Mitral Valve Repair for Functional Mitral Regurgitation-Description of A New Technique and Classification System
Article Information
Antonio Chiricolo1*, Leonard Y Lee2
1 Department of Anesthesiology, Rutgers Robert Wood Johnson Medical School, New Brunswick, NJ, USA
2 Department of Surgery, Rutgers Robert Wood Johnson Medical School, New Brunswick, NJ, USA
*Corresponding Author: Antonio Chiricolo, Department of Anesthesiology, Rutgers Robert Wood Johnson Medical School, New Brunswick, NJ, USA,
Received: 19 November 2017; Accepted: 27 November 2017; Published: 29 November 2017
Share at FacebookAbstract
Herein is reported a case of a 59-year-old male with history of non-compliance, insulin dependent diabetes and congestive heart failure admitted with recurrent symptoms of chest pain with exertion, shortness of breath, and lower extremity edema. Work up including cardiac catheterization and echocardiography revealed severe triple vessel coronary artery disease, severely depressed left ventricular (LV) ejection fraction (EF) of 15% with an enlarged LV cavity (>7.0 cm internal dimension during diastole), severe functional mitral regurgitation (MR; Carpentier type IIIb) [1] and pulmonary artery hypertension (PAP 60/30). A cardiac MRI was obtained to assess viability. Although much of the LV was non-viable, there were patchy areas of viability throughout the LV. Given his relatively young age and the results of the viability, the patient was taken to the operating room for coronary artery bypass (left internal mammary artery to the left anterior descending artery, saphenous vein graft to the obtuse marginal number 1, and saphenous vein graft to the right posterior descending artery), LV assist device standby, and a complex mitral valve repair with release of secondary chordae to the anterior and posterior leaflets, reduction annuloplasty with a 28 mm rigid Carpentier-Edwards Physio annuloplasty ring (Edwards Lifesciences, Irvine, CA, USA) and an Alfieri stitch [2] with 5-0 polypropylene suture to approximate A2 to P2. At the termination of the procedure, there was no mitral regurgitation, preserved LV EF of 15-20%. After 7-day recovery in the hospital the patient was discharged to home and a follow up echocardiogram was performed 6 months post procedure, which demonstrated an LV EF of 15-20% and no mitral regurgitation (no change in the mitral valve over several months). In addition, the patient reported marked improvement in his symptoms with improved exercise tolerance despite the persistently poor ventricular function.
Keywords
Mitral regurgitation; Functional mitral regurgitation; Dysfunction of the mitral valve apparatus; Treatment of functional mitral regurgitation; Surgical management of mitral regurgitation; Mitral regurgitation echocardiography findings; Effective regurgitant orifice area for mitral regurgitation
Article Details
1. Introduction (Pathology and Repair)
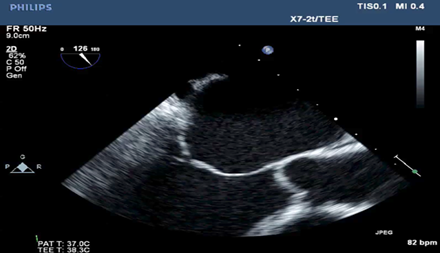
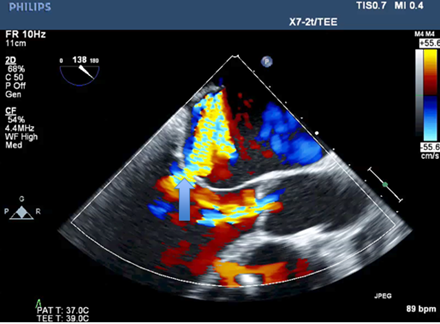
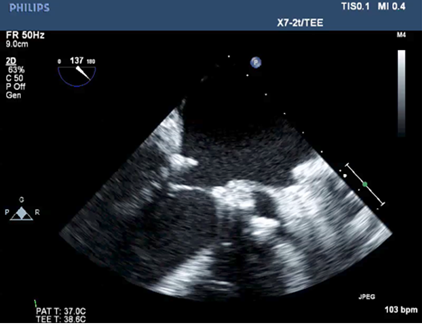
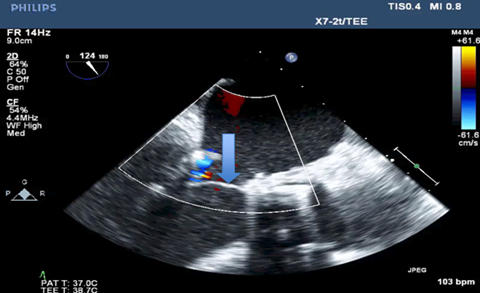
The repair of functional MR has recently been described as ineffective in correcting the regurgitation, and in providing a durable outcome, which often leads to patients developing recurrent MR with no alleviation of symptoms [3-7]. The pre-procedure two- dimensional echocardiography exam noted severe mitral regurgitation based on qualitative data, including severe dilation of the entire mitral valve apparatus (Left atrium, mitral annulus and left ventricle). Color flow Doppler was used to confirm the diagnosis and a large (>40% jet area) regurgitant jet (RJ) directed in a posterior direction was visualized. The direction of the RJ is in part due to the chordae from the posteromedial papillary muscle having attachments to both mitral leaflets. Additional criteria include systolic flow reversal using pulsed wave Doppler in the pulmonary veins and severe pulmonary hypertension. We decided to avoid other quantitative parameters for grading mitral regurgitation due to the complex geometric distortions of the coaptation point in patients with functional disease. These changes may lead to under or overestimation of the effective regurgitant orifice area and regurgitant volumes using the proximal isovelocity surface area (PISA). The cause of MR in patients as described herein, is the tethering of the valve leaflets causing the poor captain principally due to ventricular dilatation and downward and outward movement of the papillary muscles[8,9], causing a "hockey stick" deformity of the leaflets (Figure 1), i.e. a ventricular problem causing valvular pathology. The repair technique described addresses several issues. 1). By releasing the larger secondary chordae (mid-leaflet of anterior and posterior leaflets), it allows the leaflet to assume the billowing conformation and movement of the leaflets towards the atrium during ventricular systole needed for adequate coaptation. 2). Long-standing mitral regurgitation, regardless of the primary valve pathology, leads to worsening of MR due to annular dilatation over time, which further exacerbates the poor coaptation of the leaflets [10] during systole (Figure 2). This annular dilatation is addressed with a reduction annuloplasty [11,12] and in this case the use of a 28 mm full, rigid ring (Figure 3 and 4).
2. Recurrent Mitral Regurgitation
In these cases a flexible ring or a band (incomplete ring) would allow the annulus to develop a conformational change over time and become more circular in nature, as opposed to the native "D" conformation, which provides a mechanism for recurrent MR in these patients [13]. In addition, there is evidence that the fibrous backbone of the heart, aorto-mitral curtain, between the trigones can dilate over time, accounting for recurrent MR [14,15] due to recurrent annular dilatation. Therefore, a full rigid ring is advocated for this repair to restore the annular geometry to its native "D" form and to maintain its dimension and shape over time. Finally, the mechanism of recurrent MR after repair for functional disease is due to further tethering of the leaflets [16]. The full rigid ring addresses the annular dilatation and shape. The larger secondary chordae responsible for the "hockey stick deformity" seen in many of these functional MR patients are addressed with resection and release. Tethering of the primary chordae, inserting on the leading edge of the leaflet and responsible for coaptation, would be the mechanism of recurrent MR in these functional patients provided that the other two mechanisms had been addressed. By placing the Alfieri stitch, the zone of coaptation is maintained, thus not allowing further tethering to affect leaflet coaptation. The alfieri stitch is the only part of this repair that is not routinely used, instead reserved for those instances of severely depressed LV ejection fraction (<25%) and severely increased ventricular dimensions (6.5 cm or greater).
3. Classification (Discussion)
Carpentier in his landmark paper, “The French Correction,” describes a classification system for mitral regurgitation that still exists and is widely used today. In his classification system, type I refers to annular dilatation or leaflet perforation, type II refers to excessive leaflet motion with pathology such as prolapse, ruptured chordae, fibro-elastic deficiency, etc., type III lesions are the result of restrictive leaflet motion. This category is differentiated into two types of restriction. Type IIIa refers to restricted leaflet motion during systole and diastole, the diastolic restriction causes varying degrees of mitral stenosis. Type IIIb refers to restricted leaflet motion during systole and is the result of displacement of the papillary muscles or ventricular wall apically leading to tethering of the mitral leaflets. We also know that longstanding MR leads to annular dilatation and worsening of MR. As a consequence, there are subsets of patients that exhibit characteristics of annular dilatation along with either leaflet pathology or tethering. We propose adding to the existing classification system two additional pathologies. Type IV a would represent type I + type II and type IVb would represent type I+IIIb (Table 1). Given this addition to the current classification schema, potentially all valve pathologies could be adequately described and the appropriate repair techniques determined for the long-term benefit of the patient.
| Type I | Annular Dilatation |
| Type II | Leaflet Prolapse (FED, ruptured chordae) |
| Type III
|
a. Mitral Stenosis |
| b. Functional Mitral Regurgitation (tethering) | |
| Type IV | Combined Disease |
| a. Type I + Type II | |
| b. Type I + Type IIIb |
Table 1: New classification of mitral valve disease,it explains the existing Carpentier classification in black [1], FED ? fibro-elastic deficiency, additional classification schema added in red.
4. Conclusion
The repair technique described affords a durable result for patients with type IIIb and IVb mitral valve disease. In contrast to prior techniques that had been proposed that solely utilized a small ring in an attempt to address this ventricular cause of MR 17], this technique addresses all the issues of failed repair or recurrent MR in patients with functional disease, although more patients and study will be required to ascertain the long-term durability of this repair technique. Perhaps this technique could obviate the need for mitral valve replacement in this patient population in the future. These techniques are applicable to minimally invasive approaches, right mini thoracotomy or robotic techniques. The proposed addition to the classification system as developed by Carpentier could expand the description of mitral valve disease to allow for a complete description of pathology.
References
- Carpentier A. Cardiac-valve surgery- the “French correction.” J Thorac Cardiovascular Surgery 86 (1983): 323-337.
- Alfieri O, Maisano F, De Bonis M, et al. The double-orifice technique in mitral valve repair: a simple solution for complex problems. J Thorac Cardiovascular Surgery 122 (2001): 674-681.
- Dion R, Benetis R, Elias B, et al. Mitral valve procedures in ischemic regurgitation. J Heart Valve Dis. 1995:4 Suppl 2:S124-129; discussion S129-131.
- Grigioni F, Enríquez-Sarano M, Zehr KL, Bailey KR, Tajik AJ. Ischemic mitral regurgitation. Long-term outcome and prognosis implication with quantitative Doppler assessment. Circulation 103 (2001): 1759-1764.
- Dion R, Benetis R, Elias B, Guennaoui T, Raphael D, et al. Mitral valve procedures in ischemic mitral regurgitation. J Heart Valve Dis 2 (1995): S124-S131.
- McGee EC, Gillinov AM, Blackstone EH, Rajeswaran J, Cohen G, et al. Recurrent mitral regurgitation after annuloplasty for functional ischemic mitral regurgitation. J Thorac Cardiovasc Surg 128 (2004): 916-924.
- Zhu F, Otsuji Y, Yotsumoto G, Yuasa T, Ueno T, et al. Mechanism of persistent ischemic mitral regurgitation after annuloplasty: importance of augmented posterior mitral leaflet tethering. Circulation 112 (2005): I396?I401.
- Kwan J, Shiota T, Agler DA, Popovic ZB, Qin JX, et al. Geometric differences of the mitral apparatus between ischemic and dilated cardiomyopathy with significant mitral regurgitation: real-time three-dimensional echocardiography study. Circulation 107 (2003): 1135-1140.
- Liel-Cohn N, Guerrero JL, Otsuji Y, Handschumacher MD, Rudski LG, et al. Design of a new surgical approach for ventricular remodeling to relieve ischemic mitral regurgitation: insights from 3-dimensional echocardiography. Circulation 101 (2000): 2756-2763.
- Enríquez-Sarano M, Basmadjian A, Rossi A, Bailey KR, Seward JB, et al. Progression of mitral regurgitation: a prospective Doppler echocardiographic study. J Am Coll Cardiol 34 (1999): 1137-1144.
- Castillo JG, Milla F, Anyanwu AC, Adams DH. Video-atlas on minimally invasive mitral valve surgery ? The David Adams technique. Ann Cardiothorac Surg 2 (2013): 828-832.
- Adams DH, Filsoufi F, Aklog L. Surgical treatment of the ischemic mitral valve. J Heart Valve Dis 11 (2002): S21-S25.
- McGee EC, Gillinov AM, Blackstone EH, Rajeswaaran J, Cohen G, et al. Recurrent mitral regurgitation after annuloplasty for functional ischemic mitral regurgitation. J Thorac Cardiovasc Surg 128 (2004): 916-924.
- Paul WM Fedak, Patrick M. McCarthy, Robert O. Bonow. Evolving concepts and technologies in mitral valve repair. Circulation 117 (2008): 963-974.
- Magne J, Pibarot P, Dagenais F, Hachicha Z, Dumesnil JG, Senechal M. Preoperative posterior leaflet angle accurately predicts outcome after restrictive mitral valve annuloplasty for ischemic mitral regurgitation. Circulation 115 (2007): 782-791.
- Cohn LH, Rizzo RJ, Adams DH, Couper GS, Sullivan TE, et al. The effects of pathophysiology on the surgical treatment of ischemic mitral regurgitation: operative and late risk of repair versus replacement. Eur J Cardiothorac Surg 9 (1995): 568-574.
- Tahta SA, Oury JH, Maxwell JM, Hiro SP, Duran CM. Outcome after mitral valve repair for functional ischemic mitral regurgitation. J Heart Valve Dis 11 (2002): 11-18.