Home Delivery of Antiretrovirals for HIV Clients amidst Covid-19 and Insecurity Challenges: A Protocol for a Randomized Controlled Trial
Article Information
Ajagu Nnenna1, 3*, Offu Ogochukwu3, Okengwu Ogadinma Daughter3, Ani Njieka Ifeoma2, Umeh Ifeoma Blessing1, Okoro Kenneth Obinna2, Nduka Sunday1, Ekwunife Ikechukwu Obinna1, 3
1Department of Clinical Pharmacy and Pharmacy Management, Faculty of Pharmaceutical Sciences, Nnamdi Azikiwe University, Awka, Nigeria
2Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, Enugu State University of Science and Technology, Agbani, Nigeria
3Department of Clinical Pharmacy and Biopharmaceutics, Faculty of Pharmaceutical Sciences, Enugu State University of Science and Technology, Agbani, Nigeria
*Corresponding Author: Ajagu Nnenna, Department of Clinical Pharmacy and Pharmacy Management, Nnamdi Azikiwe University, Awka, and Department of Clinical Pharmacy and Biopahrmaceutics, Enugu State University of Science and Technology, Agbani, Nigeria
Received: 29 November 2021; Accepted: 06 December 2021; Published: 13 January 2022
Citation:
Ajagu Nnenna, Offu Ogochukwu, Okengwu Ogadinma Daughter, Ani Njieka Ifeoma, Umeh Ifeoma Blessing, Okoro Kenneth Obinna, Nduka Sunday, Ekwunife Ikechukwu Obinna. Home Delivery of Antiretrovirals for HIV Clients amidst Covid-19 and Insecurity Challenges: A Protocol for a Randomized Controlled Trial. Journal of Pediatrics, Perinatology and Child Health 6 (2022): 070-082.
Share at FacebookAbstract
Background: Insecurity and Covid-19 has affected access to antiretroviral agents and it has resulted in suboptimal adherence to the antiretroviral therapy which could impact negatively on the overall treatment outcomes.
Objective: This study will evaluate the effectiveness, cost-benefit, and feasibility of a home-based antiretroviral delivery model in Anambra State, Nigeria.
Method/Design: The study is a randomized, non-blinded, controlled, and parallel-group trial, which will be conducted in two HIV treatment hospitals in Anambra State, Nigeria. Participants will be randomized into the intervention and control arm. Home delivery personnel will be trained to deliver the ARV drugs at 3 monthly intervals to the homes of those in the intervention group while those in the control group (Facility-based services group) will receive ARVs at the HIV treatment hospital. The study was approved by Nnamdi Azikiwe University Teaching Hospital Health Research Ethics Committee (NAUTHHREC) (NAUTH/CS/66/VOL.13/VER III/23/2020/011) on 02/10/2020 and the trial registration number, from Pan Africa Trial registry, is PACTR202004535536808 approved on 08/04/2020, http://www.pactr.org/PACTR202004535536808 The interim analysis will be done on the 12th month and the final analysis will be done on the 24th month.
Results: The interim analysis will be done on the 12th month and the final analysis will be done on the 24th month.
Conclusions: The results of this study could provide the rationale for a larger study involving various geo-political zones in Nigeria to examine the effectiveness of the home delivery model.
Keywords
HIV, ARV, Nigeria, Home delivery, COVID-19, Pandemic
HIV articles; ARV articles; Nigeria articles; Home delivery articles; COVID-19 articles; Pandemic articles
Article Details
Abbreviations:
AIDS: Acquired immunodefici- ency syndrome; ARV: Antiretroviral therapy; HIV:
Human immunodeficiency virus; RCT: Randomized controlled trial; SPSS: Statistical Package for the Social sciences; VL: Viral load; WHO: World Health Organization; NACA: National Agency for Control of AIDS; SIDSHAS: Strengthening Integrated Delivery of Services on HIV/AIDS; CARC: Comm- unity Antiretroviral Refill Club; C-PARP: Comm- unity Pharmacy Antiretroviral Refill Programme; HAART: Highly Active Antiretroviral Therapy; HDP: Home delivery Personnel; PLHA: People living with HIV/AIDS
1. Introduction
Eradication of human immunodeficiency virus infection (HIV) offers a huge opportunity to establish a healthy world for the future generation [1], so far, in the past 20 years, enormous global progress has been made against HIV. Success in the fight against HIV has been seen from 2002 to date where the death rate due to AIDS has reduced to 61% globally and the rate of new infections has been reduced by 41% [2]. Unfortunately, setbacks are now been seen as a result of challenges due to insecurity and COVID -19 pandemic, especially in many low-income countries which is the hardest hit of HIV epidemics [2-6]. Poor access and suboptimal adherence to high active antiretroviral therapy (HAART) have a significant impact on the overall treatment outcomes because of the chronic nature of the disease [7]. Hence, contin- uous access to HIV treatment is very important to achieving the possible eradication of this public health issue [8]. It is worrisome that Africa consti- tutes about sixty-seven percent (67%) of the global disease burden [9], with about 1.9 million people living with HIV in Nigeria [10]. Nonetheless, only about sixty percent (60%) of the affected adults had access to antiretroviral therapy (ART) in 2019 but a lesser value was recorded in 2020 due to insecurity and COVID 19 challenges in the country and these disruption patterns tend to reverse the progress made in the fight against HIV[2].
Insecurity has since remained a global problem and has threatened every aspect of development in the world especially in Nigeria [11, 12]. A few years ago, insecurity, banditry, and kidnapping have soared and have continued to have a destructive effect on the Nigerian economy with an overwhelmingly negative impact on the health sector and had since worsened health indices [11]. This is mostly due to inter- connecting religious, ethnic, political, and regional groups in the country [11-14]. The concept of insecurity is cross-cutting and multi-dimensional which has been subject to debates [11-13]. There are different views on insecurity, and some scholars associate it with how it affects individual lives and existence [11]. But Achumba et.al. in 2013 [11, 14] defined insecurity as a state of being subject to danger, exposure to risk, or anxiety i.e a person is said to be secured when such person is not being exposed to any form of danger or risk of physical or moral aggression, accident, theft or deterioration [11, 13]. However, the effect of insecurity on access to HIV care has not been studied in Nigeria but intervention to mitigate such challenges has been applied in some aspects.
In 2020, the world stood still for the COVID-19 pandemic and it impacted the world beyond imagination [2]. The COVID-19 is also known as the coronavirus, It is an ongoing pandemic of corona- virus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS- CoV-2) [15]. It was first identified in December 2019 in Wuhan, China [16] and to date, it has infected more than 135 million people, killed over 2.9 million people, and is projected to plunge up to 115 million people into extreme poverty [2]. Going by the 2020 target, there is a deficit of forty percent of the population of people living with HIV who had access to antiretrovirals (ARV) due to the COVID-19 pandemic [10, 11, 12]. In 2015, the World Health Organization (WHO) modified its CD4 cell count threshold in the treatment guidelines and recom- mended a test-and-treat strategy for all persons living with HIV [17]. This development substantially increased the number of people on ART [18] and with the resultant increased cohort of stable patients; it ensued a huge problem on the already burdened health system, especially in most developing count- ries. Conveniences, long waiting time to receive care and transportation costs were identified as challenges to achieving optimal care in HIV/AIDS management [19, 20]. Similarly, in Nigeria, a report showed that long distances to the service delivery points, transport costs, as well as long waiting times, and in addition insecurity and COVID 19 Pandemic have been identified as barriers to accessing antiretroviral agents in the country [2, 21, 22].
In April 2021, a recent survey by Global Fund showed the extent to which COVID-19 has disrupted health service delivery; it revealed a major decline in HIV care (40%) in over 500 health facilities across Africa and Asia [2]. In line with this, they recom- mended that adaptive mechanisms to increase access to care should be adopted such that stable clients access their therapy conveniently and affordably without necessarily visiting the hospital for HIV care [23, 24]. Delivery of antiretroviral therapy to patients' homes or close to their homes has the potential of improving adherence to therapy and viral load out- comes due to a significant reduction in waiting time and decongested healthcare facilities [24]. This is seen in the hub and spoke model of differentiated care, where HIV treatment hospitals provide HIV services through established linkages with several secondary facilities within a network, and patients are referred to those services when the indications for such arises [25-28]. Services provided by the secondary facilities include HIV counseling and testing (HCT), ARV re-fill, adherence counseling, and treatment of simple opportunistic infections (OIs) [25-29]. This approach has been successfully applied in some facilities in the UK [30]. Therefore, the development of new and innovative ways to increase access to antiretroviral therapy that would decongest health facilities is of utmost importance [31].
1.1 Study rationale
Different strategies to improve access to ARVs have been piloted by the Strengthening Integrated Delivery of Services on HIV/AIDS (SIDSHAS) in Nigeria. Among these strategies are, the Community Antir- etroviral Refill Club (CARC) for patients living in remotes areas with a particular interest in the riverine areas and the Community Pharmacy Antiretroviral Refill Programme (C-PARP) [20]. These programs are targeted at reducing visits to the hospitals by stable clients to decongest the hospitals. Also in response to the disruption caused by COVID-19, countries, and communities are devising new, innovative, and adaptive approaches to enable services to be implemented safely which in the long run will mitigate the negative impacts of COVID-19 on HIV services [2].
1.2 Study objectives
This study is designed to assess the effectiveness, client satisfaction, willingness to pay, and cost- benefit of home-based antiretroviral delivery services in Anambra State, Nigeria.
2. Methods
The SPIRIT guideline was used in reporting the research [32].
2.1 Study design
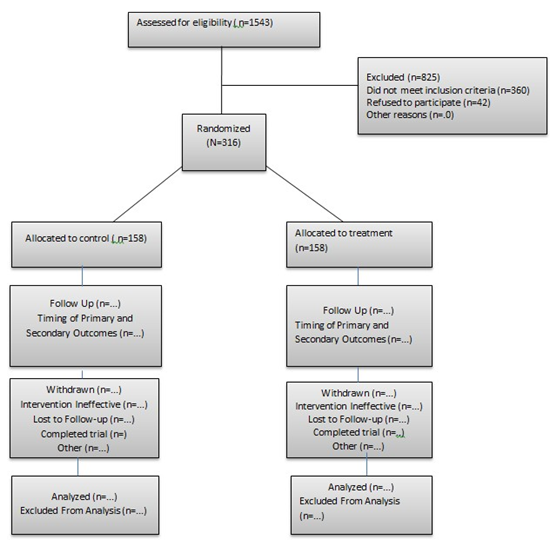
The study will be a randomized, controlled, and parallel-group balanced (1:1) trial conducted in two facilities in Anambra State, southeast Nigeria. The study aims to evaluate the effectiveness of the antiretroviral home delivery strategy in achieving undetected viral load and the SPIRIT figure was used to illustrate the timeline for the trial (see figure 1). Participants that meet the inclusion criteria in these two hospitals will be randomly assigned to the intervention or control arm (see figure 2).
2.2 Study setting
The study setting will be in Anambra state which is one of the five states in the south-eastern geopolitical zone in Nigeria. It is bounded in the northeast by Enugu state, in the east by Enugu and Abia states, in the west by Delta state while in the South and Northwest by Imo and Kogi states respectively. The State is mainly inhabited by Igbo-speaking people who are mostly Christians. Most members of the population are farmers, civil servants, artisans, and businessmen, and women [33]; with a projected population of approximately 4,177,828 (50.7% males and 49.3% females) [34]. Interestingly, about 60.5% of these populations are of ages 15-65 years [35]. Awka is the state’s capital and there are 21 local government areas in the state out of which five are urban; five are semi-urban while eleven are rural [35] and the state has an HIV prevalence rate of 2.4% [33].
2.3 Eligibility criteria
Inclusion criteria include all stable participants who commenced ART for ≥ 6months and have suppressed viral load of ≤ 1000 copies per milliliter of blood. Participants will be excluded if they: were pregnant at the time of enrolment, aged < 18years, were defaulters (i.e. those with missed appointments), had undetected viral load, or viral load <20 copies per milliliter of blood, withdrew consent, and reside outside the state.
2.4 Intervention
Enrolled participants will be directed to the research assistant responsible for introducing the study to the enrolees and they will consent to the study by signing the consent form. Those that give their consent will be sent to the physician after collecting information that will aid home delivery of their drugs including delivery points, preferred day of the week, and most available phone numbers among others. The phy- sician will then, examine the participants’ latest viral load from his/her folder and then request a baseline viral load.
Based on the physician’s recommendation that an enrolled participant qualifies for home delivery, the home delivery personnel (HDP) will deliver a three- month pre-packaged ARV in a suitable package form with a participant identifier to the participant’s chosen delivery point using a delivery motorcycle. The delivery points are jointly decided by the participant and the HDP and the drugs are to be delivered in a way to ensure confidentiality. Partici- pants in the intervention arm will be advised to visit the hospital every six months for viral load and CD4+ cell count unless there is a problem requiring the physician's attention. After each delivery, the HDPs will report back to the physician who then decides on whether to schedule the patient on appointment.
Participants in the intervention arm will be provided with a phone number to call in case of emergency or urgent consultation needs before the next appoint- tment schedule. The participant will also be referred to Nnamdi Azikwe Teaching hospital for further care in a case of non-negligent and/or harm and it will be reported to the Nnamdi Azikiwe University Teaching Hospital Ethics Committee and National Agency for Food and Drug Administration (NAFDAC) pharm- acovigilance center. In the control arm, the parti- cipants will receive their drugs in the HIV treatment hospital as usual but assess the viral load and CD4 count every six months as in the intervention group. Routine data will be collected and the patient is at liberty to withdraw from the trial at any time.
2.5 Home delivery personnel training
The community health extension worker (4 staff, who are also staff in the hospital) will serve as the home delivery personnel (HDP), and they will undergo a 5- day training based on the following guidelines: (1) the National ART training curriculum, (2) World Health Organization (WHO) guideline on manage- ment of stigma, (3) World Health Organization consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection: recommendations for a public health approach and
(4) WHO community home delivery manual. The HDPs will be trained on how to maintain utmost confidentiality when delivering the medication. They will also be trained on how to interview the parti- cipants and monitor for adherence to antiretroviral therapy during the drug delivery. The training will be carried out by an expert on HIV services.
2.6 Outcomes
The primary outcome measure will be the difference
between the HIV viral load suppression from ≤ 1000 copies per milliliter of blood to undetected viral load at the baseline, at 6, 12, 18, and 24 months in both the intervention and the control arms. The secondary outcome will measure patient satisfaction, the patient’s willingness to pay, and the cost-benefit of the home delivery model.
2.7 Sample size
The sample size for this study was calculated using a web-based Sample Size Calculator of the Clinical and Translational Science Institute, University of Califonia, San Fransico [36]. The sample size calcul- ation was based on the proportion of participants with viral load suppression of less than or equal to 1000 copies per milliliter of blood. Based on a two-sided significance level of 5% and assuming a standard deviation of 50% (increase in undetected viral load in the intervention arm compared to the control arm), the outcome in the population to be 50% and an effect size of 0.5, a minimal sample size of 264 participants was calculated to be appropriate for the study. To account for possible dropouts and loss to follow-up, an extra 20% of the calculated sample was added to the minimal sample size of 264. This resulted in a sample size of 316 participants (158 participants per group).
2.8 Randomization
Three hundred and Sixteen (316) participants living with HIV, (158 from each hospital) receiving care in the two randomly selected hospitals in Anambra and that met the eligibility criteria will be recruited for the study. Research Randomizer, a web-based computer random numbers generator will be used to generate two sets of numbers ranging from 1 – 316 [37]. Each set will contain 158 numbers. Set 1 will
represent the intervention arm while setting 2 will represent the control arm. Participants will be identified by sequential numbers, according to the order of recruitment. The allocation sequence will be conducted by an individual uninvolved in sampling and data analysis, (using the participants’ identi- fication number) and verifying which arm the participant belongs to. On the other hand, home delivery of antiretroviral which is the intervention in question would be impossible to blind since the home delivery personnel would deliver the medications to the participants at home.
2.9 Data collection techniques
Baseline data will be collected by a trained research assistant at each of the respective recruitment centers, whereas the follow-up data will be collected by the home delivery personnel. The study tools for the secondary outcome will be pretested using a pilot study at another hospital that offers ART compre- hensive services. This hospital will be located in another part of the country. Random checks will be conducted by the researchers to ensure completeness.
2.10 Statistical analysis
Statistical analysis will be conducted using IBM SPSS for Windows, Version 23 (NY, USA; IBM Corp). Descriptive statistics will be used to describe the demographic and clinical characteristics. A 2- sided p-value of 0.05 will be used to indicate statistical significance. Descriptive statistics will be used to evaluate differences in demographic and clinical characteristics. Categorical variables will be expressed as frequencies or percentages and quant- itative variables as means and standard deviations or medians and interquartile ranges. An intention-to- treat analysis will be conducted. A secondary per-protocol analysis will also be conducted. The latter includes only those participants who completed the treatment protocol originally allocated, providing results on the efficacy of the trial. Intention-to-treat analysis will only be reported if the result differs markedly from per-protocol analysis. Within-group and between-group comparisons of proportional data will be analyzed using the χ2 test or fisher’s exact test were appropriate. Within groups comparison of the mean of continuous measurement between the intervention group and control group will be condu- cted using Paired sample t-tests or Wilcoxon signed- rank tests. Between groups comparison of the mean of continuous measurement between the intervention group and control group will be conducted using Student’s t-test or Wilcoxon signed-rank tests.
Patient satisfaction will be analyzed by adopting a validated questionnaire used in a pilot study conducted in St. Mary Hospital Gwagwalada i.e the Satisfaction with Antiretroviral Home Delivery (SAHD questionnaire). The patients will be asked to respond to five categories of questionnaire items: patient demographic characteristics, Health education by the Home delivery personnel (HDP), Convenience of refill, Confidentiality, and general satisfaction level. Patient satisfaction level with home delivery will be rated on a five-point scale (ranging from ‘strongly disagree to strongly agree. The patient satisfaction will be analyzed by the Chi-square test. Contingent valuation approach using the payment card technique will be used to estimate patients’ average maximum willingness-to-pay for the home delivery service. All the resources consumed in providing the home delivery will be calculated from the health provider’s perspective. The cost-benefit ratio of the home delivery service will be calculated by dividing benefits over costs, referred to as a benefit-cost ratio (BCR). A ratio greater than 1 demonstrates a positive return on investment.
2.11 Program steering committee (PSC)
The program steering committee's role in this trial is to monitor and evaluate all the steps in research. PSC consists of five members including two patient representatives whose sole responsibility in this trial is to offer surveillance for this research.
2.12 Data monitoring committee (DMC)
The data monitoring committee consists of an independent statistician, nurse, pharmacist, clinician, and patient. The DMC reviews the data and offers advice to other committees in the trial. The DMC will also coordinate the stopping of the trial when the data has been collected. The statistician conduct a conditional power analysis of interim results or Bayesian predictive probabilities [38] will be done to identify if there is a significant difference between treatment groups, this will help the DMC make informed steps on how to end the trial. They communicate to the other trial committees to ensure that all aspect of disagreement is addressed before stopping the trial. The research assistant is respon- sible for the collation of trial data and entry into the trial database. To ensure confidentiality, the trial database will be protected by a password. The research assistant will carry out data verification and validation. The major risk that may occur in this study is the exposure of patient personal information. To curtail this risk, patient data will be pseudo- anonymized and treated with the utmost confi- dentiality.
2.13 Ethics and dissemination
Nnamdi Azikiwe University Teaching Hospital Health Research Ethics Committee (NAUTHHREC) approved this study (NAUTH/CS/66/VOL.13/VER III/23/2020/011). The trial will be conducted per the principles outlined in the Declaration of Helsinki. All identifiable data will be handled with the strictest confidentiality. Participants will fill the informed consent form and will be informed that they are free to withdraw at any time during any phase of the trial. Dissemination plans involve holding advocacy meetings with stakeholders in HIV care, publishing study outcome in an open-access peer-review journal (with an impact factor), and presentation of the outcome at an international conference.
2.14 Clinical trial registration
This trial is registered in the WHO International Clinical Trials Registry on 8th April 2020 through the WHO International Registry Network with the registration number, PACTR202004535536808.
3. Discussion
The home delivery model improves patients’ choice for care as well as minimizes interruption to patients’ HIV care [39] especially during the COVID-19 pandemic and state of insecurity that has consistently threatened the lives of the populace. This research offers a reliable option to assess the outcome of this model in the wellbeing of HIV patients. Nevertheless, related data in Nigeria on the effect of this model during the current COVID-19 and insecurity challenges is lacking. The results from this study will provide more option adoptions in this COVID-19 and insecurity challenges in this part of the globe.
We admit that this study protocol could have a potential limitation, as two HIV/AIDS sites in Nigeria would be used to access the impact of this model which may not allow for generalization of the study findings and the study cannot be blinded from both the participants and the Home delivery personnel in this study because the patient receives their ARVs at home which may not be without inherent potential bias. To curtail this potential risk we use a large sample size and randomization was to reduce inherent bias to the barest minimum.
Trial Status
The version number of this Clinical study trial is V 2.1, as various modifications have been made espe- cially to the title on the 29th August 2021, and notification has been sent to the ethics committee. The trial is expected to begin on December 1, 2021, and the recruitment will be completed approximately on Jan 30, 2022.
Availability of Data and Materials
The data used and/or analyzed during this study are available from the corresponding author on reason- able request.
Funding
This research did not receive whatsoever funding from government, private or cooperative institutions.
Competing Interests
The authors declare that they have no competing interest
Acknowledgments
We wish to acknowledge St. Charles Borromeo Specialists Hospital and Iyieanu Missionary Hospital for their approval to use their hospital for the study site as well as the provision of the HDPs. Our special appreciation goes to the PLHA support group of St. Charles Borromeo Specialist Hospital and Iyieanu Missionary Hospital for their support.
Author Contributions
NA, OE, OOD, UIB and SN designed the trial. NA, UIB, ANI, OKO and EIO drafted the first protocol. All authors participated in reviewing the protocol. NA and OO submitted the protocol for ethical clearance. All authors read and approved the final version of the protocol.
References
- 90-90-90: treatment for UNAIDS (2020).
- The impact of COVID-19 on HIV, TB and malaria services and systems for health - Updates - The Global Fund to fight AIDS, Tuberculosis, and Malaria (2021).
- Duncombe C, Ball A, Passarelli C, et Treatment 2.0: Catalyzing the next phase of treatment, care, and support. Vol. 8, Current Opinion in HIV and AIDS (2013): 4-11.
- Wolfe D, Carrieri MP, Shepard Treatment and care for injecting drug users with HIV infection: A review of barriers and ways forward. The Lancet. Elsevier B.V. 376 (2010): 355-366.
- Reda AA, Biadgilign Determinants of adherence to antiretroviral therapy among HIV-infected patients in Africa. AIDS Research and Treatment. AIDS Res Treat 2012 (2012).
- Srikantiah P, Ghidinelli M, Bachani D, et al. Scale-up of national antiretroviral therapy programs: Progress and challenges in the Asia Pacific region. AIDS 24 (2010).
- Deeks SG, Lewin SR, Havlir D V. The end of AIDS: HIV infection as a chronic disease. The Lancet. Elsevier B.V 382 (2013): 1525-
- Fast-Tracking HIV treatment; Parliamentary action and policy UNAIDS (2020).
- Global HIV & AIDS statistics — 2020 fact UNAIDS (2020).
- New survey results indicate that Nigeria has an HIV prevalence of 1.4%. UNAIDS (2021).
- Obi, Callistar. Challenges of Insecurity and Terrorism in Nigeria: Implication for National Development (February 28, 2015). OIDA International Journal of Sustainable Development 08 (2015): 11-18.
- Abadie A, Gardeazabal J. Terrorism and the world economy. Eur Econ Rev 52 (2008): 1-
- Obafemi Arinola Insecurity in northern Nigeria: Implications for maternal and child health. Clinical Epidemiology and Global Health 12 (2021).
- Eme OI, Anyadike N. Security Challenges and Security Votes in Nigeria, 2008-2013. Kuwait Chapter of Arabian Journal of Business and Management Review 2 (2013).
- World Health WHO annou- nces COVID-19 outbreak a pandemic. Geneva: World Health Organization (2020).
- Hui DS, Azhar EI, Madani TA, et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health — The latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis 91 (2020): 264-266.
- Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. WHO (2018).
- Hontelez JAC, Chang AY, Ogbuoji O, et al. Changing HIV treatment eligibility under health system constraints in sub-Saharan Africa: Investment needs, population health gains, and cost-effectiveness. AIDS 30 (2016): 2341-2350.
- Tuller DM, Bangsberg DR, Senkungu J, et Transportation costs impede sustained adherence and access to HAART in a clinic population in Southwestern Uganda: A qualitative study. AIDS Behav 14 (2010): 778-784.
- The Impact of Competing Subsistence Needs and Barriers on Access to Medical Care for Persons with Human Immuno- deficiency Virus Receiving Care in the United States on JSTOR (2021).
- Abubakar Saleh J-E. Barriers to HIV/AIDS Treatment in Am J Heal Res 3 (2015): 305.
- ANALYSIS: World AIDS Day: Why Nigeria missed 2020 HIV/AIDS target (2021).
- Security challenges in Nigeria and the implications for business activities and sustainable development - Projectchampionz Download Portal (2021).
- Global The Impact of Covid-19 on HIV, TB and Malaria Services and Systems for Health: A Snapshot From 502 Health Facilities Across Africa and Asia (2021).
- HIV/AIDS: Community models of care MSF (2020).
- Auld AF, Kamiru H, Azih C, et Evaluation of Swaziland’s hub-and-spoke model for decentralizing access to antiretroviral therapy services. J Acquir Immune Defic Syndr 69 (2015): e1-e12.
- Stafford KA, Odafe SF, Lo J, et Evaluation of the clinical outcomes of the Test and Treat strategy to implement Treat All in Nigeria: Results from the Nigeria multi-center ARt study. PLoS One 14 (2019): e0218555.
- Ware NC. Retention in Differentiated Care: Multiple Measures Analysis for a Decen- tralized HIV Care and Treatment Program in North Central Nigeria (2018).
- Geldsetzer P, Francis J, Ulenga N, et al. The impact of community health worker-led home delivery of antiretroviral therapy on virological suppression: a non-inferiority cluster-randomized health systems trial in Dar es Salaam, Tanzania. BMC Heal Serv Res 17 (2017).
- Selina Corkery Home delivery of anti-HIV drugs (2020).
- Amuron B, Levin J, Birunghi J, et al. Mort- ality in an antiretroviral therapy program in Jinja, south-east Uganda: A prospective cohort AIDS Res Ther 8 (2011): 39.
- Chan A-W, Tetzlaff JM, Gøtzsche PC, et al. SPIRIT 2013 Explanation and Elaboration: Guidance for protocols of clinical BMJ 346 (2013): e7586
- Anambra (State, Nigeria) - Population Statistics, Charts, Map and Location (2021).
- Bashorun A, Nguku P, Kawu I, et A description of HIV prevalence trends in Nigeria where is the problem?. Pan Afr Med J 18 (2014): 3.
- Olakunde BO, Ndukwe Improved Domestic Funding Enhances the Susta- inability of HIV/AIDS Response in Nigeria. Annals of Global Health. Elsevier USA 81 (2015): 684-688.
- Correlation sample size. Sample Size Cal- culators (2020).
- Research Randomizer. Michelle A. Krieger (2018).
- Heath A, Offringa M, Pechlivanoglou P, et Determining a Bayesian predictive power stopping rule for futility in a non-inferiority trial with binary outcomes. Contemp Clin Trials Commun 18 (2020): 100561.
- Smith B, Sonecha S, Murungi A, et Patient satisfaction survey for the home delivery of medication scheme. JIAS 11 (2008): 230.