Metastatic Dermatofibrosarcoma Protuberans in a Young African Lady: A Case Report
Article Information
Motsepe TA*, 1, Maritz SM 2
1Registrar, Department of General Surgery, Sefako Makgatho Health Sciences University & Dr. George Mukhari Academic Hospital, Pretoria.
2Medical Officer, Clinical Research, Sefako Makgatho Health Sciences University & Dr. George Mukhari Academic Hospital, Pretoria
*Corresponding author: Tshepang Arthur Motsepe.
Received: 16 October 2023; Accepted: 26 October 2023; Published: 08 November 2023
Citation: Motsepe TA, Maritz SM. Metastatic Dermatofibrosarcoma Protuberans in a Young African Lady: A Case Report. Fortune Journal of Health Sciences. 6 (2023): 446-449
Share at FacebookAbstract
Dermatofibrosarcoma protuberans (DFSP) is a rare tumour that originates from the dermal fibroblasts. It is often a benign lesion with a low metastatic potential. However, if left untreated, it can become locally aggressive and infiltrate surrounding fascia, muscles and bones. Distant metastases, though uncommon, are usually to the lungs with lymph node involvement being extremely rare. Below is a case of a young African lady who at presentation had a locally advanced DFSP with extensive metastases to distant lymph nodes and the lungs.
Contribution: The overall prognosis of DFSP is favourable with an excellent cure rate post surgical excision. However, with locally advanced tumours and with distant organ metastases cure becomes a non-feasible option. Therefore early diagnosis before tumour growth acceleration is key to a satisfactory patient outcome.
Keywords
dermatofibrosarcoma protuberans, metastasis, fibrosarcomatous, soft tissue tumour, fibroblast
dermatofibrosarcoma protuberans articles, metastasis articles, fibrosarcomatous articles, soft tissue tumour articles, fibroblast articles
Article Details
1. Introduction
Dermatofibrosarcoma protuberans (DFSP) is a tumour that is as rare as the literature surrounding the phenomenon. It was first described by Taylor in 1890 [1], clinically classified by Darier and Ferrand in 1924 [2] and later named by Hoffman in 1925 [3]. DFSP has an incidence rate of 0.8 to 4.5 cases per million persons annually and accounts for 1-6% of all soft tissue sarcomas. Although reported in all age groups, it presents mostly in the third to fifth decades of life [4]. DFSP is a slow growing, low- to intermediate grade malignant sarcoma [5] that originates from dermal fibroblasts [6].
It is known for its tenacious growth with asymmetrical, finger-like projections on histology. Its aggressive behaviour can be appreciated locally where it spreads within the dermis and subcutaneous tissues. If longstanding or recurrent, it can spread into the fascia, muscles and eventually bones [4, 6, 7]. DFSP has a high local recurrence rate but its metastatic potential remains low. Metastasis is involved in 1-4% of the cases, with the lungs being the most common site via a haematogenous spread. Regional lymph node involvement remains very uncommon [4].
2. Case Report
A 23 year old African lady presented at our surgical outpatient department with a 3 months history of new onset swelling of the left extremity that had been occasionally painful. She had noticed significant loss of weight since the leg swelling started. She did not have any co-morbidities, including human immunodeficiency virus (HIV) or tuberculosis. She reported sober habits and was a non-smoker. She did not use any contraceptive medication and had no recent history of prolonged air travel or rest. No family history of malignant or congenital conditions was noted.
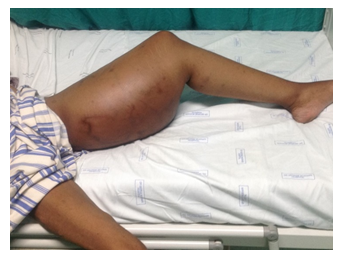
The patient had normal haemodynamic parameters. She looked chronically ill and cachectic with significant generalised lymphadenopathy. She had 2 cm x 2 cm soft subcutaneous lumps diffusely distributed throughout the upper limbs. Oro-oesophageal candidiasis was observed on general assessment. The entire left leg appeared massively swollen with a subcutaneous poorly circumscribed tumour at the posterior thigh. The size of this mass was about 20 cm x 35 cm. The skin was stretched with dilated visible tortuous veins above the area of the tumour. The left limb felt warm and full compared to the right.
The mass was firm to touch, however all the pulses were palpable and normal. The neurological assessment of the limb was unremarkable. She had difficulty walking due to the weight and size of the leg. She had a mildly laboured breathing with decreased sounds at the left lung base. All other systemic examinations were unremarkable. Differential diagnoses included neoplasms (e.g. soft tissue sarcomas, osteosarcomas and lymphomas), infective processes (e.g. massive abscess collection), and vascular disorders (e.g. lymphoedema and deep vein thrombosis).
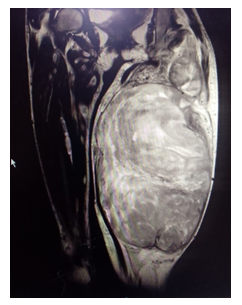
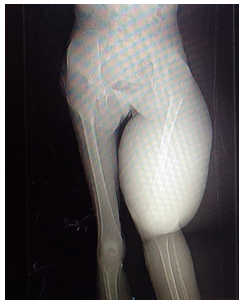
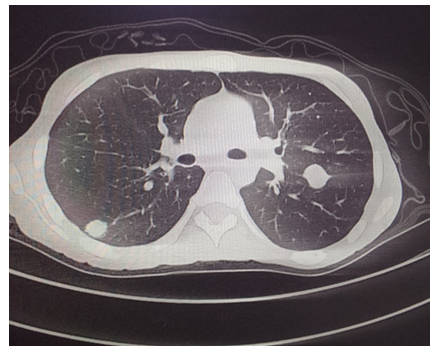
The patient’s biochemistry results showed elevated lactate dehydrogenase of 785 U/L and C-reactive protein of 100 mg/L with low serum albumin levels of 24 g/L. Peripheral smear demonstrated atypical lymphocytes with mild platelet clumping. Doppler ultrasound excluded a DVT. It indicated sluggish flow and mild reflux within the deep and superficial venous systems of the left lower limb. A chest radiograph showed multiple cannon ball lesions in the left lung field. Computed tomography (CT) scan revealed an oval shaped well delineated mass in the posterior left thigh extending from the ischium to the popliteal fossa. It had a necrotic central core with a peripheral solid consistency abutting the surrounding muscles. No bony infiltration was noted. Multiple canon ball lesions were noted in both lungs with lymphadenopathy in the bilateral cervical, axillary, iliac and inguinal regions as well as in the mediastinum. Significant hepatomegaly was noted. On magnetic resonance imaging (MRI), the mass showed low T1 and high T2 signals. Histopathology results demonstrated a tumour with features of DFSP. The patient had unresectable DFSP that had metastasised to the lungs and distant lymph nodes. Tumour showed minimal response to palliative radiotherapy, and patient demised two months post hospital discharge.
3. Discussion
DFSP is a rare tumour that accounts for less than 0.1% of all primary malignancies and only 1-6% of all soft-tissue sarcomas. This cutaneous neoplasm is known to be a slow and locally infiltrative growth that tends to recur years after surgical excision, however it seldom metastasise [8]. It carries up to 4% chance of distal haematogenous spread to vital organs, with the lungs being the most affected.4 Dissemination of DFSP to regional lymph nodes was reported in 6 out of 402 cases in an article by W. Brenner and colleagues [9]. In a study by K. Hayakawa and colleagues, a higher likelihood of metastasis and a poor prognosis was associated with fibrosarcomatous transformation in DFSP (FS-DFSP) and larger primary tumour size. FS-DFSP is an aggressive variant of DFSP associated with a predilection for metastasis and has a grave prognosis. In fact, according to Gloster et al., an ordinary DFSP seldom metastasise (likelihood of <1%) while FS DFSP has metastasis rate of up to 33%. The ordinary DFSP usually has a size of 2-5 cm, and this correlates to lower chances of it metastasising. In the same study by K. Hayakawa, four (80%) of the five cases that had distant metastases were 10 cm or larger and all were FS DFSP [10]. Our patient had a left thigh tumour measuring 20 cm x 35 cm, spanning the entire femur length and causing significant valgus deformity due to its weight. Though not formally reported on histopathology results, the patient probably had FS DFSP as this tumour had spread to lungs and lymph nodes. Multiple subcutaneous nodules (tumour deposits) were also prominent on the upper extremities.
The mainstay of treatment of DFSP is surgical excision of the tumour with either Mohs micrographic surgery (MMS) or wide local excision with tumour free margins. Though wide local excision was historically the procedure of choice it has somewhat lost popularity over the years as DFSP frequently has an unpredictable subclinical tissue penetration depth. In situation where MMS cannot be done, wide local excision can still be performed with 2 to 4 cm margins [4]. Partial or complete amputation of the digit may be performed for tumours that are located on the fingers or on toes [11]. Inoperable disease includes advanced stage tumours, recurrent disease where surgery is contraindicated due to tumour size and location, where resection would lead to significant functional and cosmetic defects, and distant organ metastasis. Under any of these conditions, non-surgical management with target therapy or/and radiation may take precedence with the aim of palliation [11].
Recurrent, unresectable or metastatic DFSP can be managed with target therapy. Imatinib mesylate (IM), a specific tyrosine kinase inhibitor, works by competitive inhibition of ATP binding to the PDGF-beta receptor. This leads to limitation of the kinase activity, suppression of tumour growth and ultimately tumour cell death. DFSP is a radio-sensitive tumour as adjuvant radiotherapy is effective at controlling the tumour growth and preventing its local recurrence. Sixty Gy are used for indeterminate or microscopic positive margins and up to seventy Gy can be used for macroscopic or gross disease. Radiation field should cover at least 3-5 cm outside the tumour borders [4]. Our patient showed modest response despite two cycles of radiation therapy. Due to the extent of cancer dissemination and the bulk of local tumour, cure was not possible to achieve. Whether target therapy would have reduced the disease load and afforded her a better quality of life is unknown as IM is currently not available in our institution.
4. Conclusion
In general, the overall prognosis of DFSP is good with a 10 year survival rate nearly 100%. However, with locally advanced tumours and distant organ metastases the coin flips to the other side; cure becomes a non-feasible option and palliation takes priority. The solution to this problem therefore partly lies in patient awareness and encouraging health-seeking attitude in the community. A small lump on the thigh or back should not be assumed to be an indolent keloid but should be raised with the primary care physician for investigation and possible biopsy early. Early diagnosis of DFSP before tumour growth acceleration is key to a satisfactory patient outcome.
Acknowledgements
Matlou Mabitsela and Branny Mthelebofu, medical writers at Sefako Makgatho Health Sciences University, are acknowledged for the technical and editorial support during the preparation of the manuscript.
Competing interests
The authors declare that they have no personal or financial interests that would compromise them in writing this article.
Author’s contribution
The authors contributed equally in the writing of this article.
Ethical considerations
Verbal and written consent was obtained from the patient, including consent for the use of body pictures, images and blood results. Ethics clearance was received from Sefako Makgatho University Research Ethics Committee (SMUREC) with the reference number: SMUREC/M/69/2023: (J) Journal.
Funding
No external funding was received.
Data availability
Patient confidentiality was upheld throughout as no personal information was shared with the public or third party.
Disclaimer
The expressed views and opinions are those of the authors and do not represent their affiliated institutions.
References
- Taylor RW. Sarcomatous tumors resembling in some respects keloid. J Cutan Genito-Urin Dis 8 (1890): 384-7.
- Darier S, Ferrand M. Dermatofibrosarcomes prograssifs at ricidivantes on fibrosarcomes de la peau (Dermatofibrosarcoma: A progressive and recurring rigid fibrosarcoma of the skin). Ann Dermatol Venereol 5 (1924): 545-62.
- Hoffman E. Ueber das knollentribende Fibrosarkom der Haut (Dermatofibrosarcoma protuberans). Dermatol Z 43 (1925): 1-28.
- Brooks J, Ramsey ML. Dermatofibrosarcoma protuberans. PubMed. Treasure Island (FL): StatPearls Publishing (2022).
- Hao X, Billings SD, Wu F, Stultz TW, Procop GW, Mirkin G, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med 9 (2020): 1752.
- Bague S, Folpe AL. Dermatofibrosarcoma protuberans presenting as a subcutaneous mass: A clinicopathological study of 15 cases with exclusive or near-exclusive subcutaneous involvement. Am J Dermatopathol 30 (2008): 327-32.
- Simon MP, Pedeutour F, Sirvent N, Grosgeorge J, Minoletti F, Coindre JM, et al. Deregulation of the platelet-derived growth factor β-chain gene via fusion with collagen gene COL1A1 in dermatofibrosarcoma protuberans and giant-cell fibroblastoma. Nature Genetics 15 (1997): 95 8.
- Paramythiotis D, Stavrou G, Panagiotou D, Petrakis G and Michalopoulos A. Dermatofibrosarcoma protuberans: a case report and review of the literature. Hippokratia 20 (2016): 80–83.
- Brenner W, Schaefler K, Chhabra H and Postel A. Dermatofibrosarcoma protuberans metastatic to a regional lymph node. Report of a case and review. Cancer 36 (1975): 1897-1902.
- Hayakawa K, Matsumoto S, Ae K, Tanizawa T, Gokita T, Funauchi Y et al. Risk factors for distant metastasis of Dermatofibrosarcoma protuberans. J Orthopaed Traumatol 17 (2016): 261-266.
- Hao X, Billings SD, Wu F, Stultz TW, Procop GW, Mirkin G, et al. Dermatofibrosarcoma protuberans: Update on the diagnosis and treatment. Journal of Clinical Medicine 9 (2020): 1752.