Glutathione Combined with Punica granatum and Acerola Cherry Extracts reduced Melanin accumulation
Article Information
Yung-Kai Lin1, Yung-Hao Lin2,Yung-Hsiang Lin3, Chi-Fu Chiang3*
1Institute of Food Safety and Risk Management, National Taiwan Ocean University, Keelung , Taiwan. Department of Food Science, National Taiwan Ocean University, Keelung, Taiwan. Graduate Institute of Biomedical Engineering, National Chung Hsing University, Taichung, Taiwan
2Baiyuete(Shanghai)Limited Company, Shanghai, China
3Research & Design Center, TCI CO., Ltd., Taipei, Taiwan
*Corresponding Author: Chiang Chi Fu, Research & Design Center, TCI CO., Ltd., Taipei, Taiwan
Received: 12 August 2021; Accepted: 19 August 2021; Published: 14 September 2021
Citation: Yung-Kai Lin, Yung-Hao Lin,Yung-Hsiang Lin, Chi-Fu Chiang. Glutathione Combined with Punica granatum and Acerola Cherry Extracts reduced Melanin accumulation. Journal of Food Science and Nutrition Research 4 (2021): 240-248.
Share at FacebookAbstract
Blue light was common in natural light and white light, and long-term blue light exposure caused skin damage. Glutathione (GSH) inhibited melanin production and brightened the skin. In addition, the high content of polyphenol in punica grantum and vitamin C in acerola cherry had whitening effects. The purpose of this study was to explore whether GSH formula that combined GSH, punica grantum, and acerola cherry can reduce melanin formation. Blue light to stimulate A375 cell line to produce melanin, used GSH formula to treat A375 cell line, analyzed melanin, tyrosinase activity, melanogenesis associated genes, and functional analysis in CCD-996SK by nCounter analysis. GSH formula significantly decreased the melanin content and tyrosinase activity under blue light exposure. GSH formula also decreased melanogenesis associated genes. And nCounter analysis showed that GSH formula had antioxidant, anti-aging, DNA repair, and anti-inflammatory abilities. GSH formula supplemented with GSH, punica granatum, and acerola cherry extracts can reduce melanin formation, and accompanied by anti-oxidation, anti-aging, DNA repair, anti-inflammatory ability.
Keywords
Acerola cherry, Glutathione, Punica Granatum, Melanin
Acerola cherry articles; Glutathione articles; Punica Granatum articles; Melanin articles
Acerola cherry articles Acerola cherry Research articles Acerola cherry review articles Acerola cherry PubMed articles Acerola cherry PubMed Central articles Acerola cherry 2023 articles Acerola cherry 2024 articles Acerola cherry Scopus articles Acerola cherry impact factor journals Acerola cherry Scopus journals Acerola cherry PubMed journals Acerola cherry medical journals Acerola cherry free journals Acerola cherry best journals Acerola cherry top journals Acerola cherry free medical journals Acerola cherry famous journals Acerola cherry Google Scholar indexed journals Glutathione articles Glutathione Research articles Glutathione review articles Glutathione PubMed articles Glutathione PubMed Central articles Glutathione 2023 articles Glutathione 2024 articles Glutathione Scopus articles Glutathione impact factor journals Glutathione Scopus journals Glutathione PubMed journals Glutathione medical journals Glutathione free journals Glutathione best journals Glutathione top journals Glutathione free medical journals Glutathione famous journals Glutathione Google Scholar indexed journals Punica Granatum articles Punica Granatum Research articles Punica Granatum review articles Punica Granatum PubMed articles Punica Granatum PubMed Central articles Punica Granatum 2023 articles Punica Granatum 2024 articles Punica Granatum Scopus articles Punica Granatum impact factor journals Punica Granatum Scopus journals Punica Granatum PubMed journals Punica Granatum medical journals Punica Granatum free journals Punica Granatum best journals Punica Granatum top journals Punica Granatum free medical journals Punica Granatum famous journals Punica Granatum Google Scholar indexed journals Melanin articles Melanin Research articles Melanin review articles Melanin PubMed articles Melanin PubMed Central articles Melanin 2023 articles Melanin 2024 articles Melanin Scopus articles Melanin impact factor journals Melanin Scopus journals Melanin PubMed journals Melanin medical journals Melanin free journals Melanin best journals Melanin top journals Melanin free medical journals Melanin famous journals Melanin Google Scholar indexed journals age-related pathologies articles age-related pathologies Research articles age-related pathologies review articles age-related pathologies PubMed articles age-related pathologies PubMed Central articles age-related pathologies 2023 articles age-related pathologies 2024 articles age-related pathologies Scopus articles age-related pathologies impact factor journals age-related pathologies Scopus journals age-related pathologies PubMed journals age-related pathologies medical journals age-related pathologies free journals age-related pathologies best journals age-related pathologies top journals age-related pathologies free medical journals age-related pathologies famous journals age-related pathologies Google Scholar indexed journals anti-aging articles anti-aging Research articles anti-aging review articles anti-aging PubMed articles anti-aging PubMed Central articles anti-aging 2023 articles anti-aging 2024 articles anti-aging Scopus articles anti-aging impact factor journals anti-aging Scopus journals anti-aging PubMed journals anti-aging medical journals anti-aging free journals anti-aging best journals anti-aging top journals anti-aging free medical journals anti-aging famous journals anti-aging Google Scholar indexed journals cherry-like berries articles cherry-like berries Research articles cherry-like berries review articles cherry-like berries PubMed articles cherry-like berries PubMed Central articles cherry-like berries 2023 articles cherry-like berries 2024 articles cherry-like berries Scopus articles cherry-like berries impact factor journals cherry-like berries Scopus journals cherry-like berries PubMed journals cherry-like berries medical journals cherry-like berries free journals cherry-like berries best journals cherry-like berries top journals cherry-like berries free medical journals cherry-like berries famous journals cherry-like berries Google Scholar indexed journals vitamin C articles vitamin C Research articles vitamin C review articles vitamin C PubMed articles vitamin C PubMed Central articles vitamin C 2023 articles vitamin C 2024 articles vitamin C Scopus articles vitamin C impact factor journals vitamin C Scopus journals vitamin C PubMed journals vitamin C medical journals vitamin C free journals vitamin C best journals vitamin C top journals vitamin C free medical journals vitamin C famous journals vitamin C Google Scholar indexed journals anti-oxidant articles anti-oxidant Research articles anti-oxidant review articles anti-oxidant PubMed articles anti-oxidant PubMed Central articles anti-oxidant 2023 articles anti-oxidant 2024 articles anti-oxidant Scopus articles anti-oxidant impact factor journals anti-oxidant Scopus journals anti-oxidant PubMed journals anti-oxidant medical journals anti-oxidant free journals anti-oxidant best journals anti-oxidant top journals anti-oxidant free medical journals anti-oxidant famous journals anti-oxidant Google Scholar indexed journals
Article Details
1. Introduction
The visible light with a shorter 600 nm wavelength can produce pigmentation and cause melanin formation. In particular blue light studies, some evidence already demonstrated the cutaneous photoaging effects in vitro and in vivo [1,2]. Especially non-pigmented epithelial cells, blue light can cause cell dysfunction by generating ROS on DNA, resulting in cellular aging, age-related pathologies, and cancer [3]. Therefore, preventing damage from exposure to blue light was an important strategy for a healthy lifestyle. Glutathione (GSH) was considered as the anti-aging strategy for preventing reactive oxygen species (ROS) accumulation. It was also acting as the oxidative stress marker in plasma, urine, and saliva measuring [4]. GSH was an antioxidant with a thiol compound, and the tripeptide structure of GSH contained cysteine, glutamic acid, and glycine, which can regulate the melanin generation pathway of the skin. Some evidence had confirmed the variety of melanogenesis mechanisms of glutathione. (e.g., pheomelanin synthesis stimulation, antioxidant effects, and melanogenic enzyme interference). Although the whitening capacity of GSH was still controversial [5], it had been widely used as additives in benefit supplements and cosmetics applications industry for anti-aging. In recent years, collagen supplements have been widely used in the nutritional intake of modern people. To increase the anti-oxidation efficacy in collagen-based supplements for blue light exposure against, the combination of GSH and natural plant extracts was a common anti-blue light strategy. The punica granatum was originating from the Middle East, and was rich in high content of polyphenols (ellagitannins, gallotannins, anthocyanins, and catechin) and commonly processed into juice, wine, and dietary ingredient [6]. These active components contributed to various biological protective effects, including anti-inflammatory [7], antibacterial [8], antiviral [9], anti-aging [10], and anticancer [11]. The punica granatum exhibited photo-irradiated damage inhibition activity in human fibroblast cells [12]. In addition, acerola cherry was native to the southern American and Caribbean, also called West Indies cherry or Barbados cherry. This cherry-like berries is not a true cherry and has been used as a folk medicine for liver aliments, diarrhea, dysentery, and cold flu [13]. Until now, the extremely high content of acerola’s vitamin C was serving as the hot ingredient in the healthy foods industry, which is around 50-100 times than orange or lemon. Besides, this fruit contains abundant phenolic compounds, including benzoic acid derivatives, carotenoids, anthocyanins, flavonoids, and phenylpropanoids [14]. Same as punica granatum, acerola cherry also performed a great value with an anti-oxidant effect. However, it was still unclear whether the combination of GSH combined with punica granatum and acerola cherry can improve melanin production. In this study, we used GSH formula that combined GSH, punica grantum, acerola cherry, and then blue light to stimulate A375 cell line to produce melanin. Using GSH formula to treat cell line, analyzed melanin, tyrosinase activity, melanogenesis associated genes, and functional analysis in CCD-996SK by nCounter analysis. Herein, we proved the potential of inhibit melanin formation of GSH formula combined with punica granatum and acerola extract in vitro.
2. Materials and Methods
2.1 Cell lines and Materials
Human melanocyte A375 (CRL-1619) and fibroblast CCD-996SK (CRL-1881) cell lines were derived from the American Type Culture Collection (Manassas, VA, USA), and culture in growth media (Gibco®; minimum essential media, 10% fetal bovine serum, 1 mM sodium pyruvate, and 1% penicillin/streptomycin). RNA extraction kit (Genaid Biotech), nCounter platform (NanoString Technologies). Glutathione with Collagen Plus Food Supplement Capsule (650 mg; ingredients: L-glutathione, fish collagen, blueberries juice powder, spinach powder, Punica grantum, Acerola berry extracat, royal jelly, sorbital, silicon dioxide, magnesium stearate and calcium phosphate) was opened, the measured dose of ingredient powder was dissolved in liquid form and performed to experiment treatment. The GSH (Sigma-Aldrich) was purchased for experimental uses.
2.2 Melanin content measurement
The experiment was following our previous publication procedure [15]. The A375 melanocyte cells were performed in this experiment. We seeded this cell line for 1.5 × 105 cell numbers in 6-well culture plates per well under 37°C and 5% CO2 condition. Then 24 hours later, we transfer the culture plate to UV chamber (8J/cm2) to mimic blue light exposure and change the medium with fresh 3 ml of DMEM medium containing 0.25 mg/ml kojic acid, or GSH formula 0.25 mg/ml fruit extract, or blank solvent as mock set. Cells were harvested after incubation at 37°C for 48 h. Rinse the collected cells with 1 × PBS twice and then resuspend in 200 μl of 1 × PBS. The total cell lysates were obtained by freeze-thaw in liquid nitrogen for 10 minutes and room temperature for 30 minutes. Then used 12,000 × g (Thermo ScientificTM HeraeusTM FrescoTM 17 Microcentrifuge, Langenselbold, Germany) centrifugation for collecting the cell lysates by 30 minutes. Suspend the pellets with 120 μl of 1 N NaOH in 60°C dry bath for 1 hour. Take 100 μl of samples to 96-well plate for spectrophotometric measurement at 405 nm. The amounts of melanin were calculated as: Relative melanin formation (%) = (ODsample/ODcontrol) × 100%.
2.3 Tyrosinase activity assay
The experiment was following our previous publication procedure [15]. The A375 melanocyte cells were performed in this experiment. We seeded this cell line for 1.5 × 105 cell numbers in 6-well culture plates per well under 37°C and 5% CO2 condition..After that, transfer the culture plate to UV chamber (8J/cm2) and change the medium with fresh 3 ml of DMEM medium containing 0.25 mg/ml kojic acid, or GSH formula 0.25 mg/ml fruit extract, or blank solvent as mock set. Cells were harvested after incubation at 37°C for 48 h. Rinse the collected cells with 1 × PBS twice and then resuspend in 200 μl of 1 × PBS. The cell lysates were obtained by freeze-thaw in liquid nitrogen for 10 min and room temperature for 30 minutes. Collect the pellets of cell lysates by centrifugation at 12,000 × g (Thermo ScientificTM HeraeusTM FrescoTM 17 Microcentrifuge, Langenselbold, Germany) for 30 minutes. After protein concentration determination. Perform the same concentration protein in a 96-well microtiter plate and adding total volume to the 90 μL. Protect from light and add 10 μL L-Dopa for 20 mins action response. After liquid becomes dark status, measure OD 450 for melanin content by ELISA reader.
2.4 Analysis of mRNA expression
1.5 × 105 CCD-966SK cells in 2 ml of the media with 0.25mg/ml of GSH formula were seeded in each well of 6-well plates and incubated for 24 hours. Then, we collected the cells and used the RNA extraction kit for RNA collection. Finally, we adjusted the RNA concentration to 75 ng/μL for mRNA expression analysis. The mRNA expression level was analyzed by qPCR. Primer sequence: TYR (F: CTCAAAGCAGCATGCACAAT/R: GCCCAGATCTTTGGATGAAA), TYRP1(F: GACACGCCTCCTTTTTATTCCA/R: ATGGGTTTGTCCCCCTGTTC), MC1R (F: CATCATCGACCCCCTCATCTAC/R: CAGGAACCAGACCACACAATATCA), MITF (F:GCCTCCAAGCCTCCGATAAG/R: GCACTCTCTGTTGCATGAACT)
Gene screening was performed nCounter gene expression platform (NanoString Technologies), and all the operation steps were referenced by the recommend protocol and our previous publication [15,16].
2.5 Statistical Analysis
All values were expressed as mean ±SD. Between sample populations differences statistical result was determined by an unpaired two-tailed Student’s t-test. Statistical significance was considered at p value < 0.05.
3. Results
3.1 GSH formula decreased melanin content under blue light exposure
To clarify whether the GSH formula can reduce melanin, we used blue light to treat human melanoma cell line A375 to induce melanin. Figure 1 showed that melanin content was increased to 113.7% under blue light exposure. Kojic acid (0.25 mg/ml) was considered as the whitening agent in cosmetics and used as the positive control in the anti-melanogenesis experiment [17]. The kojic acid treatment decreased the melanin content to 90.3%. GSH (0.25 mg/ml) treatment decreased the melanin content to 106%. Interestingly, GSH formula (0.25 mg/ml) treatment decreased the melanin content to 94.4%, and the inhibitory effect was similar to kojic acid. This result suggested that GSH formula can decrease melanin content under blue light exposure.
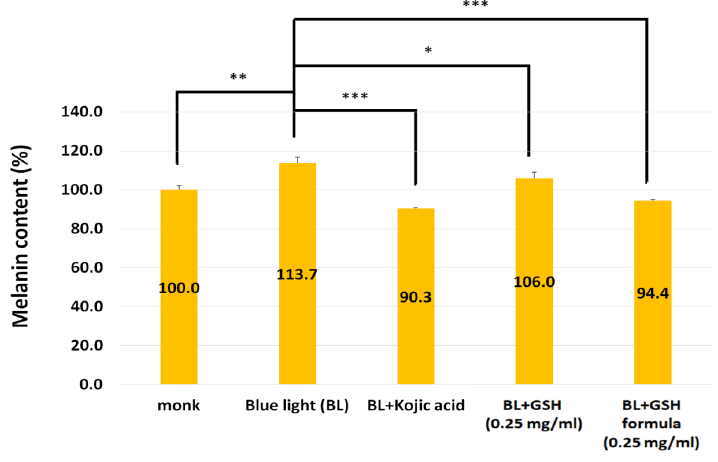
Figure 1: Effect of GSH formula reduces the melanin content compared to the GSH, kojic acid treatment only under blue light exposure. N=3, Data are means ± SD. Compared to the blue light exposed only (*, p < 0.05,**, p < 0.01; ***, p < 0.001)
3.2 GSH formula decreased tyrosinase acitivity and melanogenesis-associated gene expression.
Melanin was synthesized by tyrosinase [18]. To clarify whether the GSH formula can reduce tyrosinase acitivity, we treated the cells with GSH and then measured the tyrosinase activity. The figure 2 showed that tyrosinase activity was increased to 138.3% under blue light exposure, and kojic acid treatment decreased tyrosinase activity to 84.6%. GSH formula treatment decreased the tyrosinase activity to 66.1%. This result suggested that GSH formula can decrease tyrosinase activity under blue light exposure. Next, we examined melanogenesis-associated gene expression, TYRP1 (Tyrosinase Related Protein 1), MC1R (Melanocortin 1 receptor), MITF (Melanocyte Inducing Transcription Factor), by Q-PCR. The figure 3 also showed that GSH formula can decrease melanogenesis-associated gene expression under blue light exposure. These results suggested that the GSH formula decreased tyrosinase acitivity and melanogenesis-associated gene expression.
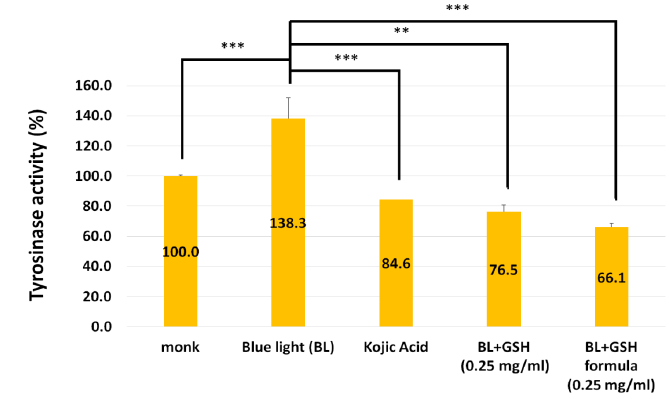
Figure 2: Effect of GSH formula reduces the tyrosinase activity compared to the kojic acid, GSH treatment only under blue light exposure. N=3, Data are means ± SD. Compared to the blue light exposed only (*, p < 0.05,**, p < 0.01; ***, p < 0.001)
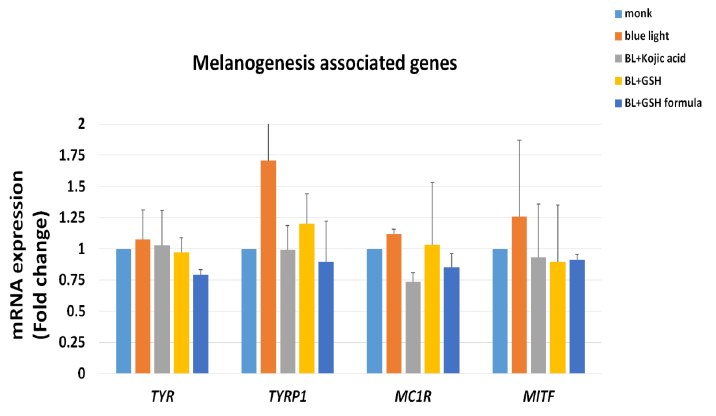
Figure 3: Melanogenesis genes mRNA expression effects of GSH formula treatment compared to the mock under blue light exposure. N=2, Data are means ± SD. Compared to the blue light exposed only. TYR (tyrosinase), TYRP1 (Tyrosinase Related Protein 1), MC1R (Melanocortin 1 receptor), MITF (Melanocyte Inducing Transcription Factor)
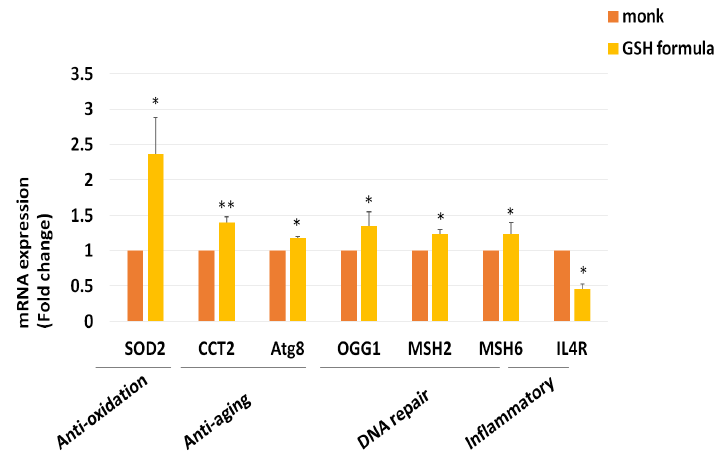
Figure 4: GSH formula treatment affected other functional genes mRNA by nCounter® screening. N=3, Data are means ± SD. Compared to the blue light exposed only (*, p < 0.05,**, p < 0.01; ***, p < 0.001) SOD2 (Superoxide dismutase 2),CCT2 (Chaperonin Containing TCP1 Subunit 2), Atg8 (Autophagy-related protein 8 ),OGG1 (8-Oxoguanine glycosylase),MSH2 (MutS Homolog 2),MSH6 (mutS homolog 6),IL4R ( interleukin-4 receptor).
3.3 The other functions of GSH formula
Next, we want to explore whether GSH formula had other functions. Using GSH formula to treat CCD-996SK cell lines, and then examined by nCounter® analysis. We examined SOD2 (Superoxide dismutase 2) (anti-oxidation gene), CCT2 (Chaperonin Containing TCP1 Subunit 2) and Atg8(Autophagy-related protein 8) (anti-aging gene), OGG1(8-Oxoguanine glycosylase), MSH2 (MutS Homolog 2) and MSH6 (mutS homolog 6) (DNA repair gene), and IL4R ( interleukin-4 receptor) (inflammatory gene). The figure 4 showed that GSH formula can increase SOD2, CCT2, Atg8, OGG1, MSH2 and MSH6 gene expression, and decrease IL4R. Thus, GSH formula had antioxidant, anti-aging, DNA repair, and anti-inflammatory abilities.
4. Discussion
At the beginning of artificial light was available, the pressure of blue light exposure was around us. More and more evidence revealed the phototoxic for the skin damage and retina. Daily supplement intake is used to common habits of modern human style. Nowadays, GSH combined with vitamin C was regarded as a synergistic improvement of anti-oxidative efficacy [19]. In this study, we demonstrated GSH formula skin-lightening effects combined with high polyphenols of punica granatum ferments and high vitamin C of acerola cherry extract. Previous evidence suggested phenolics can increase the intrinsic GSH levels of subjects [20]. In addition, naturally occurring in vegetables, fruits, and plant-derived polyphenols also performed the coordinated regulation with GSH and its related enzymes [21]. More precisely, phenolic acids combined with GSH have synergistic effects in anti-oxidation [22]. This evidence indicated our GSH formula combined with high polyphenols of punica granatum ferments is a great strategy for enhancing the anti-oxidant effects of glutathione. At the cellular level, senescence is a central hallmark of the aging process, and the senescence marker protein-30 (SMP30) expression was decreased during aging in zebrafish [23]. And acerola juice suppresses UVB-induced skin pigmentation efficacy has been provided in gluconolactonase SMP30 knockout hairless mice model [24]. In japan study also suggested acerola juice intake decrease the vitamin C excretion through urine in healthy subjects, which is in correspondence to the bioavailability of vitamin C improvement [25]. This evidence was suggested our strategy for supplementing acerola cherry in GSH formula. Taking together, the composition of GSH formula was supplemented with punica granatum ferments, acerola cherry extract with glutathione. And further examined the anti-pigmentation efficacy of this formula also performed efficacy enhancement effects (Figure.1,2,3). Despite kojic acid performed and great skin lightening efficacy in the cosmetic industry, it has been considered as a carcinogen and revealed the chronic toxicity response from dietary administration in rats [26,27]. Our combination GSH formula can successfully enhance the anti-pigmentation effect than GSH mono-treatment and performed a great efficacy closed to kojic acid mono-treatment. Moreover, our GSH formula was combined with safety botanical supplements to lessen adverse effects. GSH has been reported which can facilitate collagen-mediated fibroblast contraction and prevent cell death effect in cell line findings [28]. In our nCounter® screening findings, GSH formula can regulate anti-oxidant, anti-aging, DNA-repair, and anti-inflammation associated gene significantly (Figure 4). This result suggested the GSH formula didn’t only prevent the blue light exposure induced melanogenesis, it also performed other anti-aging efficacy.
5. Conclusion
In this study, the GSH formula was examined for the anti-pigmentation efficacy by cell line analysis. To combined high polyphenols punica granatum ferments, and acerola cherry extract. This formula successfully enhanced the anti-pigmentation efficacy of the glutathione. In addition, the skin-lightening effects of collagen also facilitated by these supplements. And GSH formula also contained other anti-aging properties through regulating different mechanisms associated with genes by nCounter screening. We demonstrated the comprehensively against blue light exposure protective function and anti-aging potential of GSH formula.
Conflict of interests
The authors have declared that there is no conflict of interest regarding this research.
References
- Mahmoud BH, Hexsel CL, Hamzavi IH, et al. Effects of visible light on the skin. Photochemistry and photobiology 84 (2008): 450-462.
- Sondenheimer K, Krutmann J. Novel Means for Photoprotection. Frontiers in medicine 5 (2018): 162.
- Godley BF, Shamsi FA, Liang FQ, et al. Blue light induces mitochondrial DNA damage and free radical production in epithelial cells. J Biol Chem 280 (2005): 21061-21066.
- Gyurászová M, Kovalcíková A, Janšáková K, et al. Markers of oxidative stress and antioxidant status in the plasma, urine and saliva of healthy mice. Physiological research 67 (2018): 921-934.
- Sonthalia S, Daulatabad D, Sarkar R. Glutathione as a skin whitening agent: Facts, myths, evidence and controversies. Indian journal of dermatology, venereology and leprology 82 (2016): 262-272.
- Middha SK, Usha T, Pande V. HPLC evaluation of phenolic profile, nutritive content, and antioxidant capacity of extracts obtained from Punica granatum fruit peel. Advances in Pharmacological Sciences 11 (2013): 236-296.
- Yahfoufi N, Alsadi N, Jambi M, et al. The immunomodulatory and anti-inflammatory role of polyphenols. Nutrients 10 (2018): 16-18.
- Daglia M. Polyphenols as antimicrobial agents. Current Opinion in Biotechnology 23 (2012): 174-181.
- Serkedjieva J, Toshkova R, Antonova-Nikolova S, et al. Effect of a plant polyphenol-rich extract on the lung protease activities of influenza-virus-infected mice. Antiviral chemistry & chemotherapy 18 (2007): 75-82.
- Shimizu C, Wakita Y, Inoue T, et al. Effects of lifelong intake of lemon polyphenols on aging and intestinal microbiome in the senescence-accelerated mouse prone 1 (SAMP1). Scientific Reports 9 (2019): 36-71.
- Niedzwiecki A, Roomi MW, Kalinovsky T, et al. Anticancer efficacy of polyphenols and their combinations. Nutrients 8 (2016): 552.
- Park HM, Moon E, Kim AJ, et al. Extract of Punica granatum inhibits skin photoaging induced by UVB irradiation. International journal of dermatology 49 (2010): 276-282.
- Nagamine I, Fujita M, Hongo I, et al. Hepatoprotective Effects of acerola cherry extract powder against d-galactosamine-induced liver injury in rats and its bioactive compounds. Czech Journal of Food Sciences 22 (2018): 159-162.
- Prakash A, Baskaran R. Acerola, an untapped functional superfruit: a review on latest frontiers. J Food Sci Technol 55 (2018): 3373-3384.
- Yu CH, Lin Y, Su HL, et al. Unripe fruit extracts of Mangifera indica L. protect against AGEs formation, melanogenesis and UVA-induced cell damage 10 (2019): 188-197.
- Kulkarni MM. Digital multiplexed gene expression analysis using the nanostring ncounter system. Current Protocols in Molecular Biology 94 (2011): 25-40.
- Lajis AFB, Hamid M, Ariff AB. Depigmenting effect of Kojic acid esters in hyperpigmented B16F1 melanoma cells. J Biomed Biotechnol 12 (2012): 952452.
- Iozumi K, Hoganson GE, Pennella R, et al. Role of tyrosinase as the determinant of pigmentation in cultured human melanocytes. The Journal of investigative dermatology 100 (1993): 806-811.
- Gitto E, Tan DX, Reiter RJ, et al. Individual and synergistic antioxidative actions of melatonin: studies with vitamin E, vitamin C, glutathione and desferrioxamine (desferoxamine) in rat liver homogenates. The Journal of pharmacy and pharmacology 53 (2001): 1393-1401.
- Visioli F, Wolfram R, Richard D, et al. Olive phenolics increase glutathione levels in healthy volunteers. Journal of agricultural and food chemistry 57 (2009): 1793- 1796.
- Giovannini C, Filesi C, D'Archivio M, et al. [Polyphenols and endogenous antioxidant defences: effects on glutathione and glutathione related enzymes]. Ann Ist Super Sanita 42 (2006): 336-347.
- Ran L, Chi Y, Huang Y, et al. Synergistic antioxidant effect of glutathione and edible phenolic acids and improvement of the activity protection by coencapsulation into chitosan-coated liposomes. LWT 127 (2020): 109409.
- Fujisawa K, Terai S, Hirose Y, et al. Senescence marker protein 30 (SMP30)/regucalcin (RGN) expression decreases with aging, acute liver injuries and tumors in zebrafish. Biochemical and biophysical research communications 414 (2011): 331-336.
- Sato Y, Uchida E, Aoki H, et al. Acerola (Malpighia emarginata DC.) Juice Intake Suppresses UVB-Induced Skin Pigmentation in SMP30/GNL Knockout Hairless Mice. Plos one 12 (2017): e0170438.
- Uchida E, Kondo Y, Amano A, et al. Absorption and excretion of ascorbic acid alone and in acerola (Malpighia emarginata) juice: comparison in healthy Japanese subjects. Biological & pharmaceutical bulletin 34 (2011): 1744-1747.
- Ota Y, Imai T, Onose J, et al. A 55-week chronic toxicity study of dietary administered kojic acid (KA) in male F344 rats. The Journal of Toxicological Sciences 34 (2009): 305-313.
- Higa Y, Kawabe M, Nabae K, et al. Kojic acid -absence of tumor-initiating activity in rat liver, and of carcinogenic and photo-genotoxic potential in mouse skin. J Toxicol Sci 32 (2007): 143-1459.
- Deveci M, Gilmont RR, Dunham WR, et al. Glutathione enhances fibroblast collagen contraction and protects keratinocytes from apoptosis in hyperglycaemic culture. British Journal of Dermatology 152 (2005): 217-224.
