Pleural Empyema in Children in Senegal-What’s New?
Article Information
Ba Idrissa Demba1, Sow Amadou2*, Dieng Yaay Joor1, Mbacké Rokhaya1, Fall Tall Fatim1, Ba Abou1, Faye Papa Moctar1, Fall Amadou Lamine1, Ndiaye Ousmane1
1Albert Royer National Children's Hospital Center, Dakar, Senegal
2Abass Ndao Hospital Center, Dakar, Senegal
*Corresponding Author: Amadou Sow, Pediatrician, University Hospital Center of Dakar, Senegal
Received: 22 June 2020; Accepted: 20 July 2020; Published: 24 July 2020
Citation:
Ba Idrissa Demba, Sow Amadou, Dieng Yaay Joor, Mbacké Rokhaya, Fall Tall Fatim, Ba Abou, Faye Papa Moctar, Fall Amadou Lamine, Ndiaye Ousmane. Pleural Empyema in Children in Senegal-What’s New?. Journal of Pediatrics, Perinatology and Child Health 4 (2020): 058-064.
Share at FacebookAbstract
Introduction: Purulent pleurisy is a severe form of acute respiratory infection. They are a major concern in pediatric practice. The aim of our work is to study the epidemiological, clinical, therapeutic and progressive profile of purulent pleurisy (PP) in children in Senegal.
Patients and methods: This is a retrospective and descriptive study, including children aged 0 to 15 years hospitalized for PP from January 2014 to December 2017 at the Albert Royer Hospital Center. The positive diagnosis of PP was retained in front of the purulent, cloudy or citrine aspect of the pleural puncture fluid with the presence of altered neutrophilic polymorphonuclear cells.
Results: The hospital frequency of PP was 0.6%. Sex ratio 1.42. The average age was 49 months. The age group of 6 to 36 months was the most represented. Malnutrition was present in 13 patients. In 91% of cases pleurisy was unilateral and right in 59% of cases. Pneumonia was radiologically associated in 54 children and pneumothorax in 19 children. The lethality was 6.6% (8 patients). The low socioeconomic level (p=0.0001) and the appearance of frankly purulent pleural fluid (p=0.002) were statistically significant associated with this mortality.
Conclusion: Purulent pleurisy is associated with significant morbidity and mortality. Prevention involves vaccination, strengthening strategies to combat malnutrition and supplementation with zinc and vitamin A.
Keywords
Pleurisy, Purulent, Children, Drainage, Senegal
Pleurisy articles, Purulent articles, Children articles, Drainage articles, Senegal articles
Article Details
1. Introduction
Pleuro-pulmonary suppurations represent the severe form of acute respiratory infections (ARI). They are a common concern in pediatric practice in our context. According to the World Health Organization, acute respiratory infections, pneumonia, are the leading cause of death in children under the age of 5 worldwide, especially in low-income countries [1, 2]. Pneumonia is often complicated by purulent pleurisy, which can be life-threatening. Since the 1990s, an increase in purulent pleurisy has been reported worldwide [3, 4]. Many studies have focused on this subject in children in Africa and Senegal in recent decades. The aim of our study is to make a recent inventory by studying the epidemiological, clinical, therapeutic and progressive profile of purulent pleurisy in children in a Dakar hospital center.
2. Methodology
Our study took place at the National Hospital Center for Children Albert Royer (CHNEAR). It is a retrospective and descriptive study. It included children aged 0 to 15 years hospitalized for purulent pleurisy (PP) during the period from January 1, 2014 to December 31, 2017 (4 years). The positive diagnosis of purulent pleurisy was retained in front of a purulent pleural puncture fluid, or cloudy or citrin with the presence of altered neutrophilic polymorphonuclear cells. Data were collected from hospital patient records. For each patient, we noted socio-demographic data, personal and family history, co-morbidities, clinical, radiological, biological, bacteriological, therapeutic and evolutionary data. The data were entered and analyzed using epi-info software (version 3.5.4) and Microsoft office Excel 2007 on Windows 7. The analysis of our data was done using the chi-square method (chi 2), and a p ≤ 0.05 is statistically significant.
3. Results
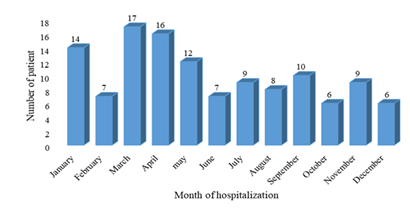
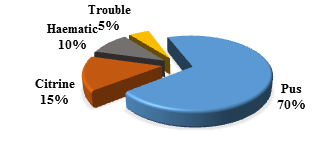
During the study period, 121 patients were hospitalized for purulent pleurisy (PP) out of 19608 patients, i.e. a hospital prevalence of 0.6%. The sex ratio was 1.42. The mean age of the children was 49 months [1 to 192 months]. The 6 to 36-month age group represented 44.63%. Peak hospitalizations were noted in March and April (Figure 1). A low socioeconomic level was found in 99 patients (82%). Vaccination was up to date, according to the vaccination program of Senegal, in 110 patients (91%), and 11 patients (9%) were incompletely vaccinated. The average consultation time was 13 days [1 to 60 days]. Nine (9) children received ibuprofen and 35 children received outpatient antibiotic therapy before hospitalization. The main sites and/or comorbidities found in our children were malnutrition in 13 cases, anemia in 44 cases and sickle cell anemia in 7 cases. The main reasons for consultation were fever in 94%, cough in 65% and difficulty breathing in 72%, as shown in the Table 1. The pleurisy was unilateral in 91% of the cases and localized on the right in 59% of the cases. Pleural effusion was of medium abundance in 53% (or 64 children) and of high abundance in 20% (or 24 children). An image of pneumonia was radiologically associated in 54 children and a pneumothorax in 19 children. The distribution according to the macroscopic aspect of the pleural fluid is represented in the figure 2. The average cellularity of the pleural fluid was 2457 elements/mm³ [5 to 54000/mm³] with altered neutrophils (PNA) in all cases. Bacteriologically, the blood culture carried out was positive in 4 children. The bacteriological research carried out on all the pleural samples returned positive on 69 samples (57%): latex alone (34 cases; 23%), culture alone (35 cases; 51%) and the two positive in (18cases, 26%) (Table 2). The main germs found were Streptococcus pneumoniae (36%), staphylococcus aureus (33%) and Klebsiella pneumoniae (10%) as shown in the Table 3. We did not note a strain of pneumococcus with reduced sensitivity to penicillin. Eighty percent (82.6%) of the staph strains were sensitive to methicillin. Therapeutically, pleural drainage was performed in 62 patients (51%). The average duration of pleural drainage was 9 days [2 and 20 days] and the average amount of fluid drained was 1175 ml [60 to 3900 ml]. The main complications found were pachypleuritis in 32 cases, pneumothorax in 8 cases and septic shock in 5 cases. In our study, 8 children died (6.6%). The majority of deaths occurred after 72 hours in 75% (6 patients). The low socioeconomic level constitutes a prognostic factor (p=0.0001).
|
Characteristics |
Effective (N=121) |
Percentage (%) |
|
Age range (in months) |
||
|
0 to 6 months |
10 |
8.2 |
|
6 to 36 months |
60 |
49.5 |
|
36 to 72 months |
20 |
16.5 |
|
> 72 months |
31 |
25.6 |
|
Sex |
||
|
Feminine |
50 |
41 |
|
Male |
71 |
59 |
|
Socio-economic level |
99 |
82 |
|
Terrain and / or co-morbidities |
||
|
Malnutrition |
13 |
10.7 |
|
Anemia |
44 |
36 |
|
Sickle cell anemia |
7 |
5.8 |
|
Clinical signs |
||
|
Fever |
114 |
94 |
|
Cough |
79 |
65 |
|
Breathing difficulties |
87 |
72 |
|
Fluid effusion syndrome |
100 |
82 |
|
Pulmonary condensation syndrome |
57 |
47 |
|
Complications |
||
|
Pachypleuritis |
32 |
26 |
|
Pneumothorax |
8 |
6.6 |
|
Septic shock |
5 |
4.1 |
|
Acute respiratory failure |
2 |
1.6 |
|
Death |
8 |
6.6 |
Table 1: Overall characteristics of children with purulent pleurisy.
|
Bacteriological research |
effective |
Percentage |
|
Latex alone positive |
34 |
23% |
|
Culture alone positive |
35 |
51% |
|
Latex and culture positive |
18 |
26% |
|
Total |
69 |
100% |
Table 2: Results of bacteriological examinations.
|
Isolated germs |
Effective (n=69) |
Percentage (%) |
|
Streptococcus pneumoniae |
26 |
37.6 |
|
Staphylococcus aureus |
23 |
33.3 |
|
Klebsiella pneumoniae |
6 |
8.7 |
|
Eschericha coli |
4 |
5.8 |
|
Pseudomonas aeruginosa |
3 |
4.3 |
|
Staaphylococus spp |
2 |
3 |
|
Other |
5 |
7.3 |
Table 3: The main germs isolated in children.
4. Discussion
The hospital incidence of PP is 0.6% at the Albert Royer Children's Hospital in Dakar. This incidence has gradually decreased in recent years in Senegal. Ndiaye in 2000 and then Thiam reported respective incidences of 1% and 0.7% [5, 6]. This downward trend is global. In fact, during the 20th century, the incidence of pleuropneumonia worldwide fell sharply mainly due to advances in the medical field, notably antibiotic therapy, better access to vaccination and improvement in conditions hygiene [7]. In our series, PP occur throughout the year with peaks of occurrence noted in the months of March and April. These months correspond to the cold and dry period of the year. This climate favors the occurrence of nasopharyngeal infections, thus weakening the respiratory mucosa and facilitating the spread of germs [6, 8]. PP occurs at any age. However, infants and young children under the age of 5 are the most likely. The average age of our children (49 months) is comparable to that found in the literature in Africa [9, 10, 5, 6]. In France an average age of 43.2 months has been reported [11]. In the Kouéta study in Burkina Faso, infants aged 6 to 36 months were the most represented [9]. The vulnerability of young infants is mainly explained by the immaturity of their immune system, the pharyngeal carriage of germs and the high frequency of otolaryngeal and skin infections which constitute the main entry points for respiratory infections. [12, 13]. In our series, this tranche represents 44.5% of the total workforce. In addition, we noticed that 82% of the children came from families with a low socio-economic level. This could contribute to limited access to health services, delayed diagnosis and treatment with an impact on the life prognosis of these serious conditions. Our average consultation time was long (13 days on average). Longer consultation times have been described in the literature [5, 6]. Malnutrition was found in 10.7% of our children, while Ndiaye reported 32.78% in 2000 [5]. Malnutrition is a debilitated field favoring the occurrence of bacterial infections. However, we have not found any case of HIV infection in our patients. This is explained on the one hand by the fact that the prevalence of HIV infection in Senegal is low at 0.5% [14]. On the other hand, not all the children had systematically benefited from HIV serology. Prospective studies are needed to assess the association between HIV and purulent pleurisy. In the series, 9 children received ibuprofen before hospitalization. Taking non-steroidal anti-inflammatory drugs during a viral infection of the upper respiratory tract (angina, pharyngitis) or pneumonia is a factor in the occurrence of purulent pleurisy in children [15, 16]. The bacteriological study of pleural fluid isolated a germ in 57% of the cases in our series. This low bacteriological yield is explained by the excessive prescription of antibiotics, which are often unsuitable. However, the use of molecular detection techniques such as Polymerase Chain Reaction (PCR) could improve the bacteriological yield, as is the case in developed countries [17, 18]. In our study Streptococcus pneumoniae was the first isolated germ followed by Staphylococcus aureus. The pneumococcal strains isolated from our children have a good sensitivity to the usual antibiotics. NDIAYE [4] reported a rate of pneumococcus with reduced penicillin sensitivity of 42%. In series studies as in that of Thiam [5], no strain of PSDP was found. Serotyping of isolated pneumococcal strains is not available in the context of our study. This constitutes a real weakness of our study to determine whether it was a vaccine pneumococcal strain or the emergence of other non-vaccine strains. Because 13 valence pneumococcal conjugate vaccine (PCV13) has been introduced in the Senegalese expanded vaccination program since November 2013 and 91% of our children had up-to-date vaccination coverage. All of our children had received intravenous antibiotic therapy. The use of corticosteroid therapy is very controversial in PP. Corticosteroid therapy has no effect on the development of purulent pleurisy in children [5]. In our series, 5 children had received corticosteroid therapy. Percutaneous pleural drainage remains a widespread, effective and less costly method of removing pleural pus without general anesthesia. It must be carried out urgently [19]. Its effectiveness is reduced when the drain is placed late. Improving the accessibility of chest drainage kits in our reception and pediatric emergency services could improve the prognosis of PP in our regions, as well as the use of fibrinolytics. Other pus removal techniques such as video-assisted thoracoscopy and surgery are very expensive because they require general anesthesia in the operating room [2].
5. Conclusion
Purulent pleurisy remains a very common concern in pediatric practice in our regions. Pneumococcus is the first germ involved. The lethality is significant, hence the need to strengthen prevention strategies through routine vaccination, the fight against malnutrition and a generalization of micronutrient supplementation such as zinc and vitamin A.
References
- World Health Organisatin 2015 WHO. WHO fact sheet: Pneumonia (2015).
- Bryce J, Boschi-Pinto C, Shibuya K, et al. WHO estimates of the causes of death in children. Lancet 365 (2005): 1147-1152.
- Eastham KM, Freeman R, Kearns AM, et al. Clinical features etiology and outcome of empyema in children in the north east of England. Thorax 59 (2004): 522-525.
- Byington C, Spencer l, Johnson T, et al. An epidemiological Investigation of a sustained high rate of pediatric parapneumonic empyema: risk factors and microbiological associations. Clin Fac Dis 34 (2002): 434-440.
- Ndiaye O, Diack-Mbaye A, Ba M, et al. Purulent pleurisy with childhood staphylococcus aureus. Experience of the Albert-Royer Children's Hospital of the Fann CHU in Dakar. Health 10 (2000): 93-96.
- Thiam L, Faye PM, Ba ID, et al. Purulent pleurisy in children: experience from the Albert Royer National Children's Hospital in Dakar (Senegal). Rev. HEALTH CAMES 5 (2017): 34-40.
- Blanc P, Dubus JC, Bosdure E, et al. Community purulent pleurisy of the child. Where are we? Arch Pediatrician 14 (2007): 64-72.
- Ba ID, Diagne G, Ba A, et al. Epidemiological, clinical, microbiological therapeutic and evolutionary aspect of the acute respiratory affections of the child about a prospective study at the Albert-Royer center of Dakar. Rev Mal Resp 36 (2019): 221-222.
- Koueta F, Ouedraogo SO, Ouedraogo G, et al. Pleuresies in children: epidemiological, clinical, paraclinical, therapeutic and progressive aspects at the Carles De Gaulle pediatric university hospital in Ouagadougou (Burkina Fasso). Clincs in Mother and Child Health 8 (2011): 1-6.
- Sawadogo A, Koweta F, Sanou I, et al. Pleurum-pulmonary staphylococcal disease in infants in hospitals in Ouagadouguou (Burkina Fasso). Cahiers Santé 9 (1999): 209-213.
- Schultz KD, Fan LL, Pinsky J, et al. In children, there is no consensus in the management of purulent pleurisy. Pediatrics 113 (2004): 1735-1740.
- Amon-Tanoh-Dick F, Timite-Konan AM, Biafry MM, et al. Bacterial non-tuberculous pneumonia the child in Abidjan. Cahiers Santé 9 (1999): 145-149.
- Alao MJ, Sagbo GG, Diakite AA, et al. Pleurisy in children at the national hospital and university center of Cotonou: epidemiological, clinical, paraclinical and therapeutic aspects. Mali Med 4 (2010): 47-51.
- Republic of Senegal. National Agency for Statistics and Demography. Demographic and Health Survey continues (EDS-continues 2017). Dakar: ANSD (2018).
- Le Bourgeois M, Le Bourgeois M. Nonsteroidal Anti-Inflammatory Drug withoutAntibiotics for Acute Viral Infection Increases the EmpyemaRisk in Children: A Matched Case-Control Study. J Pediatr 175 (2016): 47-53.
- Voiriot G. Risks associated with the use of non-steroidal anti-inflammatory drugs during pneumonia. Rev Mal Respir 35 (2018): 430-440.
- Weil-olivier C, Levy C, Marguet C, et al. Multicentric retrospective survey on infectious pleuropneumopathies in children in France. Arch Pediatrician 12 (2005): 823-826.
- Desrumaux A, Desrumaux A, François P, et al. Epidemiology and clinical characteristics of suppurative complications in childhood pneumonia. Arch Pediatr 14 (2007): 1298-1303.
- Debesse B, Bellemy J. Pleural drainage and eradication of the pulmonary focus: standard treatment of acute purulent pleurisy with banal germ. Rev Fr Mal Resp 11 (1983): 245-246.