Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION)
Article Information
Jaya Shree D*, Rajganesh Ravichandran, Raveena P B, Abarna Lakshmi R, Nikhil Cherian Sam
Department of Pharmacy Practice, C.L. Baid Metha College of Pharmacy, The Tamil Nadu Dr.M.G.R. Medical University, Chennai, Tamil Nadu, India
Corresponding Author: Jaya Shree D, Department of Pharmacy Practice, C.L. Baid Metha College of Pharmacy, The Tamil Nadu Dr.M.G.R. Medical University, Chennai, Tamil Nadu, India
Received: 19 July 2020; Accepted: 29 July 2020; Published: 03 August 2020
Citation: Jaya Shree D, Rajganesh Ravichandran, Raveena P B, Abarna Lakshmi R, Nikhil Cherian Sam. Non- Arteritic Anterior Ischemic Optic Neuropathy (NAION)
Share at FacebookAbstract
We present a non-arteritic anterior ischemic optic neuropathy (NAION) with unknown cause, but good recovery after the management with antiplatelet therapy in a 45-year-old male patient who came to the hospital with complaints of sudden painless loss of vision.
Keywords
Optic Disc Edema; Non-Arteritic Anterior Ischemic Optic Neuropathy (NA-AION); Visual Field; Optic Nerve Atrophy
Article Details
Introduction
Ischemic optic neuropathy is an acute ischemic disorder of the optic nerve. The characteristic of Anterior Ischemic Optic Neuropathy (AION) is visible disc edema because of ischemic damage to the anterior portion of the optic nerve that is supplied by Posterior Ciliary Arteries (PCA). AION is the most common cause of acute optic nerve ischemia related vision loss in adults and secondary cause of optic nerve-related permanent vision loss in adults after glaucoma [1]. AION is of two types i.e. arteritic and non-arteritic types. Arteritic Anterior Ischemic Optic Neuropathy (AAION) is secondary to inflammatory small vessel disease, i.e., vasculitis, whereas Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION) is secondary to non-inflammatory small vessel disease [2]. NAION is mostly of unknown cause, though many systemic and ocular risk factors have been identified. Among systemic factors, nocturnal hypotension, hypertension, diabetes mellitus, hyperlipidemia, and smoking are associated, whereas among ocular risk factors crowding of optic nerve head is the important one. Some of the common causes of NAION are sleep apnea syndrome, optic disc drusen, and medications such as interferon and sildenafil [3]. NAION is diagnosed using fundus fluorescein angiography, ICG angiography, Colour Doppler, and laser Doppler flow. There is no complete cure to the disease. NAION is managed only through antiplatelet and anticoagulant drugs [4].
NAION is the most common form, constitutes 95% of AION, and is the most common cause of acute optic neuropathy over the age of 50 years [5]. Annual prevalence of NAION was reported as 0.30% [6] and incidence was found to be as high as 82 cases per 100,000 [7] in large Medicare database study. This occurs most commonly in the white population than black or Hispanic individuals. Inter-ethnic differences in frequency of NAION may be attributed to the different size of optic disc which occurs predominately in small optic nerve head. It occurs equally in frequency in both the gender. Mean age reported was 66 years in IONDT (ischemic optic nerve decompression trial), while in most other studies documented age range is 57-65 years [5].
Case Report
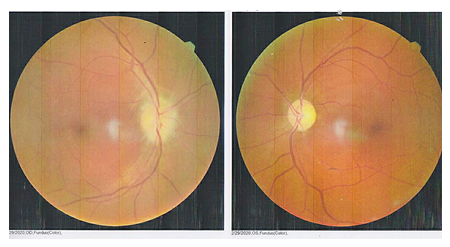
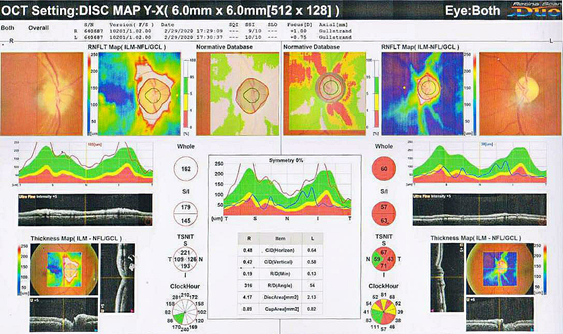
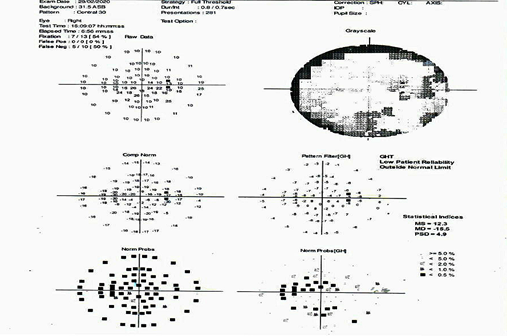
A 45-year-old male came to the hospital with complaints of sudden onset of painless vision loss in the right eye for 1 month. His height is 168 cm; body weight is 75 Kg with a body mass index is 26.6 kg/m2. He is a known case of type II diabetes mellitus and hypertension for the past 12 years. On examination of the patient eyes, it was found that visual acuity was 6/60 in RightEye (RE) and 6/6 in Left Eye (LE), near vision, was N36 in RE and N6 in LE. The intraocular pressure was normal in both the eye and was found to be 18mm Hg. It was observed that during the swinging flashlight test the RE had a sluggish relative afferent pupillary defect (RAPD) but LE showed a positive response. The anterior segment of the lens was found to be clear for both the eye. On examining both the eyes for color vision it was found that there was a partial defect in RE. Fundus examination showed that optic nerve disc in the RE was imprecisely delimited, had a swollen appearance and the cupping was absent, which represents for papillary edema (Figure 1). Visual field examination showed peripheral constriction and inferior arcuate defect of the right eye and normal of the left eye. Optical coherence tomography (OCT) of the optic nerve showed thickening of the nerve fiber layer of the RE (Figure 2). The field study showed all hemianopia in the right eye. Perimetry was assessed by a Humphrey Visual Field Analyzer, Central 30 for both the eyes to analyze the presence of any glaucoma (Figure 3 & 4). This entire investigation shows the patient has bilateral mild non-proliferative diabetic retinopathy with optic atrophy due to diabetes mellitus.
We had done a few more clinical, paraclinical, and laboratory complementary investigations to rule out autoimmune disease. The complete blood count and erythrocyte sedimentation rate had normal values; the biochemistry showed normal values for Phospholipid Ab-IgM, Ab-IgG, Ab-IgA, antinuclear antibody (ANA) screening and Antineutrophil cytoplasmic antibodies (P-ANCA & C-ANCA). The patient had moderate dyslipidemia as there was an increase in the LDL level. There were no significant findings at the neurological exam, that included a cerebral MRI, and the ENT exam showed the existence of a septum deviation irrelevant to the patient’s ophthalmologic pathology.
So, from all these investigations the patient was diagnosed with non-arteritic anterior optic ischemic neuropathy. He was treated with T. Ecospirin AV 150mg per oral for once daily for 3 months, Inj. Renerve Plus IM once daily for 10 days, and T. Rosuvas F 10mg per oral once daily for 10 days. Other regular medications he has been prescribed were T. Reclimet 80/500 per oral twice daily, T. Telvas AM 40/5mg per oral once daily in the morning, Inj. LupIsulin M 30 S.C. 18 units for morning and 15 units for the night and should be administered before food. These drugs were prescribed to control diabetes mellitus and hypertension.
Discussion
Non-ischemic arteritic optic neuropathy was found to be the most common form, which constitutes 95% of AION and is the most common cause of acute optic neuropathy over the age of 50 years. There no proper guidelines have still been established for the management of NAION, but investigative tests such as color Doppler, fundus fluorescein angiography and ICG angiography may help in establishing the early diagnosis and proper management of such cases.
Declaration of Patient Consent
The authors certify that they have obtained all appropriate patient consent forms. The patient has given his consent for his images and other clinical information to be reported in the journal. The patient understood that his name and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial Support and Sponsorship
Nil.
Conflicts of Interest
There are no conflicts of interest.
References
- Miller NR, Arnold AC. Current concepts in the diagnosis, pathogenesis and management of nonarteritic anterior ischaemic optic neuropathy. Eye 29 (2015): 65-79.
- Arnold AC. Pathogenesis of nonarteritic anterior ischemic optic neuropathy. Journal of Neuro-ophthalmology 23 (2003): 157-163.
- Etiology, signs and symptoms, pathogenesis assessed from (https://eyewiki.aao.org/Non-Arteritic_Anterior_Ischemic_Optic_Neuropathy_(NAION)) (2020).
- Patil M, Ganger A, Saxena R. Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION)—A Brief Review. Open Journal of Ophthalmology 6 (2016): 158.
- Guyer DR, Miller NR, Auer CL, et al. The risk of cerebrovascular and cardiovascular disease in patients with anterior ischemic optic neuropathy. Archives of Ophthalmology 103 (1985): 1136-1142.
- Gordon LK, Yu F, Coleman AL. Medicare database analysis of prevalence and risk factors for ischemic optic neuropathy. Ophthalmology 110 (2003): 238.
- Lee MS, Grossman D, Arnold AC, Sloan FA. Incidence of nonarteritic anterior ischemic optic neuropathy: increased risk among diabetic patients. Ophthalmology 118 (2011): 959-963.