Medicinal Plants and Female Reproduction Disorders due to Oxidative Stress
Article Information
Deutcheu Nienga Sorelle, Ngoula Ferdinand, Vemo Bertin Narcisse, Tchoumboue
Department of Animal Production, Faculty of Agronomy and Agricultural Sciences, University of Dschang, Cameroon
*Corresponding Author: Deutcheu Nienga Sorelle, Department of Animal Production, Faculty of Agronomy and Agricultural Sciences, University of Dschang-Cameroon
Received: 17 November 2019; Accepted: 30 November 2019; Published: 31 Decrember 2019
Citation: Deutcheu Nienga Sorelle, Ngoula Ferdinand, Vemo Bertin Narcisse, Tchoumboue. Medicinal plants and female reproduction disorders due to oxidative stress. Archives of Veterinary Science and Medicine 2 (2019): 058-073.
Share at FacebookAbstract
Reactive oxygen species (ROS) are highly reactive oxidizing agents. Cells, under aerobic conditions, have a defence system against ROS, and in normal circumstances, there is an appropriate balance between pro?oxidants and antioxidants. When an overproduction of ROS develops or the body fails to eliminate ROS in excess, oxidative stress arises, during which ROS accumulate and damage cells and tissues. Besides their noxious effects, accumulating data have shown that controlled of adequate ROS concentrations exerts physiologic functions. Different studies have confirmed the presence of ROS and the transcripts of the various antioxidant enzymes in the female reproductive tract. When ROS production overwhelms antioxidant defenses, oxidative stress occurs, which may deeply threaten the anatomical and functional integrity of the genital tract. This review addresses the main physiological and pathological roles exerted by ROS and their scavenging systems in several processes involved in the main physiological functions of the female reproductive tract of domestic animals. Stress, of both physical and emotional origin, has effects on the reproductive system. Accordingly, impaired follicular development appears to be the most common cause of reproductive dysfunction attributable to stress in the human female. New developments in the understanding of the role of stress in reproduction must take into consideration the many differences between the hormonal responses to stress in the human and laboratory animals. Particularly, the involvement of the oxidant system in several reproductive processes is investigated, such as follicular development, ovarian steroidogenesis, ovulation, corpus luteum formation and function, luteolysis, germ cell function, maintenance of pregnancy and beginning of parturition. The use of medicinal plants has gained the world to solve health problems, notably reproductive disturbance. A large number of tropical plants and their extracts have shown ben
Keywords
Reproduction; Oxidative stress; Medicinal plants; Female
Reproduction articles, Oxidative stress articles, Medicinal plants articles, Female articles
Article Details
Deutcheu Nienga Sorelle, Ngoula Ferdinand, Vemo Bertin Narcisse, Tchoumboue
Department of Animal Production, Faculty of Agronomy and Agricultural Sciences, University of Dschang, Cameroon
*Corresponding Author: Deutcheu Nienga Sorelle, Department of Animal Production, Faculty of Agronomy and Agricultural Sciences, University of Dschang-Cameroon
Received: 17 November 2019; Accepted: 30 November 2019; Published: 31 Decrember 2019
1. Introduction
Reproduction is the essential function that allows species continuity and improves the productivity and perpetuates animal species [1]. Its perturbation or dysfunction leads to negative consequences on the animal productivity [2]. Many factors can be responsible for this perturbation for example stress (heat, feed, oxidative…), ageing, some drugs [3] and xenobiotics such as heavy metals. The infertility rate has increased tremendously in the past few decades [4], [5]. Environmental toxicants like heavy metal is one of the most widespread contaminants amongst the myriad of xenobiotic [6] which might be a major risk factor considering the hormonal disruption [7] associated with the reduction of sex organs weight and decrease of fertility [8]. Moreover, a reduction in fertilization and implantation rate [9] as well as mitotic inhibition and chromosome aberration [10] and decrease in reproductive performance [11], [12] were registered in experimental animals. As a matter of fact, one of the mechanisms by which they toxicans are effected is the over production of free radicals, whose excessive accumulation leads to oxidative stress and hence plays a major role in reproductive tissue damage [13]. This intoxication could generate oxidative stress [14-16] and thus reproduction impairment. Many studies have elucidated oxidative damage as the central mechanism of reproduction toxicity [17], [18]. Oxidative damage primarily occurs through excessive production of ROS that are generated during the reaction and react with biological molecules, eventually damaging membranes and other tissues [19], [20]. Many insecticides are hydrophobic molecules that bind extensively to biological membranes, especially phospholipids bilayers [21] and they may damage membranes by inducing lipid peroxidation [22], [20]. As mechanism of environmental toxicants involved oxidative stress, numerous efforts were done to identify natural compound able to strengthen the cellular antioxidant defense so as to counteract the reproductive stress. In this respect, medicines derived from plant extracts are being increasingly utilized to treat a wide variety of clinical diseases. More attention has been paid to the protective and therapeutic effects of natural antioxidants against ROS [23], [24]. The use of medicinal plants has gradually gained popularity [25] to solvehealth problems, notably reproductive disturbance. This is probably due to their low cost, great availability and lesser side effects [26], [27]. A large number of tropical plants and their extracts have shown beneficial therapeutic activities including fertility enhancement, antioxidant, anti-microbial and aphrodisiac activities [28]. Hence, plant extracts such as aqueous extract of green tea [29], guava leaves [30] and ethanolic extract of Bersama engleriana [31] have antioxidant properties. According to these authors, the administrations of these extracts to stressed animals significantly ameliorated the animal’s fertility and oxidative stress parameters.
2. Overview of the Female Reproduction Function
The female reproductive system functions to produce gametes and reproductive hormones, just like the male reproductive system; however, it also has the additional task of supporting the developing fetus and delivering it to the outside world. Unlike its male counterpart, the female reproductive system is located primarily inside the pelvic cavity [32], [33]. The organs are Ovaries, Fallopian tubes, Uterus (corpus + cervix), Lower genital tract (vagina and vulva) and Placenta while The functions are Conception Delivery of the baby and Synthesis of estrogen and progesterone (hormonal organ).
2.1. Ovary
The ovaries of all mammals have an identical basic structure. However, their overall appearance varies considerably by species, in the course of the ovarian cycle and the cycle period when the ovary is examined (1) Two of them are essential organs in sexual reproduction because it is at their level that there is development and differentiation of eggs and synthesis of certain reproductive hormones [34]. The ovaries provide a dual function; endocrine function (hormone production) and exocrine function (oogenesis and folliculogenesis) leading to oocyte production [35].
2.2. Uterus
The uterus, which is a gestational organ, follows the oviduct and has two parts, which are the uterine horns and the body [36]. It has a highly variable form from specie to other. The separation between the left and right uterus is more or less marked. In rabbits, rat and guinea pig, the division is complete (two horns, two bodies, two cervix). This uterus is duplex type that is to say double. There are in principle two independent uteri of about 7cm which open separately by two cervical ducts in the vagina [1]. The base connects to the uterine body and at this level the two horns lean against each other. The cervix or discernible cervix on the surface is usually more cylindroids than the body uterine and the great thickness of its wall makes it easy to recognize it with palpation. [37] showed a significant increase in the weight of the uterus and uterine proteins in rats fed 8 mg / kg aqueous extract of Senecio biafrae.
2.3. Vagina and Vulva
The vulva is the outer part of the reproductive tract, so it ends the genital tract. It is located immediately below the anus from which it is separated by the ano-vulvar bridge. The vulva is consisting of two pairs of lips: the big lips, covered with hair on their lateral face and the internal labia minor to the previous and finer [38]. Their appearance can change over time. In the rabbit, it is pale pink in normal times, but in sexual period receptivity, it will tend to swell and turn purple-red. It's in the vulva that opens the canal of the urethra and that is the clitoris [36], [38]. The vagina is a membranous canal extended horizontally in the pelvis between the rectum and the bladder and is in the vulva. It is the organ of female copulation and the exit channel of the small at birth [39]. It is very extensible because of the mucous folds that allow it to dilate, to receive the penis during mating and the passage of the fetus during parturition. The vestibule of the vagina, which corresponds to the common terminal path of the genital and urinary, is more or less long depending on the species.
2.4. Female Reproductive Functions: Hormones Productions, Conception and Delivery
2.4.1. Reproductive Hormones
Reproductive function is under the control of sex hormones. Several hormones ( FSH, LH, estradiol, Progesterone …) produced by the hypothalamic-pituitary complex and the sex organs come in to play in a precise and coordinated way in the control of folliculogenesis, oogenesis, the estrous cycle, sexual behavior and ovulation [40]. The synthesis and action of these hormones can be influenced by endogenous and/or exogenous factors. Stress may play an inhibitory role on Hypothalamus pituitary ovarian axis. Exogenous factors, in particular Plant extracts influence the serum sex hormone levels. [41] have showed a reduction in serum concentrations of estradiol and pituitary hormones in animals treated with the ethanolic extract of Spondias mombin while [42] found a significant increase in sex hormones (FSH, LH and estradiol) in animals receiving aqueous and ethanolic extracts of Tribulus terrestris, Phoenix dactylifera and Nasturtium officinale.
2.4.2. Conception and Delivery
The establishment of a gestation is one of the most complex improvements in evolution of mammals which involve the specialization of an organ, the uterus, for the conception and embryonic development [38]. This is the period during which the egg is develops into an individual. It goes from fertilization to parturition. Gestation in rabbits lasts 31-32 days [44], with variation observed by race and individuals ranging from 29 to 35 days while it is about 65 to 70 days in guinea pigs.
2.4.3. Fertility Traits, Litter Size, Weight, and Viability Rate
As a measure, fertility rate is the number of offspring born per mating pair, individual or population. Fertility differs from fecundity, which is defined as the potential for reproduction (influenced by gamete production, fertilization and carrying a pregnancy to term). A lack of fertility is infertility while a lack of fecundity would be called sterility. Fertility is generally used to indicate the actual reproductive performance of a female or groups of females. The crude birth rate (number of birth per 1000 population per year) is only one measure of fertility. The size of the litter depends on the breed, parity, reproductive fertility, age, season,viability and growth of the fetuses. The average litter size is from 3 to 12, with limits from 1 to 20 [36]. Small breeds tend to have litters of 4-5 young/litter, whereas large breeds can have an average litter size between 8 and 12 young. For parity, primiparous usually have the smallest litters [45], [46], [43]. The most important litter sare observed in the 2nd and 3rd gestation, and then their size decreases with the following gestations. Finally, for the season, it is during the warmest months that the smallest litter sizes are observed [45], [46], [43]. At birth, the weight of the rabbits is more or less variable within the same litter. Depending on the size of the litter, rabbit can weigh from 25 to 80 [47], [39].The changing weight of young animals from birth to weaning depends on their own growth potential and the mother's ability to breastfeed [48].
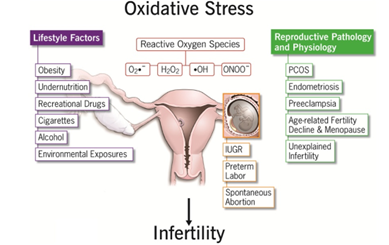
3. Female Oxidative Stress Linked Reproductive Disorders/ Dysfunctions (with Emphasis on those due to Environmental Toxicants)
3.1. Oxidative Stress
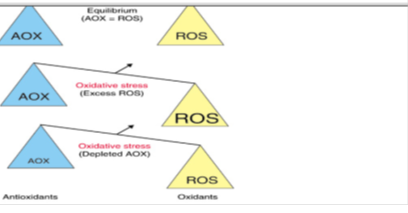
Oxidative stress is a state which results from a disequilibrium within an individual, between the production of oxidants and the defense mechanism of antioxidants [49], [50] in favour of oxidants. Its impact on the organism depends on the type of oxidant, site and intensity of its production, composition and activities of various antioxidants, and on the ability of repair systems [51]. This is evident in biological systems when there is either an excessive production of reactive oxygen species (ROS) and/or a deficiency of enzymatic and non-enzymatic antioxidants [52] (Figure 1).
Reactive oxygen species can be produced from either endogenous or exogenous sources. The
endogenous sources of ROS include different cellular organs such as mitochondria, peroxisomes and endoplasmic reticulum and phagocytosis where the oxygen consumption is high. Exogenous sources of ROS are air and water pollution, cigarette smoking, alcohol, certain drugs (cyclosporine, tacrolimus), industrial solvents, cooking gas, radiation and heavy metals. After penetration into the body, these exogenous compounds (eg. heavy metals) are decomposed into free radicals [51]. Heavy metals are natural metallic elements, metals or in certain cases metalloids, characterized by atomic number greater than 20 [53]. It has a relatively high density and is toxicat low quantity is referred to as ‘‘heavy metal’’ e.g. arsenic (As), lead (Pb), mercury (Hg), cadmium (Cd), chromium (Cr) etc. Some trace elements are also known as heavy metals e.g.copper (Cu), selenium (Se) and zinc (Zn). They are essential to maintain the body metabolism, but they are toxic at higher concentrations. Heavy metals can enter the body to a small extent viafood, drinking water and air [53]. The detrimental effects/toxicities associated with Arsenic, Lead, Mercury and Cadmium are shown on (Table 1).
|
Heavy metal |
Detrimental effect |
LD50 (oral) |
|
Arsenic (As) |
Very high concentrationcauses infertility |
10-300mg/kg |
|
Lead (Pb) |
Anaemia, reproductive problems |
5 μg/l |
|
Mercury (Hg) |
DNA and chromosomal damage, birth defects and miscarriages |
1 mg/kg |
|
Cadmium (Cd) |
Damage kidney, reproductive problems |
63-1125 mg/kg |
LD50: lethal dose 50
Table1: Detrimental effects/toxicities associated with Arsenic, Lead, Mercury and Cadmium [54].
3.2. Effects of Oxidative Stress on Female Reproductive Functions
Reactive oxygen species appear to have physiological role in female reproductive tract in many different processes such as: oocyte maturation, luteal regression, and endometrial shedding. It may affect the ovulation, fertilization, and implantation. ROS not only alters most types of cellular molecules but also induces early embryonic developmental block and retardation [55], embryonic mortality, fetal death, and low birth weight [56],cell membrane damage, DNA damage, and apoptosis [57], [55]. The harmful action of these oxidative stress can however be blocked by antioxidant (AO) substances which scavenge the free radicals and detoxify the organism [58].
The oral administration of lead (Pb) acetate at a dose of 2500ppm to rats [60] recorded a slight decrease in weight of ovaries+uterus (0.47 ± 0.12 g) in Pb-treated rats with respect to the control group (0.50 ± 0.10 g). Co-treatment with aqueous leaves extract of Costus afer (1150 mg/kg bw) led to a significant increase in weight of these organs (0.90 ± 0.35g). Another observation was noted by Dumitrescu [61] in the weights of the uterus and fallopian tube in rats exposed to lead. Oxidative stress appears to have a detrimental effect on the development of embryo. ROS may originate from embryo metabolism and from the surrounding environment. They not only altermost types of cellular molecules but also induces early embryonic developmental block andretardation [55]. High levels of ROS and apoptosis were reported in fragmented embryos compared to non-fragmented embryos [59]. The oral administration of lead (Pb) acetate at a dose of 2500ppm to rats [60] recorded a slight decrease in weight of ovaries+uterus (0.47 ± 0.12 g) in Pb-treated rats with respect to the control group (0.50 ± 0.10 g). Morphometric analysis of the cadmium (25.00 μg/g bw) treated mice indicated a significant
reduction in fetal body weight (812.8 ± 119.82 mg), crown rump length (14.85 ± 2.48 mm), head circumference (9.08 ± 1.97 mm), forelimb (5.29 ± 2.16 mm) and hind limb lengths (5.59 ± 1.23 mm) and tail size (4.16 ± 0.89 mm) compared to control fetuses (1277.24 ± 74.25 mg, 27.95 ± 2.22 mm, 23.48 ± 1.30 mm, 6.67 ± 0.50 mm, 7.29 ± 0.80 mm, 7.33 ± 1.26 mm), respectively. A, vehicle control; B, from antidote +6.25 μg/g bw; C, from antidote + 2.5 μg/g BW; D, from antidote +25.0μg/g bw. Oxidative stress appears to have a detrimental effect on the development of embryo. ROS may originate from embryo metabolism and from the surrounding environment. They not only alter most types of cellular molecules but also induce early embryonic developmental block and delay [55]. High levels of ROS and apoptosis were reported in fragmented embryos compared to non-fragmented embryos. Embryonic mortality represents the death of the product of conception during the embryonic period of gestation. Based on the work of [36], the majority of embryonic mortalities occur between 0 and 15 days of gestation. These side effects of many heavy metals can occur in high doses close to the toxic level for the dam or the pregnant animal. Evidence of the harmful effects of lead on the organism in general and reproduction in particular have been widely studied [61]. The exposure of rodents to lead increased the tendency to fetal death, growth retardation, resorption and congenital malformations [60]. Induction of oxidative stress is one of the mechanisms implicated in lead induced fetal-toxicity [56].
4. Potential (Preventive/ Therapeutic) Effects of Medicinal Plants
Apart from animal organisms, plants have several types of antioxidants, among which it has been widely proven that they contain a high concentration of polyphenols, alkaloids, flavonoids or triterpenes, tannins, that act as antioxidants both in vitro and in vivo (in animal and human cells), thus reducing and controlling ROS molecules. Phenolic compounds are known as powerful chain breaking antioxidants. Halliwell and Gutteridge [62] defined antioxidants as “any substance that, when present at low concentrations compared with that of an oxidizable substrate, significantly delays or inhibits oxidation of that substrate”, but later defined them as “any substance that delays, prevents or removes oxidative damage to a target molecule” [62]. In the same year [63] defined antioxidants as “any substance that directly scavenges ROS or indirectly actsto up-regulate antioxidant defenses or inhibit ROS production”. Another property that a compound should have to be considered an antioxidant is the ability, after scavenging the radical, to form a new radical that is stable through intra-molecular hydrogen bonding on further oxidation [64]. They are present in two types, enzymatic and non-enzymatic forms. Enzymatic antioxidants, including superoxide dismutase (SOD), catalase (CAT), glutathione (GSH) peroxidase and glutathione (GSH) reductase are also known as natural antioxidants orendogenous antioxidants [56]. The non-enzymatic antioxidants, also known as exogenous antioxidants, are obtained from dietary fruits and vegetables. These include taurine, hypotaurine, carotene, selenium, zinc, vitamin C and vitamin E [59]. Otherforms of exogenous antioxidants are ascorbic acid or vitamin C, Vitamin E (α-tocopherol),Vitamin K, flavonoids (flavonols, flavanols, anthocyanins, isoflavonoids, flavanones andflavones), phenolic acids, Beta-carotene, minerals and synthetic antioxidants suc as butylated hydroxyanisole (BHA) and buty lated hydroxyl- toluene (BHT) [52], [56], [64], [65]. Phenols are very important plant constituents because of their scavenging ability due to their hydroxyl groups [51]. It has been recognized that flavonoids have antioxidant activity and their effects on human nutrition and health are considerable. The mechanisms of action of flavonoids are through scavenging processes. These substances contain hydroxyl functional groups, responsible for antioxidant effect in plants. Some other very useful medicinal plants include green tea, Spirulina platensis, Spirulina maxima and Crassocephalum bauchiense [66], [58]. Also the antimicrobial and antioxidant activity of some species of the Spirulina genus are well established [67]. The administrations of some plant extracts have been reported to either counteract the environmental toxicity concerning the reproductive function or to simply have positive effects on this latter. This is because such plants contain compounds like steroids, saponins and phenols which have androgenic properties [68]. Plants constitute the main source of natural AO molecules, which have the capacity to eliminate or neutralize the deleterious ROS [66].
4.1. Effects of Medicinal Plants on Reproductive Hormones Associated to Oxidative Stress
The oral administration of lead (Pb) actate at a dose of 2500 ppm to rats [60] and co-treatment with aqueous leave extract of Costus afer (2250 mg/kg bw) resulted in asignificant decrease in LH (3.13 ± 4.04 mIU/ml) and FSH (3.50 ± 1.57 mIU/ml) of Pb-treated group compared with those co-exposed to lead and extract of C. afer(2.43 ± 0.25 mIU/ml and2.90 ± 0.40 mIU/ml respectively).). Flavonoids and phenols are thought to be involved in increasing serum sex hormone levels by inhibiting cyclooxygenase-2, the enzyme responsible for inhibiting the StAr gene and thus steroidogenesis .
4.2. Effects of Medicinal Plants on Fertility Traits, Litter Size, Weight and Viability Rate
Raji et al. [69] found a high rate of live born rabbits in animals that received 200 and 400
mg/kg bw of the aqueous garlic extract (Allium sativum) compared to the control group. In
another study, Kareema and Abdul-Hadi [70] orally treated adult mice with 500 and 1000 ppmof potassium dichromate dissolved in tap water for 10, 20, and 30 days. The findings showed that this compound induced a significant decrease in values of some reproductive parameters including corpora lutea and thickness of uterine layers. Also, the oral administration of female mice with chromium for 30 days resulted in a significant reduction in the number of pregnant females and the number and weight of litters. Overall, the study proved that this compound has a potential toxicity in female reproductive system and fertility of albino mice. Al-neamah, [71] observed an increase in implantation sites and the number of pups in female cadmium poisoning rats receiving hydro alcoholic extracts from Zingiber officinale. The hydro-alcoholic extracts of Zingiber officinale would effectively increase reproduction in female rats that were poisoned with cadmium [71]. Ainehchi and Zahedi, [72] showed a significant increase in the fertilization rate in females receiving 200 and 400 mg / kg of hydro-alcoholic extract of Artemisia lanata. Karacat and Simsek, [73] observed an improvement in ovarian weight and structure in lead exposed females receiving Spirulina platensis extract. The improvement of these parameters could be attributed to the androgenic properties of the compounds (phenols and flavonoids) revealed by our phytochemical tests [74].
4.3. Effects of Medicinal Plants on Fetal Toxicity Associated to Oxidactive Stress
Fetuses treated with cadmium chloride along with guava fruit extract revealed ameliorative effects of these birth defects with respective values of (886.20 ± 125.96 mg, 17.19 ± 3.15 mm, 10.80 ± 0.76 mm, 5.45 ± 2.12 mm, 6.85 ± 1.46 mm and 6.69 ± 1.22 mm). This indicates that guava fruit extract exerted a therapeutic effect on developmental anomalies induced by cadmium exposure in prenatal mice (Figure 2). Sharma et al. [75] reported a significant decrease in individual pups weight of mice exposed to lead (1.00 gm) as compared to control (1.78 gm) and those administered lead along with vitamin E recorded improved weight (1.22 gm). Increased number of resorptions and growth retardation in pregnant mice given different doses of cadmium were reported by [74]. Distorted limbs were observed by the [77] in fetus of pregnant mice injected with 4mg/kg cadmium on days 8-10 of gestation.
A, vehicle control; B, from antidote +6.25 μg/g bw; C, from antidote+ 12.5 μg/g BW; D, from antidote+ 25.0μg/g bw. A, control skeleton showing well ossified skeleton; B, from 6.25 μg/g BW; C, from 12.5 μg/g BW; D, from 25.0 μg/g bw. Cl, Clavical; Eo, Exoccipital; Fe, Femur; Fi, Fibula; F, Frontal; Ri, Ribs; U, Ulna; N, Nasal; Pm, Premaxilla; Z, Zygomatic; H,Humerus; So, Supraoccipital; Ip, Interparital; P, Parietal; R, Radius; Ro, Reduced ossification; Uo, Unossifie.
5. Critical Assessment
The stability of reproductive cells and tissues is dependent on balanced concentrations of antioxidants and oxidants varied levels of ROS can have both positive and negative impacts on female reproduction. The excess production of free radicals and subsequent induction of OS, however, have long been known to significantly affect reproductive functions. Reactive oxygen and nitrogen species can negatively affect embryo implantation and
may influence the development of reproductive disorders such as endometriosis and preeclampsia. These effects have been reported to improve with the aid of antioxidants, and thus could minimize the associated risk for infertility. The medicinal plants gained popularity to solve health problems, notably reproductive disturbance. This is probably due to their low cost, great availability and lesser side effects [78], [79]. A large number of tropical plants and their extracts have shown beneficial therapeutic activities
including fertility enhancement, antioxidant, anti-microbial and aphrodisiac activities. That is the effects on animal reproduction of aqueous extracts of these plants, for example wing (Allium sativum) extracts [69], leaves of Tribulus terrestris. Phoenix dactylifera and Nasturtium officinale (80), essential oils of Syzygium aromaticum [81], leaves of Momordica charantia [82] and guava leaves [30] were tested. Results obtained were interesting for some and less important for other researches.
6. Conclusion
Reproduction is the essential function that allows species continuity. Fertility is generally used to indicate the actual reproductive performance of a female or groups
of females. Its dysfunction leads to negative consequences on the animal productivity. Many factors can be responsible for this perturbation namely stress (heat, feed, oxidative), ageing, some drugs and xenobiotics such as heavy metals. One of the main
mechanisms by which these factors induce low reproduction is oxidative stress. Oxidative stress can be responsible for this perturbation. This intoxication could generate oxidative and thus reproduction impairment. The harmful action of these oxidative stress can however be blocked by antioxidant substances which scavenge the free radicals and detoxify the organism. The use of medicinal plants has gradually gained popularity to solve health problems, notably reproductive disturbance. This is probably due to their low cost, great availability and lesser side effects. A large number of tropical plants and their extracts have shown beneficial therapeutic activities including fertility enhancement, antioxidant, anti-microbial and aphrodisiac activities.
References
- Gayrard V. Physiologie de la reproduction des mammifères. Ecole Nationale Vétérinaire Toulouse I (2007): 198.
- Tamboura HH, Bayala B, Pellicer MT, et al. Effets ostrogéniques du macéré aqueux des feuilles de Holarrhena floribunda (G. Don) chez la rate ovariectomisée. Base 10 (2006): 173-180.
- Hafez ES, Hafez S D. Erectile dysfunction: anatomical parameters, etiology, diagnosis,and therapy, Archives of Andrology 51 (2005): 15-31.
- Oehninger S. Strategies for the infertile man. Semin Reprod Med 19 (2001): 231.
- Venkatesh S, Deecaraman M, Kumar R, Shamsi BM. Role of reactive oxygen species in the pathogenesis of mitochondrial DNA (mtDNA) mutations in male infertility. Indian J Med Res 129 (2009): 127-137.
- Mateo R,. Lead poisoning in wild birds in Europe and the regulations adopted by different countries. In: Watson, R.T., Fuller, M., Pokras, M., Hunt,W.G. (Eds.), Ingestion of Lead from Spent Ammunition: Implications for Wildlife and Humans. The Peregrine Fund, Boise, Idaho (2009): 71-98.
- Sarkar M, Chaudhuri GR, chattopadhayay A, et al. Effect of sodium arsenita on spermatogenesis, plasma gonadotrophins and testosterone in rats. Asian J. Andro 1 (2003): 27-31.
- Ait HN, Slimani M, Aous AEK. Reproductive toxicity of lead acetate in albinos’ rat. American journal of scientific research (2009): 38-50.
- Cinar O, Simez O, Can A. Carbofuran alters centrosome and spindle organization, and delays cell division in oocytes and mitotic cells. Toxicolocal Science 28 (2015): 437-444.
- Chauhan LK, Kumar M, Paul BN, et al Cytogenetic effects of commercial formulations of Deltamethrin and/or Isoproturon on human peripheral lymphocytes and mouse bone marrow cells. Environmental and Molecular Mutagenesis 48 (2007): 636-643.
- Kenfack A, Tchoumboué J, Kamtchouing P, et al. Influence du chlorpiryphos-éthyle (pesticide agricole) sur la croissance etle fonctionnement de l’appareil reproducteur mâle chez le rat wistar. Biosciences Proceedings 11 (2005): 250-257.
- Ngoula F, Watcho P, Dongmo MC, et al. Effects of pirimiphos-methyl (an organophosphate insecticide) on the fertility of adult male rats. African Health Sciences 7 (2007): 3-9.
- Sangha GK, Kaur K, Khera KS. Cypermethrin induced pathological and biochemical changes in reproductive organs of female rats. Journal of Environmental Biology, 34 (2013): 99-105.
- Zhang JJ, Wang Y, Xiang H, et al. Oxidative stress: Role in acetamiprid induced impairment of the male mice reproductive system. Agricultural Sciences in China 10 (2010): 786-796.
- Doltade S, Lonare M, Raut S et al. Evaluation of Acetamiprid Mediated Oxidative Stress and Pathological Changes in Male Rats: Ameliorative Effect of Curcumin. Proceedings of the National Academy of Sciences, India - Section B: Biological Sciences (2017: 1-9.
- Keshta AT, Hataba AA, Mead HMI et al. Oxidative stress and biochemical changes induced by thiamethoxam and acetamiprid insecticides in rats. World Journal of Pharmacy and Pharmaceutical Science 5 (2016): 44-60.
- Verma RS, Mehta A, Srivastava N. In vivo chlorpyrifos induced oxidative stress: Attenuation by antioxidant vitamins. PesticBiochem Phys 88 (2007): 191-196.
- Amin KA, Hashem KS. Deltamethrin-induced oxidative stress and biochemical changes in tissues and blood of catfish (Clariasgariepinus): antioxidant defense and roleof alpha-tocopherol. Veterinary Research 8 (2012): 1-8.
- Kalender Y, Kaya S, Durak D, Uzun FG, Demir F. 2010. Protective effects of catechin and quercetin on antioxidant status, lipid peroxidation and testis-histoarchitecture induced by chlorpyrifos in male rats. Environ Toxicol Pharmacol 3 (2010): 141-148.
- Heikal TM, Mossa TH, Marei GIKH, et al. Cyromazine and chlorpyrifos induced renal toxicity in rats: the ameliorating effects of green tea extract. Journal Environmental Analytical Toxicology, 2 (2012): 146-152.
- Ogutcu A, Suludere Z, Kalender Y. Dichlorvos-induced hepatotoxicity in ratsand the protective effects of vitamin C and E. Environ ToxicolPharmacol 26 (2008): 355-361.
- Mossa AH, Abbassy MA. Haemato-biochemical effects of formulated and technical cypermethrin and deltamethrin insecticides in male rat. Journal of Pharmacology and Toxicology 7 (2012): 312-321.
- Ikpeme EV, Okonko LE, Udensi OU. Detrimental effects of chlorpyriphos andcypermethrin on reproductive physiology of male albino rats. Research Journal of Environmental Toxicology, 10 (2016): 68-74.
- Molla MR, Rahman MM, Akter F, et al. Effects of Nishyinda, black pepper and cinnamon extract as growth promoter in broilers. The Bangladesh Vet 29 (2012): 69-77.
- Neelesh M, Sanjay J, Bihari GV. et al. Recent studies on aphrodisiac herbs for the management of male sexual dysfunction. A review. ActaPoloniae Pharmaceutica-Drug Research 68 (2011): 3-8.
- Dahanukar SA, Kulkarni RA, Rege NN. Pharmacology of medicinal plants and natural products. Indian Journal of Pharmacology 32 (2000): 81-118.
- Morsi RMY, EL-Tahan NR, El-Hadad AMA. Effect of aqueous extract Mangiferaindicaleaves, as functional foods. Journal of Applied Sciences Research 6 (2010): 712-721.
- Petrovska BB. Historical review of medicinal plants’ usage. Pharmacognosy Review 6 (2012): 1-5.
- Abshenas J, Homayoon B, Zare MH, et al. The effects of green tea (Camellia sinensis) extract on mouse semen quality after scrotal heat stress.Veterinary Research Forum 2 (2011): 242-247.
- Ngoula F, Nouboudem SC, Kenfack Aet al. Effect of guava (Psidium guajava) leaves essential oil on some reproductive parameters in male guinea pig (Cavia porcellus).Biological systems 3 (2014) : 1-4.
- Vemo BN, Kenfack A, Ngoula F, et alEffects of ethanol extract of Bersama engleriana leaves on oxidative stress and reproductive parameters in male Guinea pig (Cavia porcellus) exposed to cypermethrin. International Journal of Biological and Chemical Sciences 11 (2017): 2243- 2253.
- Whittier JC. Reproductive Anatomy and Physiology of the Bull (1993).
- Turner J. Reproductive Tract Anatomy and Physiology of the cow (2014).
- Othmani-Mecif K et Benazzoug Y. Caractéristiques de certains paramètresbiochimiques et histologiques (tractus génital femelle) chez la population locale de lapin (Oryctolaguscuniculus) non gestante et au cours de la gestation. Sciences et technologies 23 (2005): 91-96.
- Thibault C, Beaumont A et Levasseur MC. La reproduction des vertèbres. Masson, Paris (1998): 307.
- Lebas F, Courdet P, Rouvier R et Rochambeau H . The rabbit: husbandry, health and production. Food and Agricultural Organization of the United Nation, Rome.N° 21 (1997): 209.
- Lienou LL, Telefo BP, Bale B, et al. Effect of the aqueous extract of Senecio biafrae on sexual maturation of immature, female rat (2012).
- Gayrard V. Physiologie de la reproduction des mammifères. Ecole nationale de Toulouse 198 (2007): 119-151.
- Salissard M. La lapine, une espèce à ovulation provoquée. Mécanismes et dysfonctionnement associé: la pseudo-gestation. Thèse d'exercice, Médecine vétérinaire, Ecole Nationale Vétérinaire de Toulouse-ENVT (2013): 102.
- Boiti C, Besenfelder U, Brecchia G, et al. Reproductive physiology of the rabbit does in recent advance. Rabbit science (2006): 12-19.
- Ashok Agarwal, Anamar Aponte-Mellado, Beena J Premkumar, et al. The effects of oxidative stress on female reproduction: are view. Reproductive Biology and Endocrinology, (2012).
- Adaay A, Mosa AR. Evaluation of the effect of aqueous extract of Tribulusterrestris on some reproductive parameters in female mice. Journal of Environemental Science 3 (2012): 1153-1162.
- Lebas F. La Biologie du Lapin. http://www.cuniculture.info/Docs/Biologie/Biologie- 50.htm (2011).
- Cheeke PR, Patton NM, Lukefahr SD, et al Rabbit Production. Danville: Interstate Printers and Publishers (1987): 472.
- Donnelly TM. Rabbit: Basic Anatomy, Physiology and Husbandry. In Ferret Rabbits and Rodents: Clinical Medicine and Surgery.2nd edition. Philadelphia Saunders: (2004): 136-146.
- Chavatte-Palmer P, Laigre P, Simonof FE, et al. Caractérisation de la croissance fœtale in utero par échographie chez la lapine. In 1e Journées de la Recherche Cunicole Paris (2005): 83-86.
- Berchiche M, Cherfaoui D, Lounaouci G, et al. Utilisation de lapins de population locale en élevage rationnel: Aperçu des performances de reproduction et de croissance en Algérie. 3ème Congres Franco-Maghrébin de Zoologie et d’Ichtyologie, Marrakech, Maroc, (2012): 42.
- Garreau H, Rochambeau H. La sélection des qualités maternelles pour la croissance du lapereau. 10èmes Journées de la Recherche Cunicole. INRA-ITAVI, Paris, ITAVI, Ed. Paris, (2003): 61-64.
- Morena M, Martin-Mateo M, Cristol J.-P, et al. Stress oxydant, hémoincompatibilité et complications au cours de la dialyse. Néphrologie 23 (2002): 201-208.
- Pastre JO. Intérêt de la supplémentassions en antioxydants dans l’alimentation des carnivores domestiques. Thèse de doctorat vétérinaire. Ecole Nationale Vétérinaire de Toulouse, (2005): 120.
- Sivo?ová Z, Tatarková Z, ?ura?ková D, et al. Relationship between antioxidant potential and oxidative damage to lipids, proteins and DNA in aged rats. Physiological Research 56 (2007): 757-764.
- Ahmadi MF Oxidative Stress and Antioxidants. Review Article 1 (2011): 1-11.
- Heavy metals, Home process (2012): 1-12.
- Pandey G, Madhuri S. Heavy Metals Causing Toxicity in Animals and Fishes. Research Journal of Animal, Veterinary and Fishery Sciences 2 (2014): 17-23.
- Guerin P, Mouatassim SE, Menezo Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Article Human Reproduction 7 (2001): 175-189.
- Agarwal A, Gupta S, Sikka S. The role of free radicals and antioxidants inreproduction. Current opinion in obstetrics and gynecology 18 (2006): 325-332.
- Jurisicova A, Varmuza S, Casper RF. Programmed cell death and human embryo fragmentation. Molecular Human Reproduction 2 (1996): 93-98.
- Mouokeu RS, Ngono RAN, Lunga PK, et al. Antibacterial and dermal toxicological profiles of ethyl acetate extract from Crassocephalum bauchiense (Hutch.) Milne-Redh (Asteraceae). BMC Complementary and Alternative Medicine 11 (2011): 1-7.
- Agarwal A, Aponte-Mellado A, Premkumar BJ, et al Theeffects of oxidative stress on female reproduction: a review. Reproductive Biology and Endocrinology 10 (2012): 49.
- Ezejiofor AN, Orisakwe OE. Evaluation of protective effect of aqueous leave extract of Costus afer on female albino Wistar rats exposed to lead acetate. EC pharmacology and toxicology 4 (2017): 75-92.
- Dumitrescu E. The consequences of chronic lead acetate intake on exposure and morphological integrity biomarkers (lead level and weight of sexual organs) in female. Lucr?ri $Tiin?ifice Medicin? Veterinar? 41 (2008): 619-622.
- Halliwell B, Gutteridge JMC. Free radicals in biology and medicine, Oxford; Oxford University Press 4 (2007): 704.
- Khlebnikov AI, Schepetkin IA, Domina NG, et al. Improved quantitative structure–activity relationship models to predict antioxidant activity of flavonoids in chemical, enzymatic, and cellular systems. Bioorganic and medical Chemistry 15 (2007): 1749-1770.
- Halliwell B. Free radicals and antioxidants: A personnal view. Nutrition reviews 52 (1994): 253-65.
- Garait B. Le stress oxydant induit par voie métabolique (régimes alimentaires) ou parvoie gazeuse (hyperoxie) et effet de la Glisodin. Thèse pour obtenir le grade de docteur de l’Université Joseph Fourier-Grenoble I (2006): 198.
- Sguera S. Spirulina platensis and its constituents, nutritional benefits and therapeutic activities. Doctorate Thesis, University of Henri Poincare – Nancy 1 (2008): 24-100.
- Cruchot H. La Spiruline, Bilan et Perspective. Thèse de doctorat en pharmacie. Université de France-Comite, (2008).
- Ahangarpour A, Oroojan AA, Heydari H. Effect of hydro-alcoholic extract of Dorema aucheri on serum levels of testosterone, FSH and sperm count in nicotinamide-STZi duced diabetic rat models. Zanjan University of Medical Sciences Journal 21 (2013): 22-31.
- Raji LO, Fayemi OE, Ameen SA, et al. The effect of aqueous extract of Allium savium (garlic) on some Aspects of Reproduction in the female Albino Rat (Wistar Strain). Global Veneterinaria 8 (2012): 414-420.
- Kareema HD, Abdul-Hadi AH. Effects of Potassium Dichromate on Reproduction and Fertility in Albino Female Mice. International Journal of Pharmacological and Technical Research 9 (2016): 205-213.
- AL-Neamah AK. Protective effect of ginger (zingiber officinal) hydro alcoholic extract on cadmium chloride induced reproductive toxicity in rats female. Euphrates Journal of Agriculture Science 8 (2016): 8-16.
- Ainehchi N, Zahedi A Effects of Artemisia lanata extract on reproductiveparameters of female rats. Crescent journal of medical and biological sciences 1 (2014): 49-53.
- Karacat T, Simsek N. Effects of Spirulina on the number of ovary mast cell s in lead induced toxicity in rats. Phytother. Res 21 (2007): 44-46.
- Boutalbi S. Criblage chimique de l?activité biologique de spiruline ( Arthospira platensis) Thèse- université de Ouargla (2014): 82.
- Sharma R, Nazera Q, Sheetal M, et al. Lead induced infertility in Swiss mice and role of antioxidants. Universal Journal of Environmental Research and Technology 2 (2012): 72-82.
- Menai M, Heude B, Salama R, et al (2012). Association between maternal blood cadmium during pregnancy and birth weight and the risk of fetal growth restriction: The EDEN mother-child cohort study. Reproductive. Toxicology 34 (2012): 622-627.
- Messerle K, Webster WS. The classification and development of cadmium induced limb defects in mice. Teratology 25 (1982): 61-70.
- Dahanukar SA, Kulkarni RA, Rege NN. Pharmacology of medicinal plants and natural products. Indian Journal of Pharmacology 32 (2000): 81-118.
- Morsi RMY, EL-Tahan NR, El-Hadad AMA. Effect of aqueous extract Mangifera indica leaves, as functional foods. Journal of Applied Sciences Research 6 (2010).
- Adaay A. et Mattar AR. Evaluation of the effect of aqueous extract of Tribulus terrestris on some reproductive parameters in female mice. Journal of Environmental Science 3 (2012): 1153-1162.
- Farouk B, Abdelkrim B, Malika BS, et al Ameliorative Effects of Syzygium aromaticum Essential Oil on Fertility in Male Rats Exposed to Manganese. Advances in sexual medicine 3 (2013): 85-91.
- Osonuga OA, Osonuga IO, Osonuga A. Oral administration of leaf extracts of Momordica charantia affect reproductive hormones of adult female wistar rats. Asian Pacific Journal 4 (2014): 521-524.