Promoting Effect of Adriamycin, Cisplatinum and Etoposide on Trk Genes Expression in Human Neuroblastoma Cells
Article Information
Ling Liu1, Yang Li2,3*, Xilin Xiong2, 3, Chi Zhang2,3, Jianpei Fang2,3
1Department of Pediatric, Affiliated Hospital of Guangdong Medical University, Zhan Jiang, Guangdong 524000, P.R. China.
2Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Sun Yat-Sen University, Guangzhou, Guangdong 510120, P.R. China.
3Department of Pediatric Hematology/Oncology, Sun Yat-Sen Memorial Hospital, Sun Yat-Sen University, Guangzhou, Guangdong 510120, P.R. China.
*Corresponding Authors: Li Yang, Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation and Department of Pediatric Hematology/Oncology, Sun Yat-Sen Memorial Hospital, Sun Yat-Sen University, Guangzhou, Guangdong 510120, P.R. China
Received: 25 April 2019; Accepted: 14 May 2019; Published: 17 May 2019
Share at FacebookAbstract
Purpose: Tropomyosin receptor kinases (Trk) gene family had been strongly correlated with tumor progression and found to be highly predictive of clinical behavior. The aim of the study is to evaluated the cytotoxicity and cell cycle effect of adriamycin (ADM), cisplatinum (DDP) and etoposide (VP16) on neuroblastoma(NB) cells, and then assessed effect of three anti-cancer agents on Trk gene family expression in NB in vitro conditions.
Methods: SK-N-SH NB cells were exposed in vitro to ADM, DDP and VP16. Cell counting kit (CCK-8) assay was performed to determine the effect of anti-cancer agents on cell viability, and the cell cycle change was measured by flow cytometry. Quantitative reverse transcription polymerase chain reaction (RT-PCR) analysis was utilized to detect the expression levels of Trk gene family (TrkA, TrkB and TrkC) in a wide range of concentration of ADM, DDP and VP16 at different time.
Results: Our data illustrated that ADM, DDP and VP16 had cytotoxic activity as a single agent in both a time- and dose-dependent manner in vitro. However, NB cells resistance to high concentrations of ADM. It was confirmed that NB cells showed in vitro sensitivity to ADM, DDP and Vp16, with concentration that observed with the half-maximal inhibitory concentration (IC50) values on SK-N-SH cells being 5μg/ml, 8μg/ml and 100μg/ml, respectively. Flow cytometry analysis showed that these three drugs also leaded to enhanced accumulation of cell populations in S phase. Additionally, our results showed that in NB cells, these three drugs treatment dramatically increased expression of TrkA, TrkB and TrkC in RT-PCR analysis.
Conclusions: Collectively, our results may help to develop tailored treatment approaches and enhance the efficacy of cytotoxic agents treatment for high-risk NB with minimal or no additional toxicity by adjusting the concentration and
Keywords
Neuroblastoma; Trk gene family; adriamycin; cisplatinum; etoposide
Neuroblastoma articles, Trk gene family articles, adriamycin ar4ticles, cisplatinum articles, etoposide articles
Article Details
1. Introduction
Neuroblastoma(NB), as one of the most common and deadly extracranial solid neoplasm of childhood, accounts for nearly 8%~10% of all malignancies in pediatric patients [1]. Moreover, it is derived from pluri-potent neural crest precursor cells and behave in a significantly aggressive and unpredictable behavior associated with responsible for approximately 15% of all childhood cancer-linked mortalities [1]. Unfortunately, more than 50% of the treated high-risk NB patients ultimately die of recurrence and multifocal metastatic disease despite of responding well to intensive multimodality therapy at early stage [2, 3]. Indeed, patients with refractory or recurrent NB have poor response to salvage treatment strategies and behave locally aggressive with long-term survival under 40% [4]. During the last few years, advances in the treatment including stem cell transplants, target therapy and biological immunotherapy have been effectively applied to relapsed or refractory NB, but overall survival rates for these patients has been improved only modestly [5, 6].
Adriamycin (ADM) plays its anti-tumor activity related to inhibiting of nucleic acid synthesis [7]; cisplatinum (DDP) function therapeutically primarily by preventing DNA replication in rapidly formation of inter and intrastrand cross-links between platinum and guanine; etoposide (VP16), which can act on DNA topoisomerase II, and then form drugs-enzyme-DNA stable reversible complexes, eventually lead to DNA damage [8]. At present, these three chemotherapeutic agents are emerging as a feasible first-line choice in the treatment of NB. More importantly, the effectiveness of first-line chemotherapy is often obviously limited by development of chemoresistance and toxic effects [9]. Accordingly, better increasing the integrated effectiveness of initial chemotherapy with minimal toxicity by boosting the effectiveness of several cytotoxic agents may achieve promising strategy against relapsed or refractory NB.
In recent years, with rapid development of molecular medicine and genetics approaches, much more progress has been made in predicting the prognosis related to clinical behaviors of NB. Special emphasis has been placed on significant tumor-associated genes which are equally important as oncogenes in NB risk stratification and therapeutic approaches [10].
NB was characterized by genomic and biologic heterogeneity, and the tropomyosin receptor kinases (Trk) family as well-established molecules contributed to this heterogeneous behavior and clinical diversity [11, 12]. Moreover, the Trk family has well-defined roles in neuronal development [4] and elicits diverse cellular responses such as inducing differentiation, providing trophic support, modulating neuronal function and promoting axon outgrowth or apoptosis in neuronal development [13]. To date, the effect of ADM, DDP and VP16 on Trk gene family in NB has yet to be investigated.
The Trk gene family encompass the three highly homologous receptor isoforms, TrkA, TrkB and TrkC, which are activated upon preferential and high-affinity binding with the neurotrophin family of ligands, including nerve growth factor(NGF), brain-derived neurotrophic factor (BDNF), and neurotrophin-3 (NT-3) [4, 14]. At the cell membrane, NGF activates TrkA, BDNF interacts with TrkB, and NT-3 preferentially binds TrkC [14]. Accordingly, the TrkA ligands NGF indeed initiates differentiation and increases apoptosis in vitro of many human primary NB cells [13, 15, 16]. Differential expression of the Trk family of neurotrophin receptors is considered to be correlates with clinical behavior. TrkA overexpression is strongly predictive of favorable clinical and biological features and prone to spontaneous regression or differentiation, with an excellent outcome [17, 18]. In contrast, cumulative evidence indicates that high levels of TrkB is responsible for tumor progression such as invasion, metastasis, angiogenesis, and associated with frequently MYCN amplification with dismal prognosis [13, 19, 20]. Furthermore, TrkB overexpression through activation of the phosphatidyl-inositol-3-kinase (PI3K) /AKT survival pathway contributed to increasing chemoresistance in biologically aggressive, high-risk NB tumor cells and that by specifically targeting the critical downstream mediators of activated TrkB we could improve NB cell chemosensitivity [19, 21-23]. Moreover, TrkC which is expressed in a subset of the TrkA-expressing NB also serve as a good prognostic indicator in NB [13, 21]. Most NB expresses at least one member of the Trk family [21]. Favorable prognosis NB such as stage1, stage2 and stage4s usually express relatively high levels of TrkA, whereas unfavorable, MYCN-amplified NB such as stage3 and stage4s usually are associated with expression of TrkB [24]. This conclusion is borne out by our preliminary study [25].
In the present paper, we utilized a human NB cell line, SK-N-SH cell line treated by different concentrations and action time of ADM, DDP and VP16, to analyze the variance and significance of expression of TrkA, TrkB and TrkC. In the following study, we hope to investigate the chemotherapeutic regulation on the expression of TrkA, TrkB and TrkC, thereby achieving the rationale for modulating risk-based chemotherapy intensity and translating individual and moderate chemotherapeutic scheme into the clinic.
2. Methods and Material
2.1 Cell line
Human SK-N-SH cell line was purchased from the Sun Yat-sen University. The cells were cultured in DMEM/F12 (1:1 mixture) (Hyclone, USA) supplemented with 10% fetal bovine serum (FBS, Hyclone). Cells were grown in a humidified incubator of 5% CO2/95%.
2.2 Reagents
ADM, VP16 and DDP were purchased from Sigma. TRIzol was purchased from Invitrogen, and M-MLV reverse transcriptase was purchased from Promega. PrimeScriptTM RT reagent Kit (Perfect Real Time), Taq DNA polymerase and ExTaq DNA polymerase were purchased from Sigma Chemical. High-speed refrigerated centrifuge (J-25 model) was from Promega, and normal PCR instrument (PTC-100TM model) is from Bio-rad. Fluorescence quantitative PCR (ABI 7900 models) is from Stepone, and Micro UV spectrophotometer (NanoDrop ND-2000C) is from Promega. Flow cytometry (FCM-2012) is from BD.
2.3 Measurement of cell survival rate (CCK-8 assay)
For experiments, SK-N-SH cells were plated into 96-well plates at a concentration of approximately 104 cells per well in triplicate wells at 37 °C in a humidified incubator of 5% and allowed to adhere overnight. The next day, for determination of cell growth inhibition, cells were incubated with or without defined concentrations of ADM, DDP or VP16 in 37 ? CO2 incubator for 24 hours and 48 hours followed by the CCK-8 assay.
After treatment, 10µl of CCK-8 reagents were added to each well and incubated at 37 ? for 1-4 hours. The absorbance of the solutions was read at 450 nm by a microplate reader (NanoDrop ND-2000C, promega, USA) according to standard protocols. Percentage of cell viability rate was calculated as the percentage of CCK-8 absorption as follow: (absorbance of experimental wells/absorbance of control wells)×100%. All treatments were performed in triplicates and repeated three times. The concentration inhibiting 50% of cell proliferation (IC50) value was resulting from 50% inhibition of cell growth.
2.4 Flow cytometry for cell cycle
For experiments, cells were plated in 6-well plates at a density of approximately 3×105 cells per milliliter for 24 hours. According to the results of CCK-8, IC50 of ADM, VP16 and DDP were added to the experimental group. After this, untreated or drugs-treated cells were centrifuged, washed with PBS and fixed with ice-cold 75% ethanol overnight. Then, the fixed cells were incubated with 200 µl of binding-buffer, 10 µl of Annexin-V and 5 µl of propidium iodide incubated for 15 minutes in the dark. In the end, 200 µl of binding-buffer was added to the fixed cells. The cell cycle phase was carried out by flow cytometry in just one hour.
2.5 Total RNA isolation
Following chemotherapy treatment for a different time, total RNA was isolated from SK-N-SH cell lines using the TRIzol reagent (Bio. Basic, Shanghai, China) according to the manufacturer’s protocol. The RNA was precipitated with isopropanol, washed with 75% ethanol, and dissolved in diethylpyrocarbonate (DEPC)-treated water. The integrity and purity of total RNA were assessed by DNA gel (1% agarose gel) electrophoresis and UV detection.
2.6 Quantitative reverse transcription polymerase chain reaction (RT-PCR)
The PCR primers were designed with Primerpremier 5.0 software. The primers for TrkA were as follows: TrkA-F 5’-TCCAACACGGAGGCAATC-3’; TrkA-R 5’-CCAGGACATCCAGGTAGACAG-3’. The primers of TrkB were as follows: TrkB-F 5’-TCCGAGATTGGAGCCTAACAG-3’; TrkB-R 5’-CCACCAGGATCAGTTCAGACA-3’ The primers for TrkA were as follows: TrkC-F 5’-CAAGCCCACCCACTACAACA-3’; TrkC-R 5’-GTGGGACTCACTTCGTCAAAC-3’. Following cDNA synthesis, PCR amplification was performed in order to examine the number of Trk transcripts. The assay was carried out using a Biometra T-gradient Thermoblock thermal cycler (Gottingen, Germany). Multiplex RT-PCR was conducted in a final volume of 50 μL with 2× PCR-mix containing 200 μM dNTPs, 5% dimethyl sulfoxide (DMSO), 0.02 U HotStart Taq DNA polymerase (Qiagen GmbH, Hilden, Germany), and 50 pmol of each primer pair. The initial PCR cycle included activation of the polymerase at 95 °C for 5 min, which was followed by 40 cycles of PCR amplification (denaturation at 95 °C for 1 min, annealing at 55 °C for 1 min, and elongation at 72 °C for 1 min) and 1 cycle of elongation at 72 °C for 7 min prior to cooling at 4 °C. The products from the multiplex RT-PCR were confirmed by specific Trks using individual PCRs, which were performed separately using the specific primers for each Trk. The conditions for the individual PCRs were identical to those of the multiplex reactions. All experiments were performed in triplicate unless otherwise noted.
2.7 Statistical analysis
Computations were performed using the SPSS17 software. The collected data were analyzed by a one-way ANOVA (independent) and nonparametric-test. All statistical tests were 2-sided, and a P value of less than 0.05(P<0.05) selected as a statistically significant difference.
3. Results
3.1 ADM, DDP and VP16 inhibited the proliferation of SK-N-SH cells
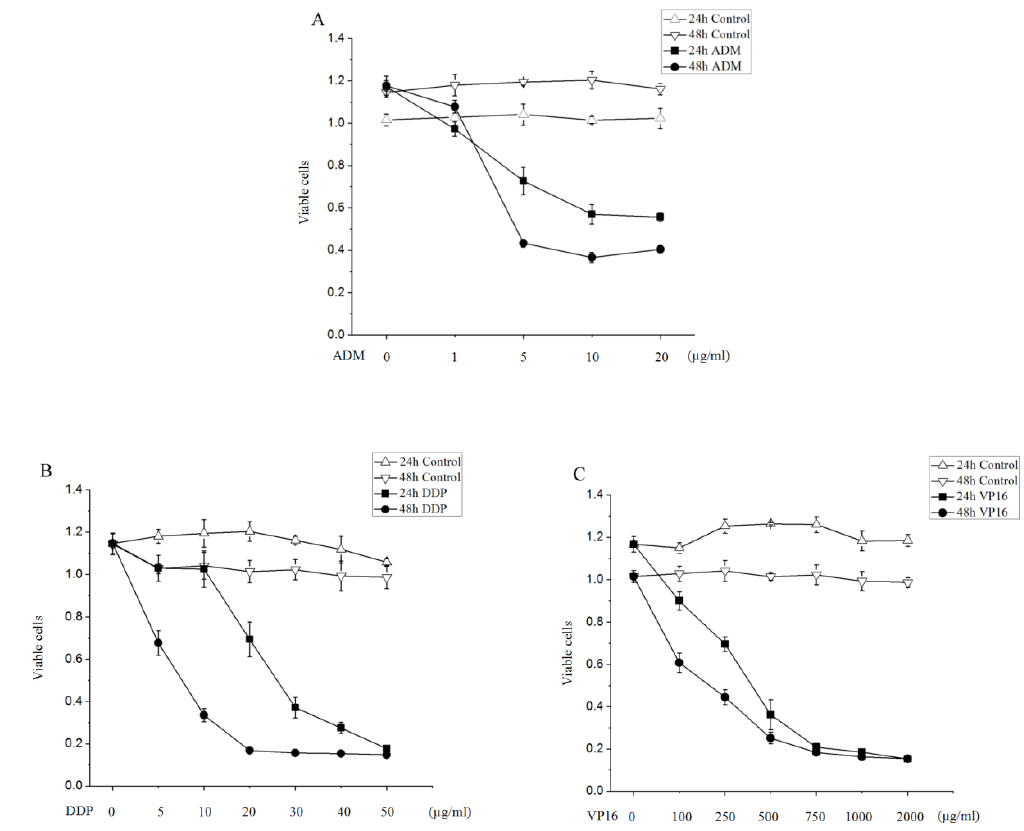
In order to determine the effects of ADM, DDP and VP16 on SK-N-SH cells, we performed CCK-8 analysis the viability and the proliferation of SK-N-SH cells. We examined the survival of SK-N-SH cell line in response to increasing concentrations of ADM, VP16 and DDP during an incubation period of 24h and 48h, cell viability was measured, and IC50 values were calculated. At first, according to the literature review [26, 27]and the results of our previous study [28], we determined the treatment conditions (drug concentration and treatment duration). The growth inhibitory effects of ADM, VP16 and DDP was determined as described under Materials and Methods. The viability of the cells was measured at different drug action time. Each point represents the mean±SD of three independent experiments.
Our results revealed that treatment of SK-N-SH cells with ADM results in a dose- and time-responsive cell death as measured by CCK-8, the effect is significant when ADM was <5 µg/ml at very low concentrations. However, the apoptosis rate of NB cells did not significantly increase, when the concentration of ADM was more than 5 µg/ml (Figure 1A). As shown in Figure 1A, there were only a little of apoptosis cells in the SK-N-SH cell line without the ADM treatment, but the viable cells were gradually decreased and reached 72.7±1.4% with a concentration of 5 µg/ml ADM at 24 hours of exposure. Moreover, only 43.3±1.9% of cells were viable at 5 µg/ml ADM of exposure, 36.6±2.4% of cells were viable by 10 µg/ml ADM, and 40.5±1.8% were viable by 20 µg/ml ADM after 48 hours of exposure.
Figure 1: Cytotoxic effect of ADM, VP16 and DDP in SK-N-SH cells. (A) The growth curves of ADM-treated SK-N-SH cells after various time induction at given concentrations. The apoptosis rate of SK-N-SH cells significantly increased upon ADM treatment when the concentration was <5 µg/ml. (B and C) Cells were exposed to the indicated concentrations of DDP and VP16 for 24 hours and 48 hours which exhibit highly exhibited dose- and time-dependent inhibitory effects on SK-N-SH cells. All results are presented as the mean±SD performed from at least 3 independent experiments.
A marked augmentation in apoptosis was observed with DDP and VP16 treatment which highly exhibited dose- and time-dependent inhibitory effects on SK-N-SH cells (Figure 1B and Figure 1C). As shown in Figure 1B, after 5 µg/ml, 10 µg/ml, 20 µg/ml, 30 µg/ml, 40 µg/ml and 50 µg/ml DDP treating SK-N-SH cells for 24 hours, the cell survival rates were (100.3±6.1)%, (100.2±8.6)%, (69.4±8.2)%, (37.1±4.9)%, (27.5±2.6)% and (17.7±4)%; after different concentrations of DDP treating SK-N-SH cells for 48 hours, the cell survival rates were (67.8±5.8)%, (33.5±3.1)%, (16.8±0.9)%, (15.6±0.3)%, (15.3±0.7)% and (14.7±0.7)%, DDP cytotoxity to SK-N-SH cells in a concentration-dependent manner. The apoptosis rates significantly increased after the same concentration of DDP treating SK-N-SH cells for 48 hours relative to 24 hours, DDP cytotoxity to SK-N-SH cells in a time-dependent manner. After 100µg/ml, 250µg/ml, 500µg/ml, 750µg/ml, 1mg/ml and 2mg/ml, VP16(Figure 1C) treating SK-N-SH cells for 24 hours, the cell survival rates were (90.9±4.2)%, (69.6±3.5)%, (36.2±7.1)%, (20.9±1.9)%, (18.5±0.6)% and (15.2±0.6)%. After 100 µg/ml, 250 µg/ml, 500 µg/ml, 750 µg/ml, 1 mg/ml and 2 mg/ml VP16 treating SK-N-SH cells for 48 hours, the cell survival rates were (60.8±4.6)%, (44.5±3.8)% , (25.2±2.8)%, (18.3±1.07)%, (16.3±0.6)% and (15.4±0.5)%, VP16 cytotoxity to SK-N-SH cells in a concentration-dependent manner. The apoptosis rates significantly increased after the same concentration of VP16 treating SK-N-SH cells for 48 hours relative to 24 hours, VP16 cytotoxity to SK-N-SH cells in a time-dependent manner.
Results show that cells have significantly different IC50 values to three drugs, the IC50 of these agents required to induce the apoptosis of SK-N-SH cells was 5 µg/ml of ADM, 8 µg/ml of DDP, and 100 µg/ml of VP16 at 48 hours treatment. Taken together, our results indicate that SK-N-SH cells were responsive to ADM, DDP and VP16-induced apoptosis in a dose- and time-dependent manner, however, NB cells resistance to high concentrations of ADM.
3.2 Effects of ADM, DDP and VP16 on cell cycle arrest of SK-N-SH cells
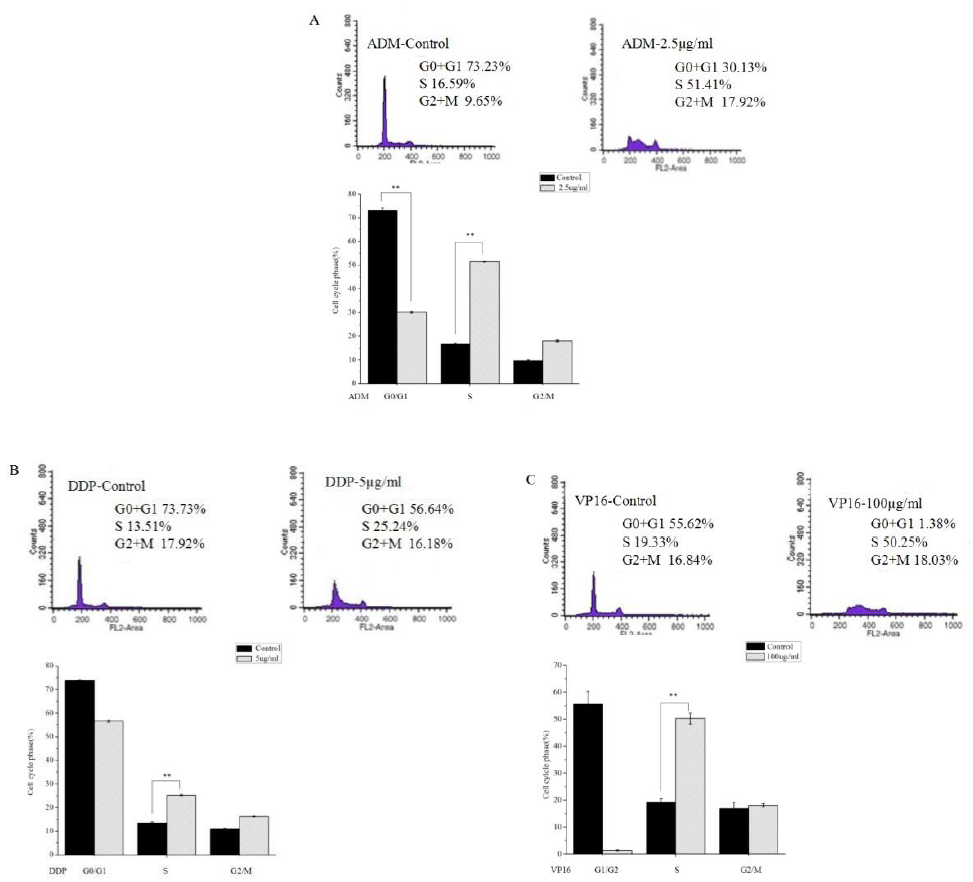
Here, to further analyzed ADM, DDP or VP16 impact on cell cycle progression of SK-N-SH cells. Analysis of cell cycle phase distribution was carried out to study the anti-proliferative mechanism of chemotherapeutic agents on SK-N-SH cells. According to the literature and our result of IC50 of the three drugs on SK-N-SH cells, we chosen 2.5 µg/ml ADM, 5 µg/ml DDP and 100 µg/ml VP16 were added to the experimental group.
It can be seen that treatment of SK-N-SH cells with ADM (Figure 2A), DDP (Figure 2B) and VP16 (Figure 2C) caused cell cycle arrest at S phase, and this was accompanied by a dramatic decrease in the percentage of cells at the G0/G1 phase. Compared with control, ADM treatment resulted in a significant accumulation of cells in the S phase, the percentage of SK-N-SH cells in the S phase increased from 16.59±0.52% to 51.41±0.13%. At time 0, only 13.51±0.39% of cells were in the S phase, and after 24 hours of DDP treatment, 25.24±0.37% of cells were in the S phase. Incubation of VP16 for 24 hours, the percentage of S phase increased to 50.25±2.06%.
Figure 2: Cell cycle arrest of the SK-N-SH cells by flow cytometry. One representative image of three is shown. After 24 hours exposure to ADM, DDP and VP16, estimation of cell cycle distribution was determined by flow cytometry. Accumulation of the cells at S phase was evident after exposure to ADM(A), DDP(B) and VP16(C). All data are expressed as mean±SD from three independent experiments. **P<0.05 a significant difference between the two groups.
t 3 independent experiments.3.3 Effects of ADM, DDP and VP16 on the expression changes of Trk gene family in SK-N-SH cells
To our knowledge, the Trk gene family in NB is highly linked with biological feature and prognosis and is responsible for multidrug resistance (MDR) formation. Given these observations, we confirmed whether ADM, DDP and VP16 are able to modulate the expression of Trk induced by different concentration gradients of ADM, DDP and VP16 with variable duration in SK-N-SH cell line using RT-PCR. Each group sets three parallel holes.
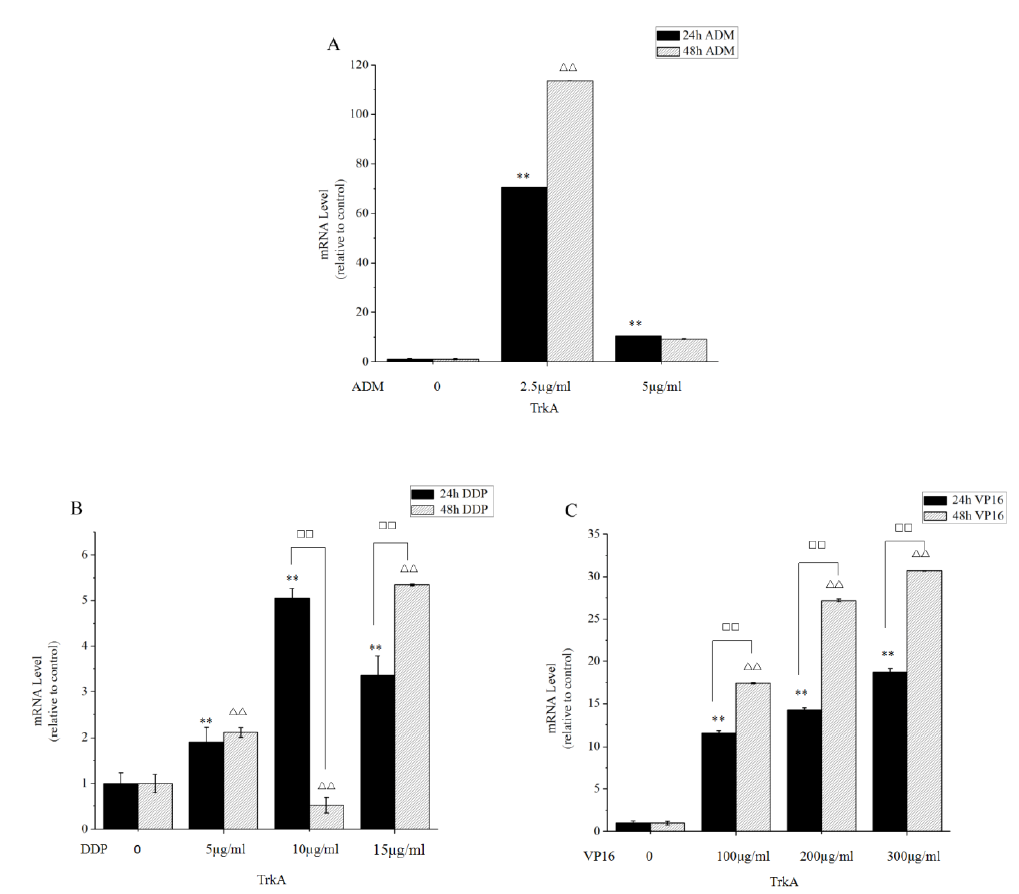
There was a statistically significant increase (at least 2-fold) in TrkA gene after treatment with ADM, DDP and VP16 in SK-N-SH cell line. To determine the extent of the observed striking increase in TrkA expression, we performed RT-PCR on TrkA expression using the column chart (Figure 3). RT-PCR demonstrated variable levels of TrkA, with the highest levels identified in SK-N-SH cells, which had approximately 100-fold higher expression levels than control group (Figure 3). RT-PCR analysis showed that ADM transiently stimulated TrkA expression, causing a highest (113-fold) increase of TrkA level after 48 hours of 2.5 µg/ml ADM, and there were no difference between 24 hours and 48 hours in the expression level of TrkA (Figure 3A). Figure 3B illustrated that 5 µg/ml, 10 µg/ml and 15 µg/ml DDP treatment for 24 hours and 48 hours both can lead to the expression of TrkA increasing (Figure 3B), with the dosage of 10 µg/ml at 24 hours being the most obvious. As shown in Figure 3C, RT-PCR revealed that VP16 treatment resulted in a time- and dose-dependent up-regulation of TrkA (P<0.05). In summary, the data suggested that we should appropriately increase the concentration and prolonging the time of DDP and Vp16 to apparently improve drug efficacy in high-expression TrkA NB. When it comes to ADM, appropriately reducing the dose was feasible, but prolonged action time not recommended.
Figure 3: ADM, DDP and VP16 strongly up-regulated TrkA expression in SK-N-SH cell lines. (A) SK-N-SH cells were treated with 2.5 µg/ml and 5 µg/ml ADM for 24 hours and 48 hours and subjected to RT-PCR assays. ADM has increased TrkA expression relative to the controls. Cells expressing TrkA show no alterations between 24 hours and 48 hours in the expression level of TrkA. RT-PCR analysis showed that ADM causing a highest (113-fold) increase of TrkA level after 48 hours of 2.5 µg/ml ADM. (B and C) The histogram showed that DDP treatment for 24 hours and 48 hours leads to increased expression of TrkA, with the dosage of 10 µg/ml at 24 hours most obvious. RT-PCR revealed that VP16 treatment resulted in a time- and dose-dependent up-regulation of TrkA. Bars represent the mean±SD of three independent experiments. **P<0.05, compared with 24 hours control. ??P<0.05, compared with 48 hours control.??P<0.05, compared with the two groups.
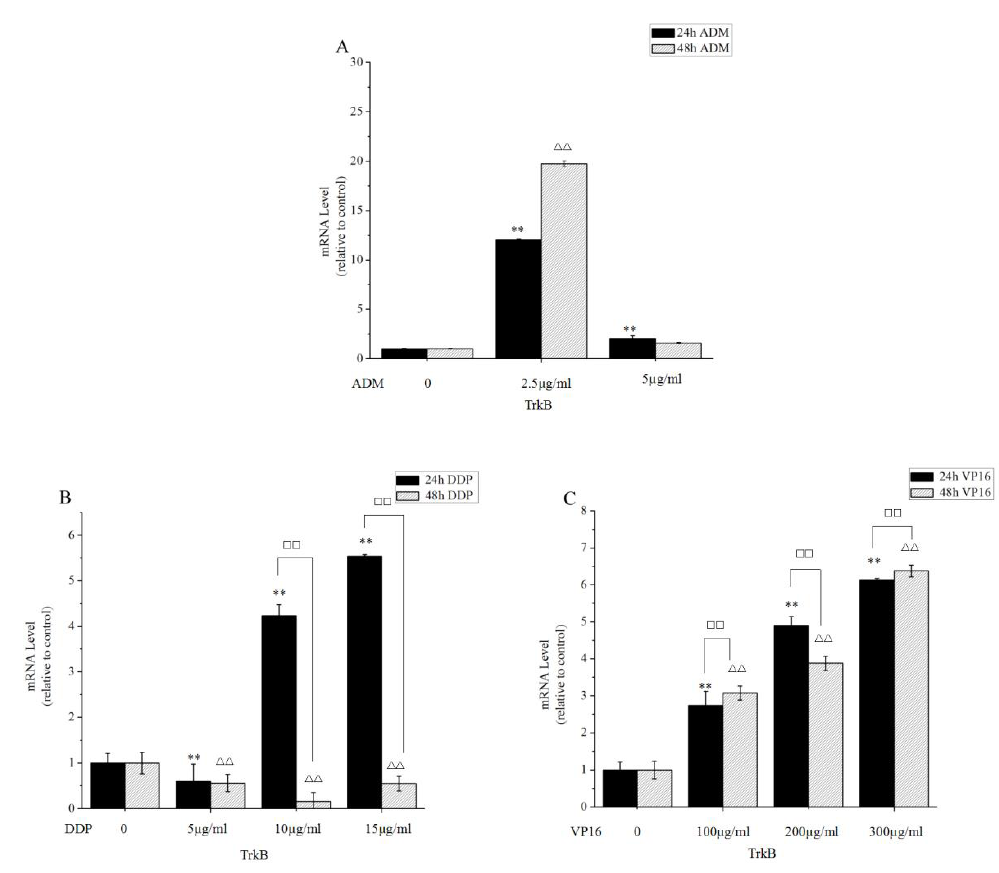
A comparison of TrkB expression in SK-N-SH cells with or without ADM, DDP and VP16 was also performed. The TrkB level was obviously higher in SK-N-SH cells in the presence of ADM, DDP and VP16 compared to the control. At 2.5 µg/ml ADM for 24 hours and 48 hours, the expression of TrkB was noted to gradually increased, but there was no significant variation in TrkB levels after ADM exposure at 24 hours and 48 hours (Figure 4A). Taken together, our data revealed that after VP16 treatment, we observed a substantially increasing in the induced TrkB expression in dose- and time-independent manner (Figure 4B). Further, we found a significant increase in TrkB expression after DDP treatment at 24 hours in dose-independent manner (Figure 4C). Also, a significant decrease in TrkB expression was noted in different concentration DDP at 48 hours. These results might improve the chemotherapy strategy that appropriately reducing the dose of DDP and VP16 and combining with other drugs should be taken into account in stage3 and stage4s of high expression of TrkB strongly associated with poor prognosis.
Figure 4: TrkB expression is significantly increased by incubating the cells in the presence of ADM, DDP and VP16. Expression levels of TrkB were detected in SK-N-SH cell lines by RT-PCR. (A) RT-PCR analysis showed that there were no difference between 24 hours and 48 hours in the expression level of TrkA after 2.5 µg/ml and 5 µg/ml ADM. (B) The histogram showed that TrkB expression was completely suppressed in different concentration DDP at 48 hours. (C) SK-N-SH cells were treated with various concentrations of VP16 for 24 hours and 48 hours. TrkB appeared overexpressed in SK-N-SH cells in comparison to controls. **P<0.05, compared with 24 hours control. ??P<0.05, compared with 48 hours control.??P<0.05, compared with the two groups. Data are expressed as the mean±SD of three independent experiments.
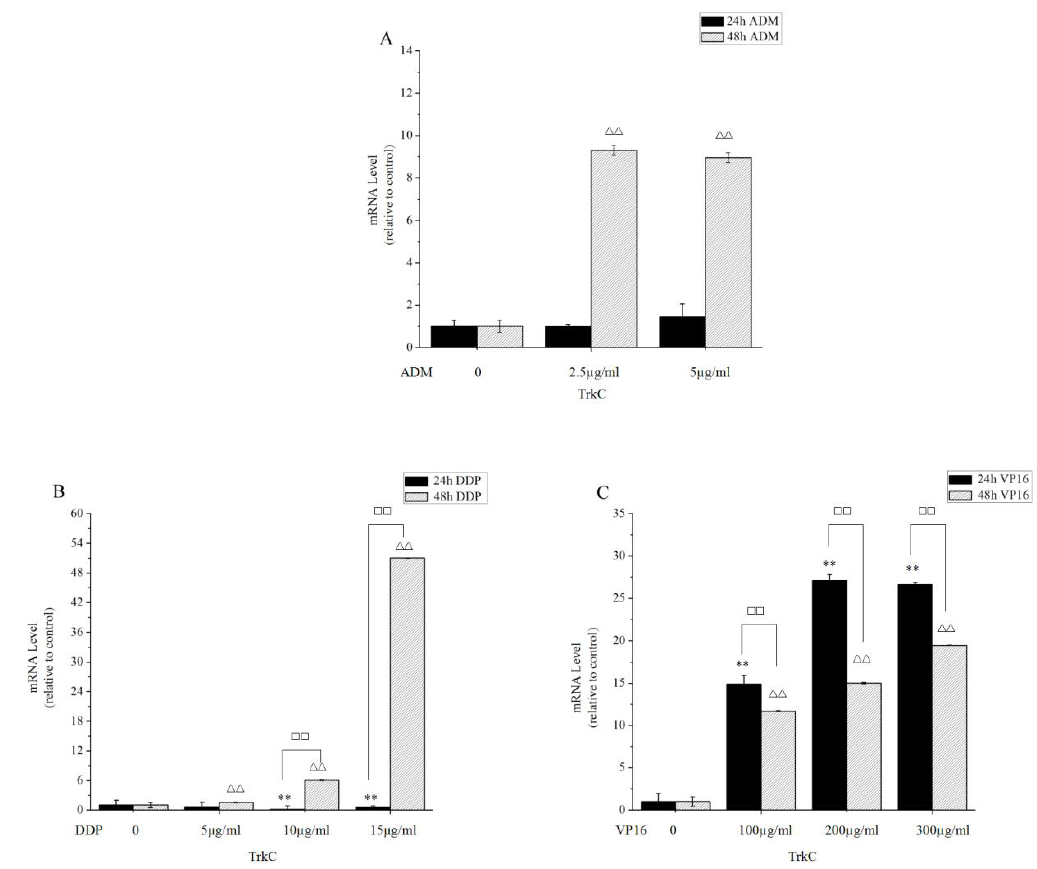
For TrkC, our results showed that DDP and Vp16 significantly increased the level of TrkC expression (Figure 5B and Figure 5C). RT-PCR further identified that the expression of TrkC in SK-N-SH cells was significantly up-regulated when treating with different concentrations of ADM at 48 hours (Figure 5A). However, this difference was not statistically significant in different concentrations of ADM for 24 hours in comparison to the control. It is noteworthy that the highest levels of TrkC were observed in 48 hours of 15 µg/ml DDP. When it comes to DDP and VP16, appropriately increasing the concentration apparently improved prognosis in high-expression TrkC NB. When it comes to ADM, appropriately reducing the dose was feasible, and that further decreased the drug related toxicity.
Figure 5: The ADM, DDP and VP16 inducible expression of TrkC determined by RT-PCR. (A) SK-N-SH cells were treated with various doses of ADM for the indicated times. Expression levels of TrkC in SK-N-SH cells were significantly up-regulated when treating with 48 hours of ADM. (B and C) SK-N-SH cells were treated with various concentrations of DDP and VP16 for 24 hours and 48 hours. DDP and VP16 significantly increased the level of TrkC expression in SK-N-SH cells. Furthermore, expression of TrkC was found at its highest level after 48 hours of 15 µg/ml DDP. **P<0.05, compared with 24 hours control. ??P<0.05, compared with 48 hours control.??P<0.05, compared with the two groups. Data are expressed as the mean±SD of three independent experiments.
Altogether, our studies concluded that commonly used chemotherapeutic agents for NB might affect Trk gene family expression, such findings prompted us to seek ways to increase the therapeutic effect of cytotoxic drugs. On the basis of these results, in the treatment of stage1, stage2 and stage 4 with high expression of TrkA or TrkC that contributes to good prognosis NB, the concentration of the ADM should be given reduced, but increasing the concentration should be recommended when using DDP and VP16 and prolong the action time of DDP; however, in the therapy of stage3 and stage4s with poor prognosis and high expression of TrkB, appropriately reducing the concentration and combining with other drugs should be recommended when using DDP and VP16.
4. Discussion
NB is the most common extracranial soild neoplasm of high malignancy with metastases and dissemination occuring in about 50% of patients for early diagnosis [29]. Recently, treatments of NB mainly involve invasive surgery, chemotherapy, targeted systemic radiotherapy, autologous stem cell transplantation (ASCT), radiotherapy, immunotherapy and gene therapy, while chemotherapy still continues to play the pivotal role in cancer therapy [6, 30-33]. The occurrence, growth and prognosis of NB depends on a very complicate polygene-related process in which involves in regulation of multi-factors, multi-stages and multi-genes, including oncogene activation and anti-oncogene inactivation. So, further study of the alterations of related regulatory genes undergoing chemotherapy is necessary, and such changes will not only promote further investigations of molecular mechanisms of NB, but will also contribute to make better characterise NB tumour biology and present new risk-stratified treatment strategies [34].
Previously, we found that ADM, DDP and VP16 apparently inhibited SK-N-SH cells growth in a dose-dependent manner, but the inhibitory effect did not obviously increase after ADM treatment at the higher concentration. The possible mechanism for this switch is related to increased expression of MRP1 (multidrug resistance-associated protein1) genes [35]. We also found that with the action time of ADM, DDP and VP16 extending, the apoptosis rate of SK-N-SH cells increased. Our experiment results suggested that by adjusting the drug concentration and action time to achieve the best therapeutic effect should rely on the various chemotherapy agent characters. Flow cytometric analysis suggested that ADM, DDP and VP16 triggered cell cycle arrest at S phase in SK-N-SH cells. The cell cycle is the series of events, when DNA synthesis inhibited cells cycle arrest in S phase [36]. The S phase arrest observed in the cells treated by ADM, DDP and VP16 suggest that these the three drugs inhibit DNA synthesis of the SK-N-SH cells [37]. Recently, a number of cell cycle proteins have been shown to exert oncogenic roles in NB, and cyclin which further invokes signal transduction pathway is key cell cycle proteins in the regulation of the cell cycle [38]. The S arrest point is associated with accumulation of cyclin A protein, which production initially increases in cells during late G1 phase [36]. These results might suggest that ADM, DDP and VP16 produces obvious antineuroblastoma effects via induced S arrest in the cell cycle progression and induced cell apoptosis.
Many previous studies found that chemotherapy drugs are not only involved with cell-killing effect on cancer cells, but also exert a considerable effect on the expression of certain genes which are associated with the occurrence of tumors and the outcome in patients. Our research strongly suggests that the expression of TrkA, TrkB and TrkC may be influenced by different concentration and action time of chemotherapeutics, and there was no similarly report at home and abroad at present. Moreover, the prevalent view currently holds that chemotherapeutics via affecting the expression of tyrosine kinase receptor are able to lead to the change of biological behaviors of tumor cells. Rubenstein [39] has proven that Cyclophosphamide(CTX) was efficacious against transforming growth factor-alpha (TGF-alpha) and its binding site, the epidermal growth factor receptor (EGFR) in PC-3 and LNCaP prostate tumors, and the greater the concentration, the stronger CTX-induced apoptosis with more significantly TGF-α and EGFR decreased. Shu [40] demonstrates that DDP enhances the apoptosis which is accompanied with increased cleaved caspase-9 expression and down-regulation of type-1 insulin-like growth factor receptor (IGF-1R). We suspect that the expression of Trk in NB might be affected depending on chemotherapy agents, because Trk belongs to tyrosine kinase receptor family which include TGF-α, EGFR and IGF-1R [41-43].
Here we addressed the effect of conventional chemotherapeutic agents on TrkA, TrkB and Trk C expression in SK-N-SH cells by using real-time RT-PCR, and we found that TrkA expression was consistently up-regulated through ADM, DDP and VP16 treatment. Clinical observation has indicated that TrkA is highly expressed in a subset of favorable NB like stage1, stage2 and stage4s [44], and may be involved in the spontaneous regression or have low tumorigenic potential [45]. Indeed, high levels of expression of TrkA in NB appear to be statistically correlated with low tumor grade and favorable prognosis [46], and upregulation of the TrkA receptor is linked to a better tumor phenotype and tumor spontaneous regression. Lavoie et al clearly indicates that TrkA when expressed at sufficient levels has ability to mediate apoptosis of NB cells by modulating the levels of a number of proteins involved in regulating cell survival and apoptotic signaling, including p53, Bcl2 and casepase-3 [47]. So, in our study we clearly concluded that we should increase the concentration of DDP and VP16 and reduce the concentration of ADM to better maintain and promote the TrkA expression when NB patients with highly expression of TrkA.
Previous studies determined that TrkB detectable in 50–60% of high-risk NB [45], and could be significantly associated with poor clinical outcome and increased resistance to chemotherapy-induced apoptosis in NB [48]. Indeed, TrkB is also a potent oncogene in NB, through activation of the PI3K/AKT pathway, a significant mechanism of facilitating resistance to several chemotherapeutic agents in NB [49]. Furthermore, Schulte [50] found that SY5Y-TrkB which highly expressed TrkB resulted in a significant increasing resistance to DDP obviously, and this was involved in the excessive expression of MDR gene1, and this result is in line with Jaboin [19] 's finding. TrkB-BDNF pathway can be blocked by pre-treatment with either the tyrosine kinase inhibitor or PI-3-kinase inhibitor, and then sensitizes the cell response to chemotherapy [21, 51], and demonstrated the potential as a realistic target in overcoming therapy resistance. However, there is no literature reporting that the TrkB inhibitors have been realized in the clinic [52]. Therefore, the way which TrkB could be inhibited by changing existing chemotherapy strategy, should be found to extensively develop an improved therapeutic treatment in NB. Here, we have experimentally validated that TrkB expression was also consistently up-regulated with the increasing concentration of DDP and VP16, due to the high expression of TrkB in NB was detected in higher stage tumors like stage3 and stage4s with MYCN-amplified [15, 27], TrkB upregulation would might induced NB cell resistance to the cytotoxic drug. In agreement with our study, Tang [53] detected that the expression of TrkB kept a trend of increasing to peak at day 1-3 during the DDP treatment and decreased at day 5 early. Thus, to down-regulate the expression of TrkB would potentially provide benefit in the patients suffering from aggressive NB which is high expression of TrkB.
Higher expression of TrkC is associated with favorable clinical outcome where TrkC expression is associated with increased sensitivity of NB cells to chemotherapy and improved patient survival [54]. Interestingly, TrkC is observed in favorable NBs, and TrkC was not expressed in MYCN-amplified tumors [55]. As with TrkA, TrkC is a novel predictive factor of NB, functioning, in part, as increasing sensitivity to DDP by targeting and downregulation of anti-apoptotic BCL2. Finally, we proposed here that appropriately increasing the concentration of DDP ad VP16 to upregulated of TrkC expression and apparently improve prognosis in high-expression TrkC NB. When it comes to ADM, appropriately reducing the dose is feasible, and that further decrease the drug related toxicity.
So far, there is no relevant report on changing Trk gene expression by adjusting commonly chemotherapy drugs to increase the efficacy of chemotherapy. In this report, our studies first provided compelling evidence that NB patients should be multi-grade treated according to tumor stages so as to increase the TrkA and TrkC expression and inhibit the TrkB expression by adjusting the action time and the concentration of different drugs appropriately. Furthermore, the results added to a growing body of literature characterizing the Trk expression in NB, thus emphasized the importance of analyzing alterations in this genetic marker for prognostic determinants.
5. Acknowledgements
This work was supported by Grant 2014A030313024 from the Guang Dong Natural Science Foundation; Grant from the Guangdong Science and Technology Department (2015B050501004).
6. Compliance with ethical standards
Yes
7. Conflict of interest
The authors declare that they have no conflict of interest.
8. Research involving human or animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
References
- Weinstein JL, Katzenstein HM and Cohn SL. Advances in the diagnosis and treatment of neuroblastoma. Oncologist 8 (2003): 278-292.
- Benard J, Raguenez G, Kauffmann A, et al. MYCN-non-amplified metastatic neuroblastoma with good prognosis and spontaneous regression: a molecular portrait of stage 4S. Mol Oncol 2 (2008): 261-271.
- Tacconelli A, Farina AR, Cappabianca L, et al. Alternative TrkAIII splicing: a potential regulated tumor-promoting switch and therapeutic target in neuroblastoma. Future Oncol 1 (2005): 689-698.
- Zage PE, Graham TC, Zeng L, et al. The selective Trk inhibitor AZ623 inhibits brain-derived neurotrophic factor-mediated neuroblastoma cell proliferation and signaling and is synergistic with topotecan. Cancer 117 (201): 1321-1391.
- Whittle SB, Smith V, Doherty E, et al. Overview and Recent Advances in the Treatment of Neuroblastoma. Expert Rev Anticancer Ther 17 (2017): 369-386.
- Bagatell R and Cohn SL. Genetic discoveries and treatment advances in neuroblastoma. Curr Opin Pediatr 28 (2016): 19-25.
- George RE, Lahti JM, Adamson PC, et al. Phase I study of decitabine with doxorubicin and cyclophosphamide in children with neuroblastoma and other solid tumors: a Children's Oncology Group study. Pediatr Blood Cancer 55 (2010): 629-638.
- Modak S and Cheung NK. Disialoganglioside directed immunotherapy of neuroblastoma. Cancer Invest 25 (2007): 67-77.
- Kim S, Kang J, Qiao J, Thomas RP, et al. Phosphatidylinositol 3-kinase inhibition down-regulates survivin and facilitates TRAIL-mediated apoptosis in neuroblastomas. J Pediatr Surg 39 (2004): 516-521.
- Nyalendo C, Sartelet H, Barrette S, et al. Identification of membrane-type 1 matrix metalloproteinase tyrosine phosphorylation in association with neuroblastoma progression. BMC Cancer 9 (2009): 422.
- Redden RA, Iyer R, Brodeur GM, et al. Rotary bioreactor culture can discern specific behavior phenotypes in Trk-null and Trk-expressing neuroblastoma cell lines. In Vitro Cell Dev Biol Anim 50 (2014): 188-193.
- Iyer R, Wehrmann L, Golden RL, et al. Entrectinib is a potent inhibitor of Trk-driven neuroblastomas in a xenograft mouse model. Cancer Lett 372 (2016): 179-186.
- Harel L, Costa B and Fainzilber M. On the death Trk. Dev Neurobiol 70 (2010): 298-303.
- Bernard-Gauthier V, Aliaga A, Aliaga A, et al. Syntheses and evaluation of carbon-11- and fluorine-18-radiolabeled pan-tropomyosin receptor kinase (Trk) inhibitors: exploration of the 4-aza-2-oxindole scaffold as Trk PET imaging agents. ACS Chem Neurosci 6 (2015): 260-276.
- Evangelopoulos ME, Weis J and Kruttgen A. Neurotrophin effects on neuroblastoma cells: correlation with trk and p75NTR expression and influence of Trk receptor bodies. J Neurooncol 66 (2004): 101-110.
- Brodeur GM, Minturn JE, Ho R, et al. Trk receptor expression and inhibition in neuroblastomas. Clin Cancer Res 15 (2009): 3244-3250.
- Lachyankar MB, Condon PJ, Daou MC, et al. Novel functional interactions between Trk kinase and p75 neurotrophin receptor in neuroblastoma cells. J Neurosci Res 71 (2003): 157-172.
- Brodeur GM. Neuroblastoma: biological insights into a clinical enigma. Nat Rev Cancer 3 (2003): 203-216.
- Jaboin J, Kim CJ, Kaplan DR, et al. Brain-derived neurotrophic factor activation of TrkB protects neuroblastoma cells from chemotherapy-induced apoptosis via phosphatidylinositol 3'-kinase pathway. Cancer Res 62 (2002): 6756-6763.
- Weinreb I, Goldstein D, Irish J, et al. Expression patterns of Trk-A, Trk-B, GRP78, and p75NRT in olfactory neuroblastoma. Hum Pathol 40 (2009): 1330-1335.
- Ho R, Eggert A, Hishiki T, et al. Resistance to chemotherapy mediated by TrkB in neuroblastomas. Cancer Res 62 (2002): 6462-6466.
- Li Z, Zhang Y, Tong Y, et al. Trk inhibitor attenuates the BDNF/TrkB-induced protection of neuroblastoma cells from etoposide in vitro and in vivo. Cancer Biol Ther 16 (2015): 477-483.
- Scala S, Wosikowski K, Giannakakou P, et al. Brain-derived neurotrophic factor protects neuroblastoma cells from vinblastine toxicity. Cancer Res 56 (1996): 3737-3742.
- Yanai T, Okazaki T, Yamataka A, et al. A rare case of bilateral stage IV adrenal neuroblastoma with multiple skin metastases in a neonate: diagnosis, management, and outcome. J Pediatr Surg 39 (2004): 1782-1783.
- Xiong X, Li Y, Liu L, et al. Arsenic trioxide induces cell cycle arrest and affects Trk receptor expression in human neuroblastoma SK-N-SH cells. Biol Res 51 (2018): 18.
- Keshelava N, Davicioni E, Wan Z, et al. Histone deacetylase 1 gene expression and sensitization of multidrug-resistant neuroblastoma cell lines to cytotoxic agents by depsipeptide. J Natl Cancer Inst 99 (2017): 1107-1119.
- Meirelles K, Benedict LA, Dombkowski D, et al. Human ovarian cancer stem/progenitor cells are stimulated by doxorubicin but inhibited by Mullerian inhibiting substance. Proc Natl Acad Sci U S A 109 (2012): 2358-2363.
- Liu L, Li Y, Xiong X, et al. Low dose of arsenic trioxide inhibits multidrug resistant-related P-glycoprotein expression in human neuroblastoma cell line. Int J Oncol 49 (2016): 2319-2330.
- Matthay KK, Reynolds CP, Seeger RC, et al. Long-term results for children with high-risk neuroblastoma treated on a randomized trial of myeloablative therapy followed by 13-cis-retinoic acid: a children's oncology group study. J Clin Oncol 27 (2009): 1007-1013.
- Berlanga P, Canete A and Castel V. Advances in emerging drugs for the treatment of neuroblastoma. Expert Opin Emerg Drugs 22 (2017): 63-75.
- Esposito MR, Aveic S, Seydel A, et al. Neuroblastoma treatment in the post-genomic era. J Biomed Sci 24 (2017): 14.
- Bartholomew J, Washington T, Bergeron S, et al. Dinutuximab: A Novel Immunotherapy in the Treatment of Pediatric Patients With High-Risk Neuroblastoma. J Pediatr Oncol Nurs 34 (2017): 5-12.
- Mora J. Dinutuximab for the treatment of pediatric patients with high-risk neuroblastoma. Expert Rev Clin Pharmacol 9 (2016): 647-653.
- Balwierz W, Wieczorek A, Klekawka T, et al. Treatment results of children with neuroblastoma: report of Polish Pediatric Solid Tumor Group. Przegl Lek 67 (2010): 387-392.
- He Z, Hu B, Tang L, et al. The overexpression of MRP4 is related to multidrug resistance in osteosarcoma cells. J Cancer Res Ther 11 (2015): 18-23.
- Siriwardana G and Seligman PA. Two cell cycle blocks caused by iron chelation of neuroblastoma cells: separating cell cycle events associated with each block. Physiol Rep 1 (2013): e00176.
- Zhang H, Thomas R, Oupicky D, et al. Synthesis and characterization of new copper thiosemicarbazone complexes with an ONNS quadridentate system: cell growth inhibition, S-phase cell cycle arrest and proapoptotic activities on cisplatin-resistant neuroblastoma cells. J Biol Inorg Chem 13 (2008): 47-55.
- Rader J, Russell MR, Hart LS, et al. Dual CDK4/CDK6 inhibition induces cell-cycle arrest and senescence in neuroblastoma. Clin Cancer Res 19 (2013): 6173-6182.
- Shu S, Yang Y, Li X, et al. Down-regulation of IGF-1R expression inhibits growth and enhances chemosensitivity of endometrial carcinoma in vitro. Mol Cell Biochem 353 (2011): 225-233.
- Van Valen F, Fulda S, Schafer KL, et al. Selective and nonselective toxicity of TRAIL/Apo2L combined with chemotherapy in human bone tumour cells vs. normal human cells. Int J Cancer 107 (2003): 929-940.
- Imhof I, Gasper WJ and Derynck R. Association of tetraspanin CD9 with transmembrane TGF{alpha} confers alterations in cell-surface presentation of TGF{alpha} and cytoskeletal organization. J Cell Sci 121 (2008): 2265-2274.
- Negi A, Ramarao P and Kumar R. Recent advancements in small molecule inhibitors of insulin-like growth factor-1 receptor (IGF-1R) tyrosine kinase as anticancer agents. Mini Rev Med Chem 13 (2013): 653-681.
- Zhang P, Xing Z, Li X, et al. Tyrosine receptor kinase B silencing inhibits anoikisresistance and improves anticancer efficiency of sorafenib in human renal cancer cells. Int J Oncol 48 (2016): 1417-1425.
- El-Sayed MI, Ali AM, Sayed HA, et al. Treatment results and prognostic factors of pediatric neuroblastoma: a retrospective study. Int Arch Med 3 (2010): 37.
- Croucher JL, Iyer R, Li N, et al. TrkB inhibition by GNF-4256 slows growth and enhances chemotherapeutic efficacy in neuroblastoma xenografts. Cancer Chemother Pharmacol 75 (2015): 131-141.
- Nakagawara A. Trk receptor tyrosine kinases: a bridge between cancer and neural development. Cancer Lett 169 (2001): 107-114.
- Lavoie JF, Lesauteur L, Kohn J, et al. TrkA induces apoptosis of neuroblastoma cells and does so via a p53-dependent mechanism. J Biol Chem 280 (2005): 29199-29207.
- Ryan J, Tivnan A, Fay J, et al. MicroRNA-204 increases sensitivity of neuroblastoma cells to cisplatin and is associated with a favourable clinical outcome. Br J Cancer 107 (2012): 967-976.
- Fujikawa H, Tanaka K, Toiyama Y, et al. High TrkB expression levels are associated with poor prognosis and EMT induction in colorectal cancer cells. J Gastroenterol 47 (2012): 775-784.
- Schulte JH, Schramm A, Pressel T, et al. Microarray-analysis: a new approach to study the molecular mechanisms of thermo-chemotherapy. Klin Padiatr 215 (2003): 298-302.
- Jaboin J, Hong A, Kim CJ, et al. Cisplatin-induced cytotoxicity is blocked by brain-derived neurotrophic factor activation of TrkB signal transduction path in neuroblastoma. Cancer Lett 193 (2003): 109-114.
- Nakamura Y, Suganami A, Fukuda M, et al. Identification of novel candidate compounds targeting TrkB to induce apoptosis in neuroblastoma. Cancer Med 3 (2014): 25-35.
- Tang Y, Wang P, Du B, et al. Expressions of neurotrophin factor receptor in spiral ganglion cell of cisplatin-induced ototoxicity. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 43 (2008): 950-955.
- Fung W, Hasan MY, Loh AH, et al. Gene expression of TRK neurotrophin receptors in advanced neuroblastomas in Singapore--a pilot study. Pediatr Hematol Oncol 28 (2011): 571-578.
- de Souza DR, Sanabani SS, de Souza AC, et al. Prognostic impact of MYCN, DDX1, TrkA, and TrkC gene transcripts expression in neuroblastoma. Pediatr Blood Cancer 56 (2011): 749-756.