Pseudo-Pseudo Meigs’ Syndrome in a Patient with Systemic Lupus Erythematosus Flare
Article Information
Muhammad Sohaib Asghar1*, Abubakar Tauseef1, Maryam Zafar1, Hiba Shariq1, Mariam Amir1, Uzma Rasheed2, Nimra Shaikh2, Gul Muhammad Memon2
1Dow University Hospital, Dow University of Health Sciences, Karachi, Pakistan
2Liaquat National Hospital and Medical College, Karachi, Pakistan
*Corresponding Author: Muhammad Sohaib Asghar, Dow University Hospital, Dow University of Health Sciences, Karachi, Pakistan
Received: 26 February 2020; Accepted: 13 March 2020; Published: 20 March 2020
Citation:
xh
Share at FacebookAbstract
Pseudo-pseudo Meig’s syndrome (PPMS) is an exceedingly uncommon disease in patients with diagnosed systemic lupus erythematosus (SLE) or mixed connective tissue disease (MCTD). This is a case report of a 23-year-old woman with a history of systemic lupus erythematosus (SLE), presented with rapidly increasing abdominal girth, facial/pedal edema, oliguria, and dyspnea, who was presumed to be developing pre-eclampsia in her 35th week of gestation. She was managed by an emergency Cesarean-section, and an incidental finding of peri-operative ascitic fluid was obtained and a right-sided pleural effusion. Upon further investigations, her CA-125 levels were found to be elevated. Her anti-double-stranded DNA titers were increased whereas complement levels (C3 and C4) were reduced indicating SLE flare. All these features suggested the diagnosis of pseudo-pseudo meig’s syndrome, a rare condition associated with SLE. It is asserted in our case that her pregnancy triggered the lupus flare.
Keywords
Lupus, Ovary, Meig’s, Ascites, Effusion, Flare
Lupus articles, Ovary articles, Meig?s articles, Ascites articles, Effusion articles, Flare articles
Lupus articles Lupus Research articles Lupus review articles Lupus PubMed articles Lupus PubMed Central articles Lupus 2023 articles Lupus 2024 articles Lupus Scopus articles Lupus impact factor journals Lupus Scopus journals Lupus PubMed journals Lupus medical journals Lupus free journals Lupus best journals Lupus top journals Lupus free medical journals Lupus famous journals Lupus Google Scholar indexed journals Ovary articles Ovary Research articles Ovary review articles Ovary PubMed articles Ovary PubMed Central articles Ovary 2023 articles Ovary 2024 articles Ovary Scopus articles Ovary impact factor journals Ovary Scopus journals Ovary PubMed journals Ovary medical journals Ovary free journals Ovary best journals Ovary top journals Ovary free medical journals Ovary famous journals Ovary Google Scholar indexed journals Meig’s articles Meig’s Research articles Meig’s review articles Meig’s PubMed articles Meig’s PubMed Central articles Meig’s 2023 articles Meig’s 2024 articles Meig’s Scopus articles Meig’s impact factor journals Meig’s Scopus journals Meig’s PubMed journals Meig’s medical journals Meig’s free journals Meig’s best journals Meig’s top journals Meig’s free medical journals Meig’s famous journals Meig’s Google Scholar indexed journals Ascites articles Ascites Research articles Ascites review articles Ascites PubMed articles Ascites PubMed Central articles Ascites 2023 articles Ascites 2024 articles Ascites Scopus articles Ascites impact factor journals Ascites Scopus journals Ascites PubMed journals Ascites medical journals Ascites free journals Ascites best journals Ascites top journals Ascites free medical journals Ascites famous journals Ascites Google Scholar indexed journals Effusion articles Effusion Research articles Effusion review articles Effusion PubMed articles Effusion PubMed Central articles Effusion 2023 articles Effusion 2024 articles Effusion Scopus articles Effusion impact factor journals Effusion Scopus journals Effusion PubMed journals Effusion medical journals Effusion free journals Effusion best journals Effusion top journals Effusion free medical journals Effusion famous journals Effusion Google Scholar indexed journals Flare articles Flare Research articles Flare review articles Flare PubMed articles Flare PubMed Central articles Flare 2023 articles Flare 2024 articles Flare Scopus articles Flare impact factor journals Flare Scopus journals Flare PubMed journals Flare medical journals Flare free journals Flare best journals Flare top journals Flare free medical journals Flare famous journals Flare Google Scholar indexed journals systemic lupus erythematosus articles systemic lupus erythematosus Research articles systemic lupus erythematosus review articles systemic lupus erythematosus PubMed articles systemic lupus erythematosus PubMed Central articles systemic lupus erythematosus 2023 articles systemic lupus erythematosus 2024 articles systemic lupus erythematosus Scopus articles systemic lupus erythematosus impact factor journals systemic lupus erythematosus Scopus journals systemic lupus erythematosus PubMed journals systemic lupus erythematosus medical journals systemic lupus erythematosus free journals systemic lupus erythematosus best journals systemic lupus erythematosus top journals systemic lupus erythematosus free medical journals systemic lupus erythematosus famous journals systemic lupus erythematosus Google Scholar indexed journals pre-eclampsia articles pre-eclampsia Research articles pre-eclampsia review articles pre-eclampsia PubMed articles pre-eclampsia PubMed Central articles pre-eclampsia 2023 articles pre-eclampsia 2024 articles pre-eclampsia Scopus articles pre-eclampsia impact factor journals pre-eclampsia Scopus journals pre-eclampsia PubMed journals pre-eclampsia medical journals pre-eclampsia free journals pre-eclampsia best journals pre-eclampsia top journals pre-eclampsia free medical journals pre-eclampsia famous journals pre-eclampsia Google Scholar indexed journals abdominal tumor, articles abdominal tumor, Research articles abdominal tumor, review articles abdominal tumor, PubMed articles abdominal tumor, PubMed Central articles abdominal tumor, 2023 articles abdominal tumor, 2024 articles abdominal tumor, Scopus articles abdominal tumor, impact factor journals abdominal tumor, Scopus journals abdominal tumor, PubMed journals abdominal tumor, medical journals abdominal tumor, free journals abdominal tumor, best journals abdominal tumor, top journals abdominal tumor, free medical journals abdominal tumor, famous journals abdominal tumor, Google Scholar indexed journals serositis articles serositis Research articles serositis review articles serositis PubMed articles serositis PubMed Central articles serositis 2023 articles serositis 2024 articles serositis Scopus articles serositis impact factor journals serositis Scopus journals serositis PubMed journals serositis medical journals serositis free journals serositis best journals serositis top journals serositis free medical journals serositis famous journals serositis Google Scholar indexed journals
Article Details
1. Introduction
The first discussion about meig’s syndrome was proposed by MEIGS and CASS in 1937 which included benign ovarian fibroma, transudative pleural effusion, and elevated level of CA 125 with the resolution on tumor removal [1]. Other variants of meig’s syndrome include Pseudo Meig’s syndrome (PMS) which can be distinguished by the presence of aforementioned features and a benign or malignant ovarian or abdominal tumor, Pseudo-pseudo meig’s syndrome (PPMS) which has no association with benign or malignant pelvic tumors and hence differentiated from the above. Pseudo-pseudo meig’s syndrome has an association with systemic lupus erythematosus (SLE) alone or occurs in SLE plus scleroderma [2, 3].
2. Case Presentation
A 23 year old female of Asian ethnicity, primigravida, weighing 60 kilogram, known case of systemic lupus erythematosus for 2 years, with a family history of tuberculosis and preeclampsia, presented to us at 35 weeks of gestation with complaints of rapidly increasing abdominal girth, facial puffiness, bilateral pitting pedal edema up to the shins, decrease urinary output with frothing, and dyspnea for 1 week. She denied any easy fatigability, abdominal pain, bleeding per vagina, yellow discoloration of eyes and skin, nausea/vomiting, fever, night sweats, or skin rash. Empirically the patient was started on oxygen inhalation, nebulization with salbutamol, and furosemide 40 mg intravenously. The emergency cesarean section was planned and a healthy male alive baby was delivered. Intraoperatively, ascitic fluid was found incidentally and 800 milliliters of ascitic fluid were drained and sent for further workup.
The physical examination was unremarkable except for blood pressure of 150/90 mmHg, mild pallor, bilateral periorbital puffiness, bilateral pitting pedal edema up to the shin, chest expansion was decreased on the right side, with stony dull percussion note, decreased breath sounds, and heart sounds were also muffled. The initial laboratory workup was hemoglobin 8.4 milligrams/deciliter, total leukocyte count (TLC) was 20.1 cells per ml of blood with neutrophil count of 60% and lymphocytes 20%, total protein was 3.99 gram per deciliter, while albumin was 1.37 gram per deciliter, and globulin was 2.62 gram per deciliter, total albumin to globulin ratio was 0.52, Erythrocyte sedimentation rate (ESR) was 98 mm/hour, prothrombin time (PT) was 29.3 seconds, international normalized ratio (INR) was 0.95. Urine analysis shows 3.5 grams per deciliter of protein along with the granular cast and red blood cells. A protein to creatinine ratio was turned out to be 1.3. A chest x-ray showed right-sided pleural effusion and enlarged heart shadow (Figure 1). A transthoracic echocardiogram showed pericardial effusion. Ultrasound abdomen and pelvis showed ascites and post-partum changes (Figure 2). The differential considerations were meig’s syndrome, pre-eclampsia, disseminated intravascular coagulation, HELLP syndrome, and abdominal tuberculosis.
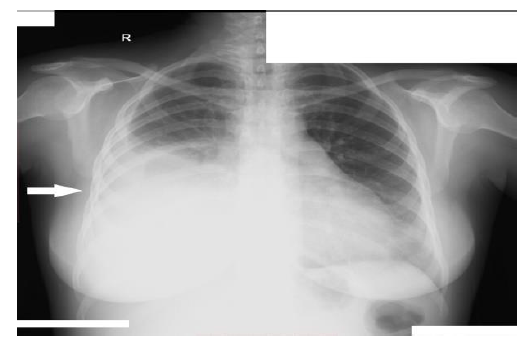
Figure 1: Chest X-ray (P-A view) showing right-sided pleural effusion.
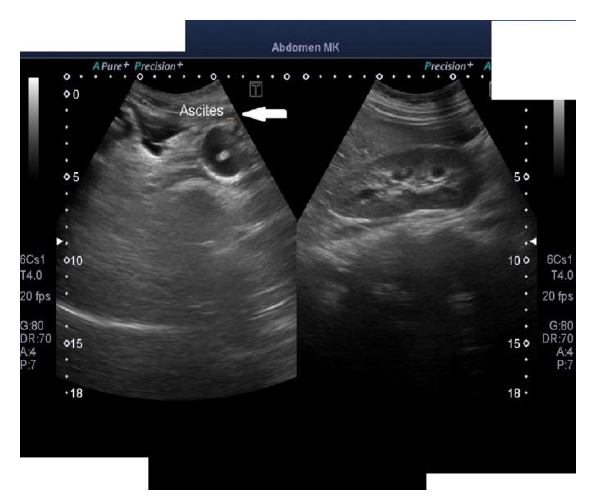
Figure 2: Ultrasound abdomen and pelvis showing gross ascites with post-partum changes and ruling out any mass or cystic lesion.
Ascitic fluid workup showed albumin of 3.4 gram per deciliter and Serum ascites albumin gradient ratio of 0.3. Ultrasound-guided tap for pleural effusion was done which showed pleural fluid albumin of 3.2 gram per deciliter and Lactate dehydrogenase of 2 units per liter. Immunologic work up index was anti-neutrophil cytoplasmic antibody (ANCA) 1:1000 (+1), Anti double-stranded DNA antibodies (Anti-DNA ds) titers were >1:80, while C3 levels were 30 milligram/deciliter and C4 levels were 6 milligram/deciliter. Ferritin levels were >1000 nanograms/milliliter. CA-125 levels were turned out to be 225 units/milliliter. The diagnosis of pseudo-pseudo meig’s syndrome was confirmed, and the patient was started on pulse therapy with injection methylprednisolone 1 gram intravenously on a daily basis for 5 days. After that the patient was switched to oral methylprednisolone 60 mg daily in two divided doses, oral leflunomide 20 mg once daily, and oral hydroxychloroquine 200 mg twice daily for 6 weeks. The patient was discharged after 1 week with regular follow-ups in the ambulatory clinics at the rheumatology and gynecology department for 6 months.
3. Discussion
Pseudo-pseudo Meig’s syndrome (PPMS) was first pioneered by TJALMA in 2005 by describing a case of a young female with concomitant history of SLE and classical characteristics of the syndrome. PPMS is an exceedingly uncommon disease in patients with diagnosed systemic lupus erythematosus or mixed connective tissue disease [3]. SLE is an autoimmune disorder that can potentially involve any organ or system. Serositis including pericardial, pleural or peritoneal membranes can lead to fluid accumulation, adherence, and even fibrosis. In a recent European study of 100 patients with SLE, the frequency of serositis was estimated to be 16% [4], with lupus peritonitis being the rarest.
The pathophysiology of PPMS is still not understood however it is suggested that an SLE phenotype which preferentially involves the serosa is responsible for the inflammation which may result in lympho-aggregation of plasma cells, immune complex deposition on the peritoneal membrane triggering a local inflammatory reaction or a vasculitic phenomenon involving the peritoneal vessels [5]. The presence of high ferritin levels (an acute phase reactant) also provides evidence to the underlying basis of this syndrome [6]. The pleural effusion can be explained by passive transfer of ascites through either trans-diaphragmatic route, lymphatic vessels or intracellular gaps, provided by the evidence that pleural effusion resolved when therapeutic paracentesis was done [1, 3]. Therapeutic control of SLE flare also settles the pleural effusion [1].
The elevation in the level of CA-125 in a patient with PPMS is a matter of concern. Considering the degree of inflammation in SLE, this can be explained as CA-125 is produced by mesovarium and omentum [7], so due to up-regulation of inflammatory mediators like interferon-gamma, Interleukin 1-b (IL-1b), Vascular endothelial growth factor (VEGF), and Fibroblast growth factor (FGF), it's synthesis is increased [8]. Few publications suggest that the diminishing of CA-125 levels after the resolution of serositis, explains a correlation between ascites and CA-125 elevation [9]. High levels of CA 125 can also occur in pelvic tuberculosis and nephrotic syndrome [10].
Our case describes a 23 year old woman with a past medical history of systemic lupus erythematosus, presented with rapidly increasing abdominal girth, facial/pedal edema, oliguria, and dyspnea, who was presumed to be developing pre-eclampsia in her 35th week of gestation based on hypertension and proteinuria, on a background of her family history. She was managed by an emergency Cesarean-section, and an incidental finding of peri-operative ascitic fluid was obtained (which was exudative), and a right-sided transudative pleural effusion. Upon further investigations, her CA-125 levels were found to be elevated. Her anti-double-stranded DNA titers were increased whereas complement levels (C3 and C4) were reduced indicating SLE flare. All these features suggested the diagnosis of pseudo-pseudo meig’s syndrome, a rare condition associated with SLE. It is asserted that her pregnancy triggered the lupus flare.
Treatment options of PPMS include treating the underlying SLE by steroids usually in the pulse dose followed by the maintenance therapy [11]. Anti-CD 20 monoclonal antibodies like rituximab also have a role in the treatment of refractory ascites based on its pathophysiology and involvement of pro-inflammatory cytokines [12].
Conflict of Interest
The authors declare no conflicts of interest with this article's content.
Financial Disclosing Statement
This work is not supported by any sponsors. No funding required in this study.
Ethical approval statement/Declaration of patient’s consent
Ethical approval was not required in this study from the institutional review board, and consent to participate has been taken from the patient’s guardian with informed written consent.
References
- Meigs JV, Cass JW. Fibroma of the ovary with ascites and hydrothorax: with a report of seven cases. Am J ObstetGynecol 33 (1937): 249-267.
- Schmitt R, Weichert W, Schneider W, et al. Pseudo-pseudo Meigs' syndrome. Lancet 366 (2005): 1672.
- McVorran S, Song J, Pochineni V, et al. Systemic Lupus Erythematosus Presenting with Massive Ascites: A Case of Pseudo-Pseudo Meigs Syndrome. Case Rep Rheumatol (2016).
- Man BL, Mok CC. Serositis related to systemic lupus erythematosus: prevalence and outcome. Lupus 14 (2005): 822-826.
- Pott Júnior H, Amate Neto A, Teixeira MAB, et al. Ascites due to lupus peritonitis: a rare form of onset of systemic lupus erythematosus. Revista Brasileira de Reumatologia 52 (2012): 116-119.
- Lim MK, Lee CK, Ju YS, et al. Serum ferritin as a serologic marker of activity in systemic lupus erythematosus. Rheumatology International 20 (2001): 89-93.
- Zeimet AG, Marth C, Offner FA, et al. Human peritoneal mesothelial cells are more potent than ovarian cancer cells in producing tumor marker CA-125. Gynecologic Oncology 62 (1996): 384-389.
- Zeimet AG, Offner FA, Marth C, et al. Modulation of CA-125 release by inflammatory cytokines in human peritoneal mesothelial and ovarian cancer cells. Anticancer Res 17 (1997): 3129-3131.
- Yang Z, Liang Y, Li C, et al. Serum CA125 elevation is independently associated with serositis in SLE patients. Clin Exp Rheumatol 30 (2012): 93-98.
- Dalvi SR, Yildirim R, Santoriello D, et al. Pseudo-pseudo Meigs' syndrome in a patient with systemic lupus erythematosus. Lupus 21 (2012): 1463-1466.
- Bes C, Soy M. Pseudo-pseudo Meigs syndrome developed under the leflunomide therapy. Rheumatology International 31 (2009): 521-523.
- Iaccarino L, Bartoloni E, Carli L, et al. Efficacy and safety of off-label use of rituximab in refractory lupus: Data from the Italian Multicentre Registry. Clinical and Experimental Rheumatology 33 (2015): 449-456.
