomy.
The immediate postoperative period did not present any complications, and the patient was kept sedated with midazolam and fentanyl for 72 hours, under norepinephrine, controlling all the parameters that could lead to cerebral aggression. A window of sedation was performed and enteral feeding was started, with no return to a state of consciousness, leading to an imaging control which had not objectified any novelties apart from the resorption of certain haemorrhagic contusion foci and the disappearance of the haematoma subject of the operative act. On the seventh day of his admission a cessation of anti- comital treatment was carried out with the introduction of an antibiotic therapy based on imipenem associated with colistine for the treatment of a pneumopathy acquired under mechanical ventilation with Acinetobacter Baumanii. The patient had a tracheotomy at D10, and progressed to a chronic phase with a vegetative status, and then paucyrelational made a spontaneous opening of the eyes with blinking at the call or sound stimulation without motor reaction of the limbs. He received supportive care such as exclusive enteral feeding, motor and respiratory physiotherapy, prevention of thromboembolic disease with low molecular weight heparin, ventilation in inspiratory aid mode.
In the second month of its evolution, the patient presented a cranial deformation with a deepening of the cutaneous plane in relation to the craniectomy (Figure 1), with deterioration of the neurological status and a generalized convulsive crisis, treated with intravenous midazolam with resumption of a continuous infusion of sedation. The brain CT scan imaging objectified an aspect of the skin flap syndrome, hence the complementary management during the following 24 hours, keeping the elevated position of the lower limbs with head and trunk at 0°, hydration and the introduction of liveteracetam for secondary prevention with the rest of the patient's usual treatment. The evolution was favourable after the sedation was stopped with clinical improvement: regaining of the state of consciousness before the deterioration and disappearance of the convulsions and skin depression.
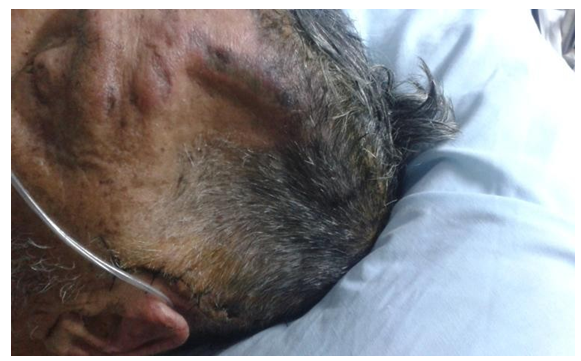
Figure 1: Scalp depression regarding craniectomy.
In the following weeks, neurological improvement was recovered with restoration of contact, movement execution and memory with weaning of mechanical ventilation and decannulation, keeping a left hemiparesis limiting walking to the possibility with assistance. The patient is then transferred to the neurosurgery department for cranioplasty.
The second case is that of a 32 year old man, without any known pathological history, motorcyclist hit by a car, admitted to the emergency room of our hospital at H6 of the trauma with an initial examination objectifying a systolic blood pressure at 137 mmHg and a diastolic blood pressure at 78 mmHg and a mean pressure at 97 mmHg, 88% pulsed oxygen saturation in ambient air in relation to a rapidly corrected tongue fall after placement of an oropharyngeal cannula and oxygen therapy, 8/15 GCS including motor response at 4, verbal response at 2 and eye opening response at 2, with an anisocoria on pupillary examination. An initial treatment consisted in the reception room of vital emergencies in the setting up of a neck brace, the taking of two peripheral venous routes, preoxygenation by a BAVU, preparation of intubation equipment, and the beginning of osmotherapy with mannitol 20% at a rate of 80g at the same time as vascular filling by crystalloid saline type crystalloid 0.9%. The patient was intubated without incident after a rapid sequence anesthetic induction including morphine, followed by volume controlled ventilation with mild hyperventilation, sedation with midazolam and fentanyl and introduction of norepinephrine to maintain MAP at 100 mmHg. A full body CT scan was performed, showing intracranial lesions with an 11 mm right subdural hematoma with a hemorrhagic contusion opposite, a 12 mm midline deviation and diffuse cerebral edema. The patient was then brought to the operating room with evacuation of the subdural hematoma through a large craniectomy, anesthesia was provided by intravenous infusion of propofol combined with fentanyl and rocuronium boulus. Intraoperative ventilation was ensured by a controlled volume with 98% SpO2 and 33 mmHg EtCO2. The hemodynamic stability and goal of PAM was achieved by infusion of norepinephrine through a femoral venous line under invasive blood pressure monitoring with a femoral arterial line with vascular filling with isotonic saline, with antibiotic prophylaxis with cefazolin. At the end of the procedure, anisocoria regressed with pupils in miosis. The patient was then transferred to the surgical intensive care unit, where further treatment was performed, maintaining the head elevated at 30°, norepinephrine infusion with a MAP target of 80 to 90 mmHg, sedation with midazolam and fentanyl, volume-controlled ventilation with a target SpO2 95% and PaCO2 between 35 and 40 mmHg, with liveteracetam introduced for seven days, prevention of stress ulcer by proton pump inhibitor and mechanical prophylaxis of pressure points and thromboembolic venous disease. Monitoring included vital signs targets, pupil status and parameters of secondary systemic attacks. A control CT scan was carried out at H24, with the objective of returning the midline to its original position, with evacuation of the subdural hematoma. On the fourth day of his stay, the patient presented a fever related to a pneumopathy acquired under early mechanical ventilation, prolonging the patient's sedation, treated with a third generation cephalosporin with good evolution with the introduction of an enteral feeding by gastric tube. At 8m day, after respiratory improvement, a sedation window was achieved allowing to objectify a neurological status authorizing an extubation. The neurological examination after the extubation showed a GCS of 14 depending on the verbal response with left hemiparesis. During the two days following the extubation, the patient had received non-invasive ventilation and physical respiratory therapy to prevent re-intubation. A swallowing test was performed and oral feeding was initiated. Two days later, the patient presented a depression of the right scalp with an aspect of skin flap syndrome on CT scan (Figure 2) without significant neurological deterioration. Management was conservative with hydration and postural measures. The patient was then transferred to the neurosurgery department for cranioplasty.
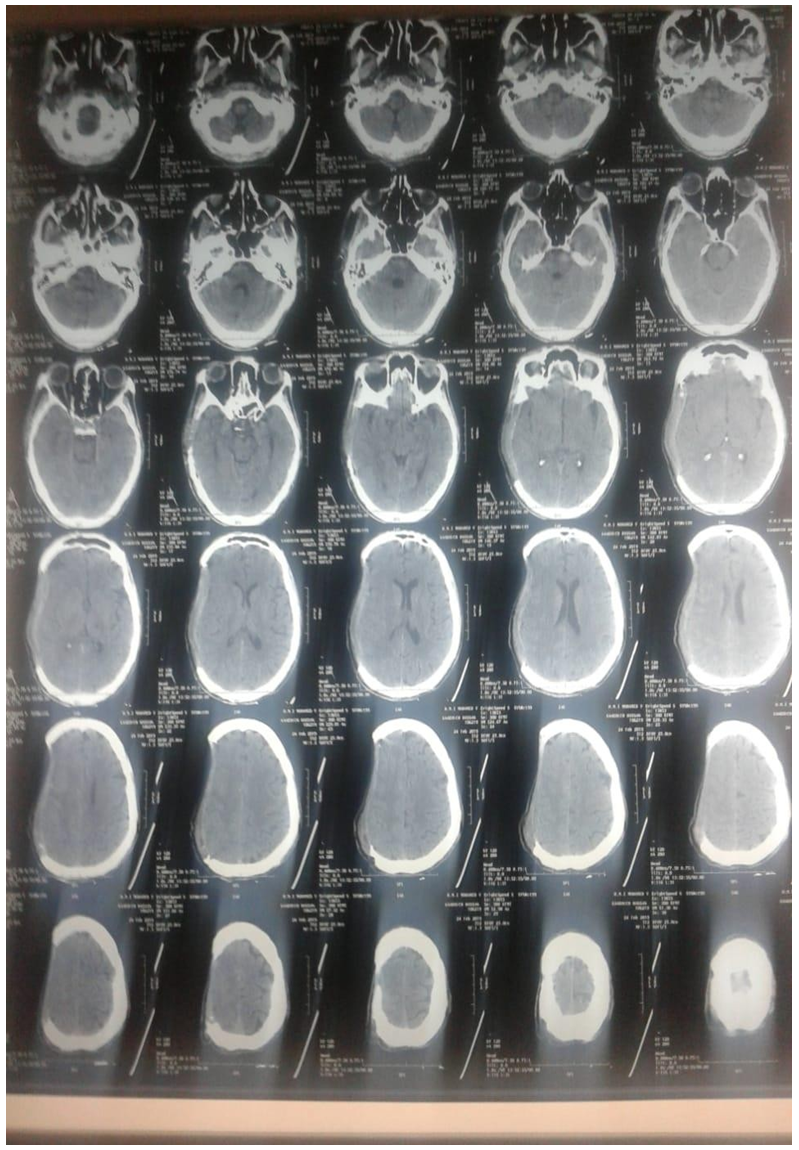
Figure 2: CT scan with Sinking Flap syndrome.
3. Discussion
The sinking skin flap syndrome is a set of neurological manifestations occurring weeks or even months after a large craniectomy performed for different reasons: severe head trauma as in the case of our two observations, malignant ischemic or hemorrhagic stroke [4], improved after a cranioplasty [5]. Since its first description, the literature has been enriched by case reports or small series of cases, making it possible to recognize and describe this syndrome. A review of the literature found 84 cases published up to June 2013 [6], and a second more recent one published in 2016 with different inclusion criteria including 54 observations [5], testifying to the rarity of the syndrome.
The delay between craniectomy and the onset of neurological deterioration varies from 1 month to 1 year, with an average of 13 +/- 8 weeks [6], consistent with the time of onset in our observations, with a very wide interval reported in the literature ranging from a few days (9 days) [7], especially in the presence of promoting factors such as lumbar puncture [8] or ventricular shunt [9], to several years. This more or less delayed delay allows the preventive effect of a possible early cranioplasty to be discussed.
The pathophysiology of this syndrome is still based on unclear theories, the most adopted is that of the direct transmission of atmospheric pressure to the intracranial contents via the bone defect [10], favoured by hypovolemia and/or a decrease in cerebrospinal fluid pressure [11] and consequently brain compression, performing a true external cerebral tamponade [12], inducing an alteration of cerebral blood flow, cerebrospinal fluid circulation and glucose metabolism leading to cortical dysfunction [5]. These consequences may be aggravated by craniectomy [13], as evidenced by their improvement after cranioplasty.
Neurological deterioration presented by patients, who may include symptoms such as headache, dizziness, fatigue, mood or language disorders, discomfort at the operating site, convulsions and altered consciousness. Delayed signs of dysautonomy may occur including orthostatic hypotension, bladder or bowel dysfunction. The physical examination finds a large concave scalp depression opposite the craniectomy, a low Glascow score, with or without motor or sensory deficit. Objective CT shows a paradoxical brain hernia, with skin depression opposite more or less deviation from the midline and compression of the intracranial structures [4]. In the systematic review by Ashayeri et al. the frequency of symptoms was as follows: 61.1% motor weakness, 44.4% cognitive deficit, 29.6% language impairment, 27.8% altered consciousness, 20.4% headache, 18.5% psychosomatic disturbances, 11.1% convulsions or EEG abnormalities and 5.6% cranial pair deficit [5]. In our presentation the first case had presented a convulsion after the onset of the syndrome, with difficulty in assessing cognitive and higher function deficits given the sequelae of the head injury and the possibility of overlap of the two causes in the cognitive outcome.
For the second case, the signs of the syndrome were early, and its transfer to the neurosurgery department for possible cranioplasty did not allow us to evaluate a neurological decline secondary to this syndrome. Therapeutic options are limited, and are aimed at restoring intracranial pressure against atmospheric pressure. Patients are admitted to intensive care with close monitoring. Measures to suppress promoting factors were described with hydration,
Trendelenburg position and temporary closure of external LCS drainage with or without a conservative approach. Other teams performed intrathecal injections of isotonic saline to reverse cerebral herniation [14] or performed a blood patch. However, the therapeutic option that has demonstrated its effectiveness remains cranioplasty, which allows for the reversibility of symptoms. This opens the debate towards the ideal timing of cranioplasty after craniectomy: early during the first three months, or late between the fourth and sixth months after craniectomy, several studies have tried to make a decision but with controversial results.
Sinking skin flap syndrome is a rare complication of depressive craniectomy, which must be known by practitioners, requiring prolonged monitoring of patients who have had this therapy, and raising the question of the safe interval between craniectomy and cranioplasty, the only effective treatment.
4. Conclusion
The role of decompressive craniectomy in neurological improvement in still uncertain, and timing of cranioplasty is more debate: early with unclear neurologic status and preventing the skin flap syndrome or delete after final outcome.
Financial Support
There is no financial support.
Conflicts of Interest
There are no conflicts of interest.
References
- Grant FC, Norcross NC. Repair of cranial defects by Ann Surg 110 (1939): 488-512.
- Yamaura A, Makino H. Neurological deficits in the presence of the sinking skin flap following decompressive Neurol Med Chir (Tokyo) 17 (1977): 43-53.
- Yamaura A, Makino H. Neurological deficits in the presence of the sinking skin flap following decompressive Neurol Med Chir (Tokyo) 1 Pt 1 17 (1977): 43-53.
- Noman Ahmed Jang Khan, Saad Ullah, Waseem Alkilani, et Sinking Skin Flap Syndrome: Phenomenon of Neurological Deterioration after Decompressive Craniectomy. Case Reports in Medicine (2018).
- Kimberly Ashayeri, Eric M Jackson, Judy Huang, et Syndrome of the Trephined: A Systematic Review. Neurosurgery 79 (2016): 525-534.
- Mariam Annan, Bertrand De Toffol, Caroline Hommet, et al. Sinking skin flap syndrome (or Syndrome of the trephined): A review. British Journal of Neurosurgery (2015).
- Joseph V, Reilly P. Syndrome of the trephined. J Neurosurg 111 (2009): 650-652.
- Gschwind M, Michel P, Siclari Life- threatening sinking skin flap syndrome due to CSF leak after lumbar puncture - treated with epidural blood patch. Eur J Neurol 19 (2012): 49.
- Schiffer J, Gur R, Nisim U, et al. Symptomatic patients after Surg Neurol 47 (1977): 231-237.
- Yamaura A, Makino H. Neurological deficits in the presence of the sinking skin flap following decompressive Neurol Med Chir (Tokyo) 17 (1977): 43-53.
- Oyelese AA, Steinberg GK, Huhn SL, et Paradoxical cerebral herniation secondary to lumbar puncture after decompressive craniectomy for a large space-occupying hemispheric stroke: case report. Neurosurgery 57 (2005): 594.
- Nicholas Fawley and Chiedozie Udeh. Sinking Skin Flap Syndrome After Decompressive Craniectomy: A Case Report. A A Pract 11 (2018): 241-243.
- Winkler PA, Stummer W, Linke R, et Influence of cranioplasty on postural blood flow regulation, cerebrovascular reserve capacity, and cerebral glucose metabolism. J Neurosurg 93 (2000): 53-61.
- Cho H, Kim CH, Kim JH, et Paradoxical herniation after decompressive craniectomy for acute subdural hematoma. Journal of Korean Neurosurgical Society 40 (2006): 51-53.
