Radiological Diagnosis of Autoimmune Pancreatitis (AIP) Type 1: A Case Report
Article Information
Diallo Moustapha1, Amar Ndeye Isseu2, Ndaw Mame Diarra Bousso2, Diack Aminata2, Diop Massamba2, Diouf Cheikh Tidiane2, Fall Amath2, Ndiaye Abdou Rahmane2, Soko Thierno Omar2, Mbengue Ababacar2
1Army Hospital Specialist, Dakar Principal Hospital, Medical Imaging Department, Dakar-Senegal.
2Dakar Principal Hospital, Medical Imaging Department, Dakar, Senegal.
*Corresponding Author: Dr Moustapha DIALLO, Army Hospital Specialist, Dakar Principal Hospital, Medical Imaging Department, Dakar-Senegal.
Received: 08 July 2023; Accepted: 18 July 2023; Published: 28 July 2023
Citation: Diallo Moustapha, Amar Ndeye Isseu, Ndaw Mame Diarra Bousso, Diack Aminata, Diop Massamba, Diouf Cheikh Tidiane, Fall Amath, Ndiaye Abdou Rahmane, Soko Thierno Omar, Mbengue Ababacar. Radiological Diagnosis of Autoimmune Pancreatitis (AIP) Type 1: A Case Report.. Journal of Radiology and Clinical Imaging. 6 (2023): 161-165
Share at FacebookAbstract
Introduction: Autoimmune pancreatitis (AIP) is a rare form of chronic inflammatory pancreatitis. It is the pancreatic localization of a systemic autoimmune disease characterized by a fibro-inflammatory lymphoplasmacytic infiltration associated with a serum elevation of IgG4. The imaging plays a key role in diagnosis.
Objective: To display the contribution of imaging in the diagnosis of type 1 PAI
Observation: A 38-year-old man was received for the exploration of cholestatic jaundice that has been evolving for 3 weeks. The CT scan showed diffuse hypertrophy of the pancreas with loss of lobulations and presence of a hypodense peripancreatic halo, without peripancreatic fat infiltration. There was a dilation of the bile ducts upstream of a stenosis with enhancement of the distal bile duct, associated with a focus of hypodensity at the cortical base and not enhanced in the left kidney. The diagnosis of pancreatitis and autoimmune cholangitis was evoked. Magnetic Resonance Imaging (MRI) performed secondarily confirmed pancreatic and biliary involvement with diffuse pancreatic hypertrophy in T1 hyposignal, peripheral halo in T2 and T1 hyposignal and long stenosis without dilatation upstream of the main pancreatic duct. The walls of the bile ducts and of the gallbladder were thickened and moved upward. The dosage of serum IgG4 had shown an elevation that reached up to 10 times normal. Drawing on imaging and serum IgG4 levels, the diagnosis of autoimmune pancreatitis type I, associated with chonlangitis and autoimmune nephritis, was taken as a conclusion. The clinical and biological abnormalities have completely regressed under corticosteroid therapy with a disappearance of the bilio-pancreatic abnormalities on the control imaging.
Conclusion: Type 1 PAI is a rare condition whose diagnosis in practice is based on a range of arguments including typical pancreatic involvement on imaging, elevation of IgG4, histological evidence, and or regression of abnormalities on corticosteroid therapy
Keywords
Autoimmune pancreatitis; IgG 4; Scanner; MRI.
Autoimmune pancreatitis articles; IgG 4 articles; Scanner articles; MRI articles.
Autoimmune pancreatitis articles Autoimmune pancreatitis Research articles Autoimmune pancreatitis review articles Autoimmune pancreatitis PubMed articles Autoimmune pancreatitis PubMed Central articles Autoimmune pancreatitis 2023 articles Autoimmune pancreatitis 2024 articles Autoimmune pancreatitis Scopus articles Autoimmune pancreatitis impact factor journals Autoimmune pancreatitis Scopus journals Autoimmune pancreatitis PubMed journals Autoimmune pancreatitis medical journals Autoimmune pancreatitis free journals Autoimmune pancreatitis best journals Autoimmune pancreatitis top journals Autoimmune pancreatitis free medical journals Autoimmune pancreatitis famous journals Autoimmune pancreatitis Google Scholar indexed journals IgG 4 articles IgG 4 Research articles IgG 4 review articles IgG 4 PubMed articles IgG 4 PubMed Central articles IgG 4 2023 articles IgG 4 2024 articles IgG 4 Scopus articles IgG 4 impact factor journals IgG 4 Scopus journals IgG 4 PubMed journals IgG 4 medical journals IgG 4 free journals IgG 4 best journals IgG 4 top journals IgG 4 free medical journals IgG 4 famous journals IgG 4 Google Scholar indexed journals Scanner articles Scanner Research articles Scanner review articles Scanner PubMed articles Scanner PubMed Central articles Scanner 2023 articles Scanner 2024 articles Scanner Scopus articles Scanner impact factor journals Scanner Scopus journals Scanner PubMed journals Scanner medical journals Scanner free journals Scanner best journals Scanner top journals Scanner free medical journals Scanner famous journals Scanner Google Scholar indexed journals MRI articles MRI Research articles MRI review articles MRI PubMed articles MRI PubMed Central articles MRI 2023 articles MRI 2024 articles MRI Scopus articles MRI impact factor journals MRI Scopus journals MRI PubMed journals MRI medical journals MRI free journals MRI best journals MRI top journals MRI free medical journals MRI famous journals MRI Google Scholar indexed journals Radiological articles Radiological Research articles Radiological review articles Radiological PubMed articles Radiological PubMed Central articles Radiological 2023 articles Radiological 2024 articles Radiological Scopus articles Radiological impact factor journals Radiological Scopus journals Radiological PubMed journals Radiological medical journals Radiological free journals Radiological best journals Radiological top journals Radiological free medical journals Radiological famous journals Radiological Google Scholar indexed journals CT scan articles CT scan Research articles CT scan review articles CT scan PubMed articles CT scan PubMed Central articles CT scan 2023 articles CT scan 2024 articles CT scan Scopus articles CT scan impact factor journals CT scan Scopus journals CT scan PubMed journals CT scan medical journals CT scan free journals CT scan best journals CT scan top journals CT scan free medical journals CT scan famous journals CT scan Google Scholar indexed journals diffuse hypertrophy articles diffuse hypertrophy Research articles diffuse hypertrophy review articles diffuse hypertrophy PubMed articles diffuse hypertrophy PubMed Central articles diffuse hypertrophy 2023 articles diffuse hypertrophy 2024 articles diffuse hypertrophy Scopus articles diffuse hypertrophy impact factor journals diffuse hypertrophy Scopus journals diffuse hypertrophy PubMed journals diffuse hypertrophy medical journals diffuse hypertrophy free journals diffuse hypertrophy best journals diffuse hypertrophy top journals diffuse hypertrophy free medical journals diffuse hypertrophy famous journals diffuse hypertrophy Google Scholar indexed journals apparent diffusion coefficient articles apparent diffusion coefficient Research articles apparent diffusion coefficient review articles apparent diffusion coefficient PubMed articles apparent diffusion coefficient PubMed Central articles apparent diffusion coefficient 2023 articles apparent diffusion coefficient 2024 articles apparent diffusion coefficient Scopus articles apparent diffusion coefficient impact factor journals apparent diffusion coefficient Scopus journals apparent diffusion coefficient PubMed journals apparent diffusion coefficient medical journals apparent diffusion coefficient free journals apparent diffusion coefficient best journals apparent diffusion coefficient top journals apparent diffusion coefficient free medical journals apparent diffusion coefficient famous journals apparent diffusion coefficient Google Scholar indexed journals Magnetic Resonance Imaging articles Magnetic Resonance Imaging Research articles Magnetic Resonance Imaging review articles Magnetic Resonance Imaging PubMed articles Magnetic Resonance Imaging PubMed Central articles Magnetic Resonance Imaging 2023 articles Magnetic Resonance Imaging 2024 articles Magnetic Resonance Imaging Scopus articles Magnetic Resonance Imaging impact factor journals Magnetic Resonance Imaging Scopus journals Magnetic Resonance Imaging PubMed journals Magnetic Resonance Imaging medical journals Magnetic Resonance Imaging free journals Magnetic Resonance Imaging best journals Magnetic Resonance Imaging top journals Magnetic Resonance Imaging free medical journals Magnetic Resonance Imaging famous journals Magnetic Resonance Imaging Google Scholar indexed journals Pancreatology articles Pancreatology Research articles Pancreatology review articles Pancreatology PubMed articles Pancreatology PubMed Central articles Pancreatology 2023 articles Pancreatology 2024 articles Pancreatology Scopus articles Pancreatology impact factor journals Pancreatology Scopus journals Pancreatology PubMed journals Pancreatology medical journals Pancreatology free journals Pancreatology best journals Pancreatology top journals Pancreatology free medical journals Pancreatology famous journals Pancreatology Google Scholar indexed journals
Article Details
1. Introduction
Acute pancreatitis remains mainly of alcoholic or biliary origin. After excluding these causes, rarer etiologies are sought, among which we find autoimmune pancreatitis. There are two types (PAI type 1 and 2) with distinct anatomo-pathological forms and different clinical profiles [1]. The type 1 autoimmune pancreatitis that we have presented through this clinical case, is part of the systemic IgG4 disease, and affects elderly patients (between 50 to 70 years old). It is characterized by the fact that it first reaches several organs by the same fibro-inflammatory infiltrate. Its diagnosis is based on imaging, biological (serum IgG4 assay), evolutionary (remission by corticosteroid therapy) and histological criteria [2]. Thus, through a short description of a particular clinical case of PAI type 1, because it occurs in a young subject, we present the clinical-biological and diagnostic aspects of this rare entity and thus show the contribution of imaging.
2. Observation
A 38-year-old man was seen for the exploration of cholestatic jaundice that has been evolving for 3 weeks and for which an abdominal CT scan was performed. The protocol covered the volume of the abdomen and pelvis with volume acquisitions in thin slices before and after injection of contrast product and at the arterial and portal stages. It showed diffuse hypertrophy of the pancreas with loss of lobulations and the presence of a hypodense peripancreatic halo, without infiltration of peripancreatic fat (figure 1). There was dilatation of the bile ducts upstream of a stenosis with parietal enhancement of the distal bile duct, associated with a focus of parenchymal hypodensity at the cortical base of the right kidney. The diagnosis of autoimmune pancreatitis and cholangitis was evoked. Magnetic Resonance Imaging (MRI) was performed secondarily with T1, T2, cholangio-MRI and dynamic T1 weighted sequences after gadolinium injection and fat removal. It confirmed the pancreatic and biliary infection with diffuse hypertrophy of the pancreas in hyposignal T1 and a peripheral halo in hyposignal T2 and T1. There was a regular stenosis of the lower bile duct associated with a long stenosis without dilatation upstream of the main pancreatic duct. The walls of the bile ducts and gallbladder were thickened and rmoved upward (Figure 2). The serum IgG4 assay was 3.634 g/l, an increase that is 10 times normal. On the basis of imaging and serum IgG4 levels, the diagnosis of type I autoimmune pancreatitis, associated with cholangitis and autoimmune nephritis, was retained, falling within the nosological framework of the disease IgG4. The clinical and biological abnormalities completely regressed under corticosteroid therapy with a disappearance of the bilio-pancreatic and renal abnormalities on the control imaging (figure 3)
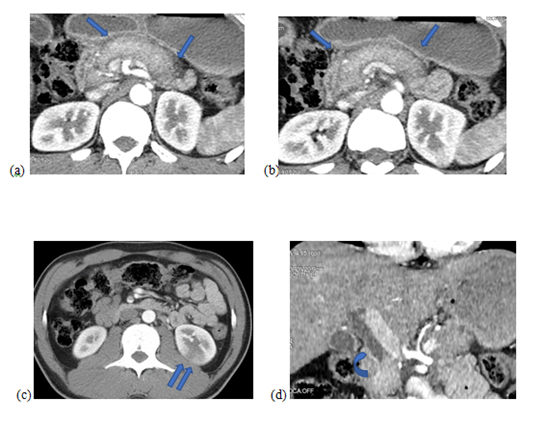
Figure 1: Abdominal CT scan with axial sections in the arterial phase after contrast injection (a, b and c) and coronal reconstruction (d): Diffuse hypertrophy of the pancreas with loss of lobulations and peripancreatic hypodense halo (single arrow). Posterior cortical hypodensity of the left kidney in connection with a hearth of nephritis (double arrow). Lower bile duct stenosis (curved arrow)
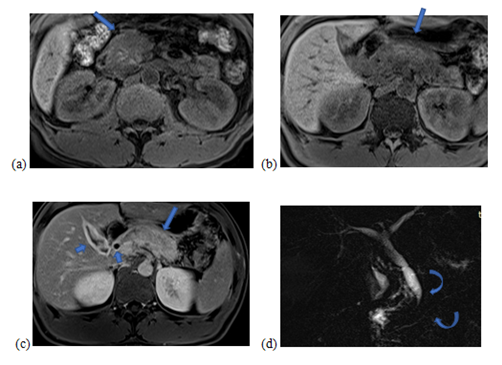
Figure 2: Abdominal MRI with axial slices in T1-weighted sequences with fat suppression (a and b), T1 with fat suppression and portal phase Gadolinium injection (c) and 3D Bili-MRI sequence (d): Edematous infiltration diffuse pancreas in T1 hyposignal with a peripancreatic halo in T1 hyposignal. Heterogeneous enhancement of the pancreas after injection of contrast (long arrows). Thickening and intense contrast uptake of the wall of the bile ducts (short arrows). Filiform and regular stenosis of the lower bile duct and long stenosis of the main pancreatic duct without dilation upstream (curved arrows).
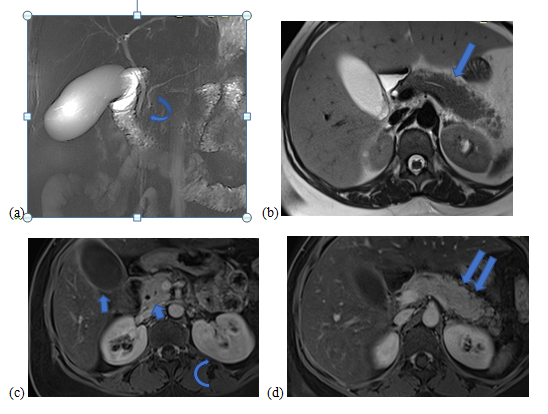
Figure 3: Control abdominal MRI after treatment: 3D Bili-MRI sequence (a), axial slices in T2-weighted sequence (b) and T1 with fat suppression and Gadolinium injection at the portal phase (c) and (d): Normalization of the stenosis of the lower bile duct and the main pancreatic duct (curved arrow), disappearance of the peripancreatic halo with normal aspect of the lobulations of the pancreas (long arrow). Homogeneous enhancement of the pancreas (double arrow) and normal appearance of the wall of the gallbladder and bile ducts (short arrows). The left renal focal nephritis also regressed (arc arrow).
3. Discussion
It was in 1961 that Sarles et al [3] coined the term idiopathic chronic pancreatitis for a patient who presented with obstructive jaundice and hypergammaglobulinemia. Yoshida et al. [4] in 1995 introduced the term “autoimmune pancreatitis” (AIP). There are two subtypes, type 1 PAI and type 2 PAI. Type 1 autoimmune pancreatitis, which is under study in our presentation, is characterized by fibrotic and inflammatory pancreatic damage associated with a specific elevation serum IgG4, as well as possible damage to other organs. Infiltration of multiple organs by IgG4+ plasma cells defines systemic IgG4 disease (IgG4-related sclerosing disease) [2]. Autoimmune pancreatitis (AIP) remains little known and is part of the differential diagnosis of acute non-alcoholic-non-biliary pancreatitis. Type 1 PAI is very common in Asia (more than 90% of Japanese series), and represents less than 20% of Western series [5]. The age of diagnosis is classically between 50 and 70 years with a predominance of the male sex [2]. Our patient, on the other hand, was 38 years old and male. Clinically, patients present with obstructive jaundice and chronic, mild and recurrent abdominal pain, as was the case in our patient [6]. Isolated or associated extra-pancreatic damage is multiple, synchronous or spaced out over time. They concern the biliary tree (cholangitis), the thorax (pulmonary nodules, mediastinal fibrosis, adenopathy), the retroperitoneum (retroperitoneal fibrosis, chronic periaortitis), the salivary glands (sclerosing sialadenitis), the kidneys (interstitial nephritis) and orbits (pseudolymphoma ). Intestinal infection is rare. Our patient had cholangitis and nephritis associated with pancreatitis.The imaging examinations of choice for diagnosis are CT scan with injection of contrast product and Magnetic Resonance Imaging (MRI) with cholangio-MRI sequences [6]. The CT is useful for looking for changes in the pancreatic parenchyma and excluding malignancies, while magnetic resonance cholangiopancreatography more specifically assesses abnormalities of the bile and pancreatic ducts [6]. Three types of morphological change in the pancreas are described: diffuse, focal and multifocal. Diffuse infection is characterized by enlargement of the pancreas with loss of lobulations giving the "sausage" appearance. There is no peripancreatic fat infiltration. A peripancreatic halo, hypodense on the CT scan and hyposignal T1 and T2 on the MRI, is noted late after injection due to the presence of fibro-inflammatory cells around the pancreas [6]. This evocative appearance of diffuse PAI was found in our patient. The differential diagnosis of this form is oedemato-interstitial pancreatitis. The focal form presents as a hypovascular mass in the arterial phase and enhancing late after injection of contrast and without infiltration of peripancreatic fat. It therefore poses the differential diagnosis with pancreatic adenocarcinoma. [6] The sign of the pancreatic duct crossing the mass and highlighted on the cholangio-pancreatography-MRI points towards the PAI. [7]. Choi et al, showed that an apparent diffusion coefficient (ADC) value of less than 0.9407 × 10−3 mm2/s on diffusion-weighted MRI can be useful in differentiating AIP from ductal adenocarcinoma focal. This hypercellularity in PAI is due to a dense fibro-inflammatory, plasma cell and lymphocyte infiltration, on an edematous background. Other differential elements are noted. The adenocarcinoma remains hypovascular at all injection times and presents a T2 hyposignal on MRI. The focal pancreatitis enhancement happens late and presents as a T2 hypersignal due to inflammation and fibrosis. [6] Concerning the ductal infection of the PAI, it is long (greater than 1/3 of the ductal length) or characterized by single or multiple short stenoses and without significant dilation upstream. [6]. The aspect of long stenosis of the main pancreatic duct was found on the MRI of our patient. When there is associated biliary infection, it is characterized by long irregular strictures involving the distal part of the common bile duct and the right and left hepatic ducts with parietal enhancement [6]. Biliary infection in our patient had the same characteristics. The differential diagnosis is made with primary sclerosing cholangitis which is characterized by short multifocal stenoses with a pearly appearance. Nephritis associated with IgG4 is the most common condition after pancreatitis and is characterized on imaging by focal parenchymal hypodensity as illustrated in our patient [2]. The diagnosis of PAI is based on biological, clinical, imaging and histopathological criteria.The Asian recommendations, revised in 2009 by Okazaki et al. [9], only include three diagnostic criteria (imaging, serological and histological) whereas the American HISORt criteria [10], developed at the Mayo Clinic, and the international association of pancreatology in 2011 include 5 main diagnostic criteria (histological, imaging, serology, involvement of other organs and response to corticosteroid therapy) [11]. Imaging is an integral part of all diagnostic recommendations with a prominent place. The signs of PAI being typical and almost topognomonic. In the case of our patient, the definite diagnosis of PAI was retained, according to the recommendations of the International Association of Pancreatology, on the typical imaging criteria, the serological criteria, the infection of other organs (nephritis and cholangitis) and on a clinical-biological and radiological response after corticosteroid treatment.
4. Conclusion
Type 1 PAI is a rare condition whose diagnosis in practice is based on a range of arguments including typical pancreatic infection on imaging, elevation of IgG4, histological evidence, and or regression of abnormalities under corticosteroid therapy. Imaging occupies an important place in the diagnostic criteria with almost pathognomic signs that have yet to be known.
References
- Rebours M, Levy P. Les deux formes de la pancréatite auto-immune. Press Med. 41 (2012) :580-592.2
- Bennis M, Roy T, Atto YN, et al. La maladie systémique à IgG4, une cause rare de pancréatite aigüe sévère. Louvain Med 2020, 139 (2020) : 185-191
- Sarles H, Sarles JC, Muratore R, et al. Chronic inflammatory sclerosis of the pancreas. An autonomous pancreatic disease? Am J Dig Dis, 6 (1961): 688-698
- Yoshida K, Toki F, Takeuchi T, et al. Chronic pancreatitis caused by an autoimmune abnormality. Proposal of the concept of autoimmune pancreatitis. Dig Dis Sci, 40 (1995): 1561-1568
- Ebbo M, Grados A, Daniel L, et al. Maladie systemique associée aux IgG4 : une pathologie émergente? Mise au point et revue de la littérature. Rev Med Intern. 33 (2012): 23-34.
- Binit SA, Archana R. Autoimmune Pancreatitis. Pol J Radiol, 82 (2017): 233-239
- Ichikawa T, Sou H, Araki T, et al, Ishigame K, et al. Duct-penetrating sign at MRCP : Usefulness for differentiating inflammatory pancreatic mass from pancreatic carcinomas. Radiology, 221 (2001): 107-116
- Choi SY, Kim SH, Kang TW, et al. Differentiating mass-forming autoimmune pancreatitis from pancreatic ductal adenocarcinoma on the basis of contrast-enhanced MRI and DWI findings. Am J Roentgenol, 206 (2016): 291-300
- Okazaki K, Kawa S, Kamisawa T, et al. Japanese clinical guidelines for autoimmune pancreatitis. Pancreas, 38 (2009): 849-866
- Chari ST, Smyrk TC, Levy MJ, et al. Diagnosis of autoimmune pancreatitis: The Mayo Clinic experience. Clin Gastroenterol Hepatol, 4 (2006): 1010-1016
- Shimosegawa T, Chri S, Frulloni L, et al. International Consensus Diagnostic Criteria for Autoimmune Pancreatitis. Guidelines of the International Association of Pancreatology. Pancreas. 2011: 352-358
