Self-Monitoring and Self-Efficacy Increase with a Smartphone Application: A Technical Approach to Treating Bipolar Disorder?-Study Protocol for a Clinical Validation Trial
Article Information
Frederike T Fellendorf1*, Carlo Hamm1, Martina Platzer1, Christian Pendl2, Ralph Gruber2, Manfred Weiss2, Nina Dalkner1, Susanne A Bengesser1, Armin Birner1, Robert Queissner1, Rene Pilz1, Hans-Peter Kapfhammer1, Mireille van Poppel3, Eva Z Reininghaus1
1Department of Psychiatry and Psychotherapeutic Medicine, Medical University Graz, Graz, Austria
2Meemo-tec OG, Graz, Austria
3Institute for Sport-science, University of Graz, Graz, Austria
*Corresponding Authors: Frederike T Fellendorf, Department of Psychiatry and Psychotherapeutic Medicine, Medical University Graz, Graz, Austria
Received: 11 June 2019; Accepted: 27 June 2019; Published: 30 June 2019
Citation: Frederike T Fellendorf, Carlo Hamm, Martina Platzer, Christian Pendl, Ralph Gruber, Manfred Weiss, Nina Dalkner, Susanne A Bengesser, Armin Birner, Robert Queissner, Rene Pilz, Hans-Peter Kapfhammer, Mireille van Poppel, Eva Z Reininghaus. Self-Monitoring and Self-Efficacy Increase with a Smartphone Application: A Technical Approach to Treating Bipolar Disorder?-Study Protocol for a Clinical Validation Trial. Journal of Psychiatry and Psychiatric Disorders 3 (2019): 137-148.
Share at FacebookAbstract
Background: Bipolar disorder (BD) is characterized by mood fluctuations, which are often recognized late by those who are affected. Symptom monitoring via smartphone seems to be an inexpensive and feasible method to detect these fluctuations earlier. The aim of an application (app) for smartphones is to record individual activity patterns (e.g. physical activity, sleep-wake rhythm, digital communication) as characteristic for mood swings in individuals with BD.
Methods: We developed the UP! app for Android smartphones, which collects subjective mood daily and continuous data about movement, exercise, sleep duration and intensity of digital communication via GPS and sensors. This pilot trial will include 24 individuals with BD and 24 healthy controls (HC) without a mental disorder. The main aim of this study is to assess the app’s validity to detect mood fluctuations and illness episodes in comparison to a clinical psychiatric interview, validated questionnaires and a fitness tracker. A second aim is to determine whether changes in activity patterns, measured with the app, can detect early warning symptoms of depressive and/or manic episodes. The third aim is the evaluation of users’ acceptance of the app. The recruitment of participants (planned n=48) is ongoing.
Discussion: Behavior patterns recognition via smartphone could present an innovative, technological tool for the early detection of BD episodes and could be valuable for long-term research.
Registration Details: The trial is approved by the ethics committee of the Medical University Graz, Austria (29-290 ex 16/17). The trial is registered at ClinicalTrials.gov as NCT03275714.
Keywords
m-health; Bipolar disorder; E-health; Application; Smartphone; Mood tracking
m-health articles m-health Research articles m-health review articles m-health PubMed articles m-health PubMed Central articles m-health 2023 articles m-health 2024 articles m-health Scopus articles m-health impact factor journals m-health Scopus journals m-health PubMed journals m-health medical journals m-health free journals m-health best journals m-health top journals m-health free medical journals m-health famous journals m-health Google Scholar indexed journals Bipolar disorder articles Bipolar disorder Research articles Bipolar disorder review articles Bipolar disorder PubMed articles Bipolar disorder PubMed Central articles Bipolar disorder 2023 articles Bipolar disorder 2024 articles Bipolar disorder Scopus articles Bipolar disorder impact factor journals Bipolar disorder Scopus journals Bipolar disorder PubMed journals Bipolar disorder medical journals Bipolar disorder free journals Bipolar disorder best journals Bipolar disorder top journals Bipolar disorder free medical journals Bipolar disorder famous journals Bipolar disorder Google Scholar indexed journals E-health articles E-health Research articles E-health review articles E-health PubMed articles E-health PubMed Central articles E-health 2023 articles E-health 2024 articles E-health Scopus articles E-health impact factor journals E-health Scopus journals E-health PubMed journals E-health medical journals E-health free journals E-health best journals E-health top journals E-health free medical journals E-health famous journals E-health Google Scholar indexed journals Application articles Application Research articles Application review articles Application PubMed articles Application PubMed Central articles Application 2023 articles Application 2024 articles Application Scopus articles Application impact factor journals Application Scopus journals Application PubMed journals Application medical journals Application free journals Application best journals Application top journals Application free medical journals Application famous journals Application Google Scholar indexed journals Smartphone articles Smartphone Research articles Smartphone review articles Smartphone PubMed articles Smartphone PubMed Central articles Smartphone 2023 articles Smartphone 2024 articles Smartphone Scopus articles Smartphone impact factor journals Smartphone Scopus journals Smartphone PubMed journals Smartphone medical journals Smartphone free journals Smartphone best journals Smartphone top journals Smartphone free medical journals Smartphone famous journals Smartphone Google Scholar indexed journals Mood tracking articles Mood tracking Research articles Mood tracking review articles Mood tracking PubMed articles Mood tracking PubMed Central articles Mood tracking 2023 articles Mood tracking 2024 articles Mood tracking Scopus articles Mood tracking impact factor journals Mood tracking Scopus journals Mood tracking PubMed journals Mood tracking medical journals Mood tracking free journals Mood tracking best journals Mood tracking top journals Mood tracking free medical journals Mood tracking famous journals Mood tracking Google Scholar indexed journals mood swings articles mood swings Research articles mood swings review articles mood swings PubMed articles mood swings PubMed Central articles mood swings 2023 articles mood swings 2024 articles mood swings Scopus articles mood swings impact factor journals mood swings Scopus journals mood swings PubMed journals mood swings medical journals mood swings free journals mood swings best journals mood swings top journals mood swings free medical journals mood swings famous journals mood swings Google Scholar indexed journals mental disorder articles mental disorder Research articles mental disorder review articles mental disorder PubMed articles mental disorder PubMed Central articles mental disorder 2023 articles mental disorder 2024 articles mental disorder Scopus articles mental disorder impact factor journals mental disorder Scopus journals mental disorder PubMed journals mental disorder medical journals mental disorder free journals mental disorder best journals mental disorder top journals mental disorder free medical journals mental disorder famous journals mental disorder Google Scholar indexed journals psychiatric interview articles psychiatric interview Research articles psychiatric interview review articles psychiatric interview PubMed articles psychiatric interview PubMed Central articles psychiatric interview 2023 articles psychiatric interview 2024 articles psychiatric interview Scopus articles psychiatric interview impact factor journals psychiatric interview Scopus journals psychiatric interview PubMed journals psychiatric interview medical journals psychiatric interview free journals psychiatric interview best journals psychiatric interview top journals psychiatric interview free medical journals psychiatric interview famous journals psychiatric interview Google Scholar indexed journals manic episodes articles manic episodes Research articles manic episodes review articles manic episodes PubMed articles manic episodes PubMed Central articles manic episodes 2023 articles manic episodes 2024 articles manic episodes Scopus articles manic episodes impact factor journals manic episodes Scopus journals manic episodes PubMed journals manic episodes medical journals manic episodes free journals manic episodes best journals manic episodes top journals manic episodes free medical journals manic episodes famous journals manic episodes Google Scholar indexed journals
Article Details
Abbreviations: BD-bipolar disorder; BDI-II-Beck Depression Inventory-II; GPAQ-Global Physical Activity Questionnaire; GPS-global positioning system; HAMD-Hamilton Depression Scale; MSS-Manie-Selbst-Rating-Skala; YMRS-Young Mania Rating Scale PSQI Pittsburgh Sleeping Questionnaire
1. Introduction
1.1 Bipolar disorder
Bipolar affective disorder (BD), is a chronic mental disorder, which often presents a major challenge for those affected, their relatives and the entire health care system. The burden of this disease is mainly due to the characteristic recurrent mood fluctuations (depressive and manic episodes). The occurrence of physical and mental comorbidities and cognitive deficits increase the risk of incapacity to work even more and can increase premature mortality. On a socioeconomic level, the costs for the health care system, through a high number of disability and hospital stays, are tremendous [1]. In both, (hypo)manic and depressive episodes, there are alterations in mood and energy level. However, course and severity of each affective episode vary a lot between individuals with BD [2]. An intact support system of relatives, friends, physicians and therapists is essential for a favourable influence on the development of the disease [3]. A higher number of depressive or manic episodes are associated with a greater likelihood that an individual will not return to a premorbid level of functioning [4]. Every episode, both depressive as well as (hypo)manic episodes lead to a decline of cognitive function and reduced work ability, which is resulting in an all in all worse course of illness [5]. Therefore, a special emphasis is placed on the continuous monitoring and educating of those affected and their relatives, so that they can recognize mood symptoms early and apply appropriate coping strategies.
Furthermore, it has a positive effect on the course of an affective episode if it is detected as early as possible and treated by a combination of psychopharmaceuticals and psychotherapy [6]. In many cases, individuals with BD recognize the beginning of an episode too late to counteract adequately. Early warning signs often manifest in a change in activity behavior, sleep-wake rhythm and communication behavior. The individual extent of physical activity as well as the change in activity could indicate an early stage depressive or manic episode. Increased activity plays an important role as an early symptom of manic phases and reduced activity as an early warning symptom of depressive phases. Moreover, sleep disturbances (problems in falling asleep, sleeping interruptions, too much sleep or insomnia) could be interpreted as a warning sign of depressive as well as (hypo)manic episodes [7]. Disturbances of the sleep-wake rhythm, in turn, can complicate the course of the episode.
To date, the current possibilities in the treatment of BD in the western world allow regular outpatient and inpatient appointments. However, one challenge is recording behavioral patterns timely. Usually, symptoms are recorded retrospectively by personal interview or questionnaires. Therefore, it would be desirable to assess behavior patterns more directly, precisely, individually and more accurate. In the research of etiology and course of the disease it is necessary to obtain valid, objective data on behavioral patterns as well as to monitor data over a longer period of time. Thus, current possibilities seem not valid enough for follow-up studies as well as for the clinical setting.
1.2 E-health
In recent years, there have been major developments in technology in the western world. In 2017, more than 3.58 billion had access to the internet and the number should rise to 4.1 billion in 2021. In 2016, more than 51.5 percent of all households worldwide and around 84 percent in Europe had access to the internet [8]. Internet-enabled mobile phones (smartphones) are the major form of digital communication for a great number of people. This fact opens up new possibilities in the research and treatment of diseases. Electronic health (e-health) is easily accessible for everyone, inexpensive, time-efficient and strengthens autonomy [9]. One major field of e-mental health is the technology of applications (apps) for smartphones (mobile health: m-health; [10]. According to the Federal Statistical Offices, in 2018 about 52 million of over-ten-year-olds in Germany had a smartphone. In Austria between 65.7 (over 65 year olds) and 98.1 percent (16 to 24-year-olds) used their own smartphone for internet access [11, 12]. As many people almost always carry their mobile phone with them, data collection via phones provides an opportunity for continuous recording.
Bauer et al. [13] showed that individuals with BD often use the internet to inform themselves about their disease. Numerous e-health products have been developed in order to provide support in the form of screening, monitoring and therapy. Most of the products for BD treatment provide psychoeducational content with diverse outcomes concerning efficacy [14]. Another possibility underlying e-health arises from the individual entry of symptoms, emotional condition or behavior by the users. With frequent use, individuals have a retrospective overview. Even the knowledge about positive and negative patterns could lead to changes and therefore improve the self-management. Apps can record and process behaviors related to WIFI, accelerometer, global positioning system (GPS), exercise, physical activity, sleep, work and vacation time, phone calls, and digital communications. Most of the data is collected by objective sensors measuring continuously. Instant interpretation and statistical analysis of data can be performed by the app system. This, in turn, can lead to more personalized diagnostics and interventions.
1.3 E-health and mental disorders
In the last few years, there has been an explosion of new e-health products with free or chargeable access. There is evidence that e-health in general and apps in particular are effective, feasible and accepted by individuals with mental disorders. By increasing the elevation of self-management activities, depressive symptoms can be reduced
Palmius and colleagues [16] found in a study using mobile phones an association between geographic movements and depressive symptoms in a cohort of individuals with BD. A recent survey found that health care
professionals’ attitudes towards using apps in clinical practice are quite positive. However, the knowledge of technology as well as products and consequently the use in their daily clinical practice differed a lot [15].
We hypothesize that an app that could reliably measure behavior patterns as well as behavioral changes as a result of altered mood, could be a valuable technical support tool in the treatment of BD. Changes in mood, in symptoms, and behavior could be detected more quickly and individually. The access to feedback of these changes could lead to early recognition of warning signs and faster reactions of the users and could therefore increase self-efficacy. It seems likely that innovative products could bring an advantage as an add-on or self-contained therapy. Moreover, it would be an asset to medical research if individuals with BD brought structured data format of their symptoms to their physicians’ appointments, which could be used for further improvement of understanding and treating BD. In the past, physicians and therapists have had to rely on subjective, retrospective information. Even though it is unlikely that e-mental health products will replace medical and therapeutic treatments, treatment teams and scientists have to evaluate to what extent e-mental health could be a good supporting tool in BD.
1.4 Study-aim
Due to the technological developments of the last decade, the use of an app, could easily be integrated into everyday life with little additional effort for the user. As a cooperation between medical professionals of the special unit for BD of the Medical University Graz in Austria and the company meemo-tec OG, the smartphone app UP! was developed. It collects continuous data about mood, movement, exercise, sleep duration and intensity of digital communication via GPS and sensors. For this purpose the sleep duration is measured by light and motion sensors of the mobile phone. Acceleration sensors and GPS data acquisition allow a calculation of the physical activity. In addition, the current mood situation is assessed daily.
We aim to investigate whether an app can be a modern, appropriate assistance system for a self-determined life for individuals with BD. The main aim is to assess the app’s validity in comparison to a clinical psychiatric interview, validated questionnaires and a fitness tracker to detect manic or depressed episodes. The second aim is to determine whether changes in behavior patterns, measured with UP! coincide with reported early warning symptoms, thus enabling a timely intervention. The third aim is the evaluation of users’ acceptance.
2. Methods
2.1 Setting and participants
This pilot validation study will include 24 individuals with BD and 24 healthy controls without a mental disease (HC). The recruitment is ongoing at the Department of Psychiatry and Psychotherapeutic Medicine at the Medical University of Graz. Individuals with BD are either inpatients or outpatients of the special unit for BD. The diagnosis of BD is confirmed with the Structured Clinical Interview according to DSM-IV. HC are recruited from the general population. Participants have to be of legal age and have given prior written informed consent.
2.2 Design
The trial is approved by the ethics committee of the Medical University Graz, Austria (EK-number: 29-290 ex 16/17) in compliance with the current revision of the Declaration of Helsinki, ICH guideline for Good Clinical Practice and current regulations. The trial is registered at ClinicalTrials.gov as NCT03275714.
2.3 Product
The software solution UP! developed by meemo-tec OG with the medical consult of the Medical University Graz is working on Android smartphones. Data collected automatically include physical-, exercise-, sleep-, and social profiles. Physical activity is determined via acceleration sensor and localization services (GPS, WIFI, etc.) of the smartphone. Different forms of activity can be distinguished: walking, running, cycling, transport in a vehicle and standstill. These activities are recorded on a minute-by-minute basis. The autonomous location and the additional requests of how frequently these places are visited can also detect how often and how long the user is at home, at work, in restaurants, outdoors, etc.. The sleep behavior of the user is recorded using several sensors. As a result, real sleeping events can be distinguished from simple inactivity. Time of falling asleep and waking up, as well as sleep duration and possible interruptions can be determined. The use of smartphones in combination with activity profiles provides information about the (digital) social activity of a person. Smartphone usage is categorized into “Telephony”, “Messaging” (includes WhatsApp, Facebook Messenger, SMS, Snapchat, Signals, etc.), “Social” (Facebook, etc.) and “Media” (includes Netflix, Amazon Fire TV, YouTube, Spotify, etc.). Moreover, users will be asked to enter their mood with seven smiley-options once daily in the evening. The options are depressed, sad, upset, ok, fine, happy, excited (see Figure 1).
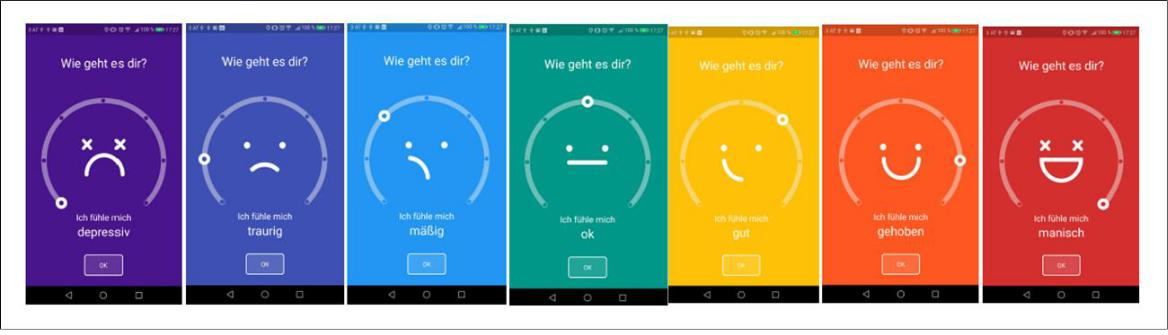
Figure 1: Mood options of UP!.
2.4 Procedure
The procedure of the trial is shown in figure 2. Participants use the app for sixth months after having been instructed to use their smartphone as usual. After one month, three months and sixth months, participants have a scheduled visit at the Department for Psychiatry and Psychotherapeutic Medicine, where a clinically psychiatric report is collected through an interview and standardized questionnaires. For the duration of one month, participants wear a validated fitness tracker on their wrist as well, which collects data 24 hours a day. The tracker measures even the smallest activities by means of acceleration sensors. During the visits, the data is transferred to the local database. At
the first visit, demographic data as well as data about illness duration, number of episodes and ‘normal’ smartphone use is collected. Moreover, mood symptoms in the last two, respectively four weeks are evaluated with the self-rating questionnaires Beck-Depression-Index II (BDI-II; 17) and Manie-Selbst-Rating-Skala (MSS; 18) as well as the rating scales Hamilton Depression Scale (HAMD; 19) and Young Mania Rating Scale (YMRS; 20). Subjective sleep patterns will be evaluated by Pittsburgh Sleeping Questionnaire (PSQI; 21). Data about the subjective physical activity patterns is collected by the Global Physical Activity Questionnaire (GPAQ; 22). Moreover the participants’ satisfaction with the interface of the app and their feedback about the value of the app in the treatment of BD is assessed.
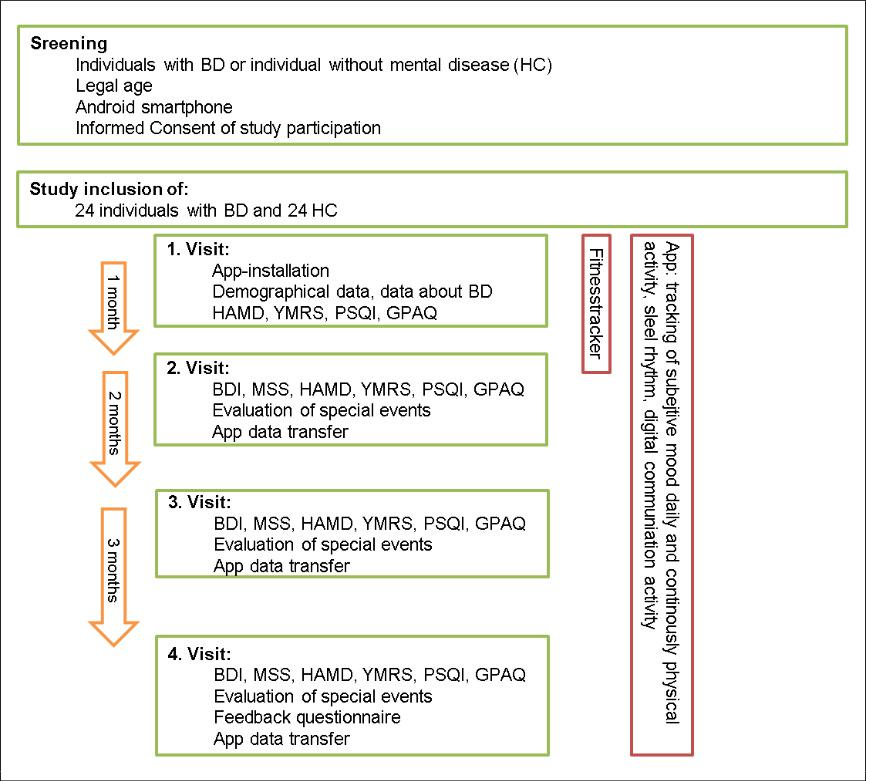
Figure 2: Procedure of the trial.
Note.: BD=Bipolar disorder; HC=Healthy controls; BDI=Beck-Depression-Index, MSS=Manie-Selbst-Rating-Skala, HAMD=Hamilton Depression Scale, YMRS=Young Mania Rating Scale, PSQI=Pittsburgh Sleeping Questionnaire, GPAQ=Global Physical Activity Questionnaire.
2.5 Safety protocol
In general, no risks or harm due to the use of the app are expected. All data is stored locally in the mobile phone. During the visits, the data are transferred via USB to a computer in the study center, which is not useable for online working. Moreover, all further steps of anonymization, storage and analyses of the data go in line with good clinical practice. Due to individual and specific issues accompanying mental disorders, the individuals with BD might experience psychological distress or psychiatric symptoms during the trial participation. An opportunity to talk to a
physician is provided during every planned visit. Psychopharmacological and psychotherapeutical treatment continues as usual.
2.6 Study hypotheses and aims
For the validation of the UP! app, the measurement accuracy of mood as well as sleep, movement and communication behavior are analyzed. Therefore, activity scores and scores for daily rhythms are calculated and compared with the validated questionnaires. Moreover, the fitness tracker data is a valid instrument of measuring activity patterns. Individual patterns of activity and sleep are generated. Changes in the patterns that could be markers for depressive or (hypo)manic episodes are calculated. Subsequently, the results are compared with the clinical interviews. Due to the long observation period of an individual patient, both daytime fluctuations and weekly rhythms can be detected in the mobility pattern. In case of deviations from ‘normal‘ behavior of the individual person, a change in the disease pattern is assumed as an input hypothesis, which is confirmed or rejected by comparison with tracker data and the interviews.
2.7 Analysis
Data of all activities (physical activity, stationary activities, sleep behavior, social behavior, etc.) of the participants must be extracted from the raw data of the smartphone sensors. Activity patterns are determined by the app and plausibility is checked by means of descriptive and graphical statistical methods. Then patterns are validated on the basis of the survey data on individual days. Moreover they are evaluated by means of multivariate statistical methods. From app- and fitness tracker data the time spent sitting per day, as well as light, moderate and intense activity and total activity time (light to intensive) are calculated. To determine validity, Spearman correlation coefficients are calculated. Relations between app data and metric questionnaire data will be investigated by correlations analysis. Moreover, factor analysis and cluster procedures in order to evaluate significant influencing factors of the temporally layered activity behavior with the health data will be conducted. If mood changes present a predictor for activity patterms change will be testet by linear multiple regressions. The HC group is needed to recognize "normal" deviations in patterns such as holidays or being ill. Last, the acceptance of, satisfaction with and demand for that app will be evaluate with the feedback questionnaire of the participants.
2.8 Sample size
Main hypothesis (Validation of the app data) - correlation hypothesis: At least a moderate correlation is expected between the physical activity and sleep data by app and fitnesstracker (correlation coefficient ρ=0.5). Settings for the calculation using G*Power: were test family exact; correlation, bivariate model; a priori; computer required simple size: 5% alpha error level, 95% power, two-sided testing and expectation of a moderate correlation (ρ=0.5). Calculation yielded a sample size of 46 people. Secondary Hypothesis (Can the mood be predicted by the motion and / or sleep data?) - Case number calculation for linear multiple regression: Two Predictors to be Tested (“Movement” and “Sleep Duration”); in total, four predictors are collected (for possible calculations or secondary hypotheses, “phone use” and “length of locations”; Criterion is the mood. Settings for G*Power: Test family: F-
Tests; Fixed model, R^2 increase; a priori; computer required simple size: 5% alpha error level, 95% power, effects of 0.35 should be found. Calculation yielded a sample size of 48 people. For both hypotheses, 48 persons are sufficient according to number of cases calculation. Furthermore, 48 subjects to be tested (observed over a six-month period) appears to be a realistic number from a clinical perspective and experience.
2.9 Dissemination
Results of the trial will be disseminated for publication in peer-reviewed journals and key findings presented at national and international conferences. Moreover, results and conclusions will be included in public health work.
3. Discussion
If the app turns out to be a valid tool, it may represent a supportive health-promoting method in the treatment of BD. One advantage for individuals with BD could be the confrontation with their behavior patterns. The early recognition of warning signs and symptoms could result in faster and more specific countermeasures. If social-sleep- and activity patterns are positively affected, the outcome will improve [23]. Furthermore, with the outcome of a favorable validation of the app, further research projects could be created on the basis of that, to investigate the individual behavioral patterns of the disease.
E-health products in general and m-health products in particular could be useful in a variety of ways. Firstly, the tools are widely used in today's everyday life and thus would provide the affected individuals with easy access. Secondly, by using their own internet access or mobile phone, individuals do not need to relay on medical appointments as much, thereby increasing their self- autonomy. Thirdly, e-health offers the possibility of minimizing outpatient waiting times and waiting times for appointments, thus increasing the quantity of care. A study found that individuals with mental illness who were treated by regular video conferencing of experienced psychotherapists were no less satisfied than individuals who had personal medical appointments [24]. Fourthly, even long distances can be overcome through the use of apps. This would have a positive impact on regions in which medical care is not readily available. Fifthly, it would also have positive effects on the environment by minimizing ways travel to and from the hospitals or doctor’s office. Sixthly, by saving time and using existing structures, it is possible to conduct cheaper diagnostics and therapies, thereby reducing costs for the entire healthcare system. It was reported that in 2004 the cost to the healthcare system for only one year per individual with depression was € 4884 and € 7779 per individual with bipolar disorder [25]. In a study of Ly et al. [26] it was shown that nearly twice as many individuals suffering from depression could be treated with e-therapy and m-therapy than with face-to-face therapy. Seventhly, the stigma around mental illness could be reduced. Affected individuals are able to inform themselves anonymously on the Internet before consulting a professional. This will likely counteract stigma as arranging an appointment with a professional presents a psychological barrier for many people. In addition, self-help forums could potentially help reduce an individual’s feeling of being alone with their illness. Eighthly, individuals with mental disorders have expressed a desire to use e-health products. A study group in the USA found, that 62.5 percent of 320 individuals with mental disorders had a smartphone and that the interest for utilizing it for symptom monitoring was measured
at 70.6 percent [27]. Lastly, it would be an asset to medical research if individuals brought records of their symptoms to their treatment appointments. In the past, physicians and therapist have had to rely on subjective, retrospective information. It is obvious that e-health products cannot replace the current treatment, but it may be a good additional opportunity for clinical and research work. Up to date, there are just a few products validated scientifically. Therefore, one of the challenges of the future is to conduct more validation studies.
However, there are some similar products for smartphones for BD developed by European study groups. Up to now, there are a few study protocols of clinical validation and evaluation trials published, but final results and evidence for efficacy is rare [28-31].
3.1 Summary
Since early detection of depressive and manic episodes is a major challenge for individuals with BD, early treatment would have a positive effect on the course of the episodes. Warning-symptoms of affective episodes often manifest in changes in physical activity, communication behaviour and sleep-wake rhythm. Therefore, early detection of warning signs by detecting changes in behavioral patterns would be a useful tool in the treatment of BD. Up to now, there was a lack of precise measurability of physical activity, changes in mood and sleep behavior. A validated instrument providing such data objectively would be of great benefit to further research projects. Consequently, UP! was developed to record individual behaviour patterns on the basis of objective, continuous and individual data.
3.2 Outlook
The final product of UP! will allow users to see graphical feedback of their working time, physical activity, sleeping rhythms and mood. Even the visualization and knowledge of patterns has the potential of changing the mind set of users and encouraging “healthy behaviours” such as doing exercises consciously or take care about an awake-sleep-rhythm. Moreover the users will be able to choose between typical warning signs of BD or even to enter their own experienced ones. If it turns out, that UP! is a valid measure, it will accurately recognize deviations of behaviour and interpret them as potential early warning symptoms. The product will then respond with feedback to the user by asking them for potential warning symptoms. Again, if individuals with BD become aware of possible illness signs, they can respond adequately. If individuals with BD were educated about their BD they will have a lot of opportunities to handle beginning symptoms and thus strengthening their self-autonomy. However, every user will get some psychoeducative suggestions. The aim of UP! is to increase the possibilities for self-management and self-efficacy and prevent relapses, severe episode courses and hospitalization.
4. Conclusion
Behavior patterns recognition (physical activity, sleep-wake-rhythm, digital communication) via smartphone could present an innovative, technical tool in the early detection of BD episodes and could moreover be used for long-term research. As the adherence of the principals of evidence based medicine is desirable, validation of products are necessary and important.
Declarations
Ethics Approval and Consent to Participate
The trial is approved by the ethics committee of the Medical University Graz, Austria (EK-number: 29-290 ex 16/17) in compliance with the current revision of the Declaration of Helsinki, ICH guideline for Good Clinical Practice and current regulations. The trial is registered at ClinicalTrials.gov as NCT03275714.
Consent for Publication
Not applicable.
Availability of Data and Materials
Up to now, there is no data applicable. The datasets of study results will be available from the corresponding author on reasonable request.
Competing Interests
The authors declare that they have no competing interests.
Funding
Costs for the monitoring of the study are taken by meemo-tec OG.
Authors’ contributions
FF, EZ, CP, RG and MW were mainly involved in the product development. FF and EZ designed the study and contributed to the recruitment of the participants. FF, CH and RP are responsible for the implementation of the study visits as well as for the data storage. All authors will partly be involved in the statistical analyses and in the interpretation of the results. All authors read and drafted the manuscript.
Acknowledgements
Not applicable.
References
- Kessing LV, Vradi E, Andersen PK. Life expectancy in bipolar disorder. Bipolar Disord 17 (2015): 543-548.
- Suppes T, Dennehy EB, Wells Gibbons E. The longitudinal course of bipolar disorder. The Journal of clinical psychiatry 9 (2000): 23-30.
- Vargas-Huicochea I, Berenzon S, Rascon ML, et al. A bittersweet relationship: What does it mean to be the caregiver of a patient with bipolar disorder?. International Journal of Social Psychiatry 64 (2018): 207-216.
- Perlis RH, Ostacher MJ, Patel JK, et al. Predictors of recurrence in bipolar disorder: primary outcomes from the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). American Journal of Psychiatry 163 (2006): 217-224.
- Harvey PD. Mood symptoms, cognition, and everyday functioning: in major depression, bipolar disorder, and schizophrenia. Innovations in clinical neuroscience 8 (2011): 14.
- Berk M, Brnabic A, Dodd S, et al. Does stage of illness impact treatment response in bipolar disorder? Empirical treatment data and their implication for the staging model and early intervention. Bipolar disorders 13 (2011): 87-98.
- Harvey AG, Talbot LS, Gershon A. Sleep disturbance in bipolar disorder across the lifespan. Clin Psychol (New York) 16 (2009): 256-277.
- Rabe L. Internet usage statistics worldwide. Statista (2019).
- Marzano L, Bardill A, Fields B, et al. The application of mHealth to mental health: opportunities and challenges. The Lancet Psychiatry 2 (2015): 942-948.
- mHealth: new horizons for health through mobile technologies. Geneva: WHO (2011).
- Two-thirds of smartphone users use security software. Destatis Federal Statistical Office (2018).
- Statistik Austria (2018).
- Bauer R, Conell J, Glenn T, et Internet use by patients with bipolar disorder: results from an international multisite survey. Psychiatry research 242 (2016): 388-394.
- Hidalgo-Mazzei D, Mateu A, Reinares M, et al. Psychoeducation in bipolar disorder with a SIMPLe smartphone application: feasibility, acceptability and satisfaction. Journal of Affective Disorders 200 (2016): 58-66.
- Kerst A, Zielasek J, Gaebel W. Smartphone applications for depression: a systematic literature review and a survey of health care professionals’ attitudes towards their use in clinical practice. European archives of psychiatry and clinical neuroscience (2019): 1-14.
- Palmius N, Tsanas A, Saunders KEA, et al. Detecting bipolar depression from geographic location data. IEEE Transactions on Biomedical Engineering 64 (2017): 1761-1771.
- Wintjen L, Petermann F. Beck-Depressions-Inventar Revision (BDI-II). Zeitschrift für Psychiatrie, Psychologie und Psychotherapie 58 (2010): 243-245.
- Krüger S, Braunig P, Shugar G. Manie-Selbstbeurteilungsskala: MSS, deutsche Bearbeitung des Self-Report manic inventory (SRMI). Beltz (1997).
- Hamilton M. The Hamilton rating scale for depression. In Assessment of depression. Springer, Berlin, Heidelberg (1986): 143-152.
- Young RC, Biggs JT, Ziegler VE, et al. A rating scale for mania: reliability, validity and sensitivity. The British Journal of Psychiatry 133 (1978): 429-435.
- Buysse DJ, Reynolds III CF, Monk TH, et al. The Pittsburgh Sleep Quality Index: a new instrument forpsychiatric practice and research. Psychiatry research 28 (1989): 193-213.
- Armstrong T, Bull F. Development of the world health organization global physical activity questionnaire (GPAQ). Journal of Public Health 14 (2006): 66-70.
- Miklowitz DJ, Otto MW, Frank E, et al. Intensive psychosocial intervention enhances functioning in patients with bipolar depression: results from a 9-month randomized controlled trial. American Journal of Psychiatry 164 (2007): 1340-1347.
- Urness D, Wass M, Gordon A, et al. Client acceptability and quality of life–telepsychiatry compared to in-person consultation. Journal of telemedicine and telecare 12 (2006): 251-254.
- Andlin-Sobocki P, Jonsson B, Wittchen HU, et al. Cost of disorders of the brain in Europe. European Journal of neurology 12 (2005): 1-27.
- Ly KH, Topooco N, Cederlund H, et al. Smartphone-supported versus full behavioural activation for depression: a randomised controlled trial. PLoS One 10 (2015): e0126559.
- Torous J, Chan SR, Tan SYM, et al. Patient smartphone ownership and interest in mobile apps to monitor symptoms of mental health conditions: a survey in four geographically distinct psychiatric clinics. JMIR Mental Health 1 (2014): 5.
- Faurholt-Jepsen M, Vinberg M, Christensen EM, et al. Daily electronic self-monitoring of subjective and objective symptoms in bipolar disorder-the MONARCA trial protocol (MONitoring, treAtment and pRediCtion of bipolAr disorder episodes): a randomised controlled single-blind trial. BMJ open 3 (2013): e003353.
- Hidalgo-Mazzei D, Mateu A, Reinares M, et al. Self-monitoring and psychoeducation in bipolar patients with a smart-phone application (SIMPLe) project: design, development and studies protocols. BMC psychiatry 15 (2015): 52.
- Mühlbauer E, Bauer M, Ebner-Priemer U, et al. Effectiveness of smartphone-based ambulatory assessment (SBAA-BD) including a predicting system for upcoming episodes in the long-term treatment of patients with bipolar disorders: study protocol for a randomized controlled single-blind trial. BMC psychiatry 18 (2018): 349.
- Slofstra C, Klein NS, Nauta MH, et al. Imagine your mood: study design and protocol of a randomized controlled micro-trial using app-based experience sampling methodology to explore processes of change during relapse prevention interventions for recurrent depression. Contemporary clinical trials communications 7 (2017): 172-178.
