Generalized lymphatic anomaly: an unusual cause of mediastinal widening -A case report
Article Information
Tamanna Khullar*, Rashmi Dixit, Monica Juneja, Gaurav Pradhan
Affiliation:
Maulana Azad Medical College,Bahadur shah zafar marg,Delhi ,India
*Corresponding author:
Tamanna Khullar, Maulana Azad medical college, Bahadur shah zafar marg, Delhi,India
Received: December 11, 2022; Accepted: December 20, 2022; Published: December 26, 2022
Citation:
Tamanna Khullar, Rashmi Dixit, Monica Juneja, Gaurav Pradhan. Generalized lymphatic anomaly: an unusual cause of mediastinal widening - A case reportJournal of Radiology and Clinical Imaging 5 (2022): 87-90.
Share at FacebookAbstract
While simple lymphatic malformations which may be microcystic or macrocystic (Cystic hygroma) are quite common, complex lymphatic anomalies such as generalized lymphatic anomaly are uncommon. Due to its rarity and nonspecific presentation, diagnosis may often be delayed. A 7yr old male presented with non-specific respiratory symptoms and knee pain. Chest imaging findings of low attenuation soft tissue mass with infiltration along the lymphatic distribution adjacent to the bronchovascular bundles and multiple punched out lytic lesions in the bones with preserved cortex were seen. In the given clinical scenario, laboratory findings, these imaging findings were highly suggestive for generalised lymphatic anomaly.We suggest a complete radiological evaluation in such children as imaging can be highly suggestive even in the absence of genetic or histopathological diagnosis.
Keywords
Generalised lymphatic anomaly; Kaposiform lymphangiomatosis; central conducting anomaly; Gorham stout disease; lymphatic anomalies
Generalised lymphatic anomaly articles; Kaposiform lymphangiomatosis articles; central conducting anomaly articles; Gorham stout disease articles; lymphatic anomalies articles
Generalised lymphatic anomaly articles Generalised lymphatic anomaly Research articles Generalised lymphatic anomaly review articles Generalised lymphatic anomaly PubMed articles Generalised lymphatic anomaly PubMed Central articles Generalised lymphatic anomaly 2023 articles Generalised lymphatic anomaly 2024 articles Generalised lymphatic anomaly Scopus articles Generalised lymphatic anomaly impact factor journals Generalised lymphatic anomaly Scopus journals Generalised lymphatic anomaly PubMed journals Generalised lymphatic anomaly medical journals Generalised lymphatic anomaly free journals Generalised lymphatic anomaly best journals Generalised lymphatic anomaly top journals Generalised lymphatic anomaly free medical journals Generalised lymphatic anomaly famous journals Generalised lymphatic anomaly Google Scholar indexed journals Kaposiform lymphangiomatosis articles Kaposiform lymphangiomatosis Research articles Kaposiform lymphangiomatosis review articles Kaposiform lymphangiomatosis PubMed articles Kaposiform lymphangiomatosis PubMed Central articles Kaposiform lymphangiomatosis 2023 articles Kaposiform lymphangiomatosis 2024 articles Kaposiform lymphangiomatosis Scopus articles Kaposiform lymphangiomatosis impact factor journals Kaposiform lymphangiomatosis Scopus journals Kaposiform lymphangiomatosis PubMed journals Kaposiform lymphangiomatosis medical journals Kaposiform lymphangiomatosis free journals Kaposiform lymphangiomatosis best journals Kaposiform lymphangiomatosis top journals Kaposiform lymphangiomatosis free medical journals Kaposiform lymphangiomatosis famous journals Kaposiform lymphangiomatosis Google Scholar indexed journals central conducting anomaly articles central conducting anomaly Research articles central conducting anomaly review articles central conducting anomaly PubMed articles central conducting anomaly PubMed Central articles central conducting anomaly 2023 articles central conducting anomaly 2024 articles central conducting anomaly Scopus articles central conducting anomaly impact factor journals central conducting anomaly Scopus journals central conducting anomaly PubMed journals central conducting anomaly medical journals central conducting anomaly free journals central conducting anomaly best journals central conducting anomaly top journals central conducting anomaly free medical journals central conducting anomaly famous journals central conducting anomaly Google Scholar indexed journals Gorham stout disease articles Gorham stout disease Research articles Gorham stout disease review articles Gorham stout disease PubMed articles Gorham stout disease PubMed Central articles Gorham stout disease 2023 articles Gorham stout disease 2024 articles Gorham stout disease Scopus articles Gorham stout disease impact factor journals Gorham stout disease Scopus journals Gorham stout disease PubMed journals Gorham stout disease medical journals Gorham stout disease free journals Gorham stout disease best journals Gorham stout disease top journals Gorham stout disease free medical journals Gorham stout disease famous journals Gorham stout disease Google Scholar indexed journals lymphatic anomalies articles lymphatic anomalies Research articles lymphatic anomalies review articles lymphatic anomalies PubMed articles lymphatic anomalies PubMed Central articles lymphatic anomalies 2023 articles lymphatic anomalies 2024 articles lymphatic anomalies Scopus articles lymphatic anomalies impact factor journals lymphatic anomalies Scopus journals lymphatic anomalies PubMed journals lymphatic anomalies medical journals lymphatic anomalies free journals lymphatic anomalies best journals lymphatic anomalies top journals lymphatic anomalies free medical journals lymphatic anomalies famous journals lymphatic anomalies Google Scholar indexed journals imaging articles imaging Research articles imaging review articles imaging PubMed articles imaging PubMed Central articles imaging 2023 articles imaging 2024 articles imaging Scopus articles imaging impact factor journals imaging Scopus journals imaging PubMed journals imaging medical journals imaging free journals imaging best journals imaging top journals imaging free medical journals imaging famous journals imaging Google Scholar indexed journals Magnetic resonance imaging articles Magnetic resonance imaging Research articles Magnetic resonance imaging review articles Magnetic resonance imaging PubMed articles Magnetic resonance imaging PubMed Central articles Magnetic resonance imaging 2023 articles Magnetic resonance imaging 2024 articles Magnetic resonance imaging Scopus articles Magnetic resonance imaging impact factor journals Magnetic resonance imaging Scopus journals Magnetic resonance imaging PubMed journals Magnetic resonance imaging medical journals Magnetic resonance imaging free journals Magnetic resonance imaging best journals Magnetic resonance imaging top journals Magnetic resonance imaging free medical journals Magnetic resonance imaging famous journals Magnetic resonance imaging Google Scholar indexed journals Mammalian target of rapamycin articles Mammalian target of rapamycin Research articles Mammalian target of rapamycin review articles Mammalian target of rapamycin PubMed articles Mammalian target of rapamycin PubMed Central articles Mammalian target of rapamycin 2023 articles Mammalian target of rapamycin 2024 articles Mammalian target of rapamycin Scopus articles Mammalian target of rapamycin impact factor journals Mammalian target of rapamycin Scopus journals Mammalian target of rapamycin PubMed journals Mammalian target of rapamycin medical journals Mammalian target of rapamycin free journals Mammalian target of rapamycin best journals Mammalian target of rapamycin top journals Mammalian target of rapamycin free medical journals Mammalian target of rapamycin famous journals Mammalian target of rapamycin Google Scholar indexed journals Computed tomography articles Computed tomography Research articles Computed tomography review articles Computed tomography PubMed articles Computed tomography PubMed Central articles Computed tomography 2023 articles Computed tomography 2024 articles Computed tomography Scopus articles Computed tomography impact factor journals Computed tomography Scopus journals Computed tomography PubMed journals Computed tomography medical journals Computed tomography free journals Computed tomography best journals Computed tomography top journals Computed tomography free medical journals Computed tomography famous journals Computed tomography Google Scholar indexed journals histopathological diagnosis articles histopathological diagnosis Research articles histopathological diagnosis review articles histopathological diagnosis PubMed articles histopathological diagnosis PubMed Central articles histopathological diagnosis 2023 articles histopathological diagnosis 2024 articles histopathological diagnosis Scopus articles histopathological diagnosis impact factor journals histopathological diagnosis Scopus journals histopathological diagnosis PubMed journals histopathological diagnosis medical journals histopathological diagnosis free journals histopathological diagnosis best journals histopathological diagnosis top journals histopathological diagnosis free medical journals histopathological diagnosis famous journals histopathological diagnosis Google Scholar indexed journals
Article Details
Introduction
Simple lymphatic malformations such as microcystic and macrocystic lymphatic malformations are commonly seen, while complex lymphatic anomalies which include Generalized lymphatic anomaly (GLA), kaposiform lymphangiomatosis( KLA), Gorham stout disease (GSD) and central conducting lymphatic anomaly (CCLA) are rare disorders. Generalized lymphatic anomaly (GLA) is a rare complex lymphatic anomaly characterized by abnormal proliferation of lymphatic vessels. Exact incidence of GLA remains unknown owing to its rarity. It is has a multiorgan involvement yet thoracic involvement is uncommon. Thoracic involvement in GLA may have a poorer prognosis when compared to other sites of involvement. Skeletal involvement have typical imaging features which can help in distinguishing it from other complex lymphatic anomalies such as Gorham stout disease. Management of this entity is largely supportive.
Case Presentation
A 7 year old male child presented to the outpatient department with complaints of dry cough and breathlessness on and off since one and half years with increase in severity from last 6 weeks. There was no history of fever or wheeze or significant loss in weight. The child also had been complaining of knee pain for past 6 months. The birth history was unremarkable .There was no family history of asthma or tuberculosis. On general physical examination the child appeared weak and emaciated with lower BMI for his age. The child had stable vitals with mild pallor, no icterus or cyanosis. There were no palpable
lymph nodes. Chest examination revealed normal air entry bilaterally with few bibasilar crackles. No organomegaly or palpable mass was noted on abdominal examination. Neurological examination was also unremarkable. Complete blood count revealed a hemoglobin of 10mg/dl and a platelet count of 141000/µL suggestive of mild anemia and thrombocytopenia. Total leukocyte and differential leucocyte counts were normal.
In view of the clinical complaints of cough and breathlessness and knee pain, an X ray chest and AP radiograph of the knee was requested. The frontal radiograph revealed a well-defined soft tissue density lesion causing
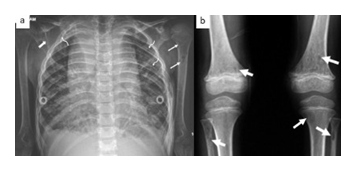
Fig 1 (a): Frontal radiograph in the child demonstrating soft tissue density lesion occupying the mediastinum from thoracic inlet to the diaphragm silhouetting bilateral cardiac borders, descending thoracic aorta suggesting a multicompartmental lesion. Multiple punched out lytic lesions are noted in ribs(curved white arrows), right scapula (notched white arrow) and humerus(straight white arrows).
Fig 1 (b): AP radiograph of bilateral knee shows multiple small punched out lytic lesions in bilateral distal femoral metaphysis and proximal metaphysis of bilateral fibula and left tibia with preserved cortical outline.(straight white arrows).No associated soft tissue lesion is seen.
symmetric mediastinal widening from the level of thoracic inlet to the level of diaphragm. The trachea was central with no displacement or luminal narrowing. The lesion was seen to silhouette the right and left cardiac borders and the descending thoracic aorta. Hilar vessels were seen through the lesion suggesting the possibility of a multi-compartmental mediastinal mass. Few reticulo nodular opacities were also noted in both lung fields, more prominent in bilateral lower zones .No pleural or pericardial effusion was seen. Few small well defined lytic lesions having a punched out appearance were also noted involving multiple ribs, right scapula and metaphysis and diaphysis of left humerus.[Fig.1a] On AP radiograph of knee, multiple punched out lytic lesions were observed in bilateral distal femoral metaphysis and proximal metaphysis of bilateral fibula and left tibia.[Fig.1b]
In view of the above findings, a contrast enhanced CT of the chest was done to know the nature and extent of the anticipated mediastinal mass and lung findings. Contrast enhanced CT chest revealed a large homogenous low attenuation, poorly enhancing multicompartmental mediastinal lesion spanning from anterior to the posterior chest wall. The lesion showed no evidence of calcification or fat within. The major aortic vessels and its branches were seen to course through the lesion without any luminal attenuation or obliteration. This soft tissue lesion was also seen to extend along the bronchovascular bundles. Lung window revealed bilateral septal thickening in peribronchovascular distribution reaching up till the pleural surface with sub pleural nodules. No pleural effusion or pericardial effusion was noted. Cardiac size and morphology appeared normal. Visualised sections of liver and spleen showed no focal lesions, minimal extension
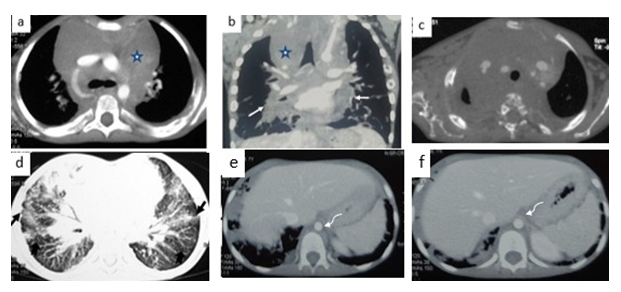
FIG2: Contrast enhanced CT axial (a) and coronal reformatted image(b) shows a low attenuation poorly enhancing multicompartmental soft tissue density lesion (star) insinuating between the vessels of mediastinum without any luminal narrowing or obliteration . It can be seen infiltrating along bronchovascular bundles bilaterally.(white arrows)Axial bone window(c) demonstrates multiple lytic lesions in right scapula and vertebral body. Axial lung window image (d) reveals nodular septal thickening along peribronchovascular distribution reaching up till pleural surface(notched black arrows) with sub pleural nodules.(straight black arrows) Axial soft tissue window(e,f) depicts no focal lesions in liver or spleen with minimal periaortic soft tissue(curved white arrows).
of soft tissue was also noted along the visualized upper retroperitoneum surrounding the aorta. [Fig.2] An ultrasound of the abdomen and neck was unremarkable. In view of the long clinical course and imaging morphology of chest and musculoskeletal lesions an imaging diagnosis of generalized lymphatic anomaly /kaposiform lymphangiomatosis was considered. Parents of the child had refused genetic testing or histopathological examination. However, absence of any hemorrhagic effusions or any clinical history of hemoptysis/ bleeding, minimal decrease in platelet count and a less aggressive clinical picture favored a diagnosis of GLA.
Discussion
Complex lymphatic anomalies are rare conditions which occur due to anomalous embryogenesis of the lymphatic system. [1]These include Generalized lymphatic anomaly (GLA), kaposiform lymphangiomatosis( KLA), Gorham stout disease (GSD) and central conducting lymphatic anomaly (CCLA). These are associated with significant morbidity and mortality. [2]Overlapping clinical symptoms and imaging appearances are often seen amongst these which pose a diagnostic difficulty. Generalised lymphatic anomaly is usually seen in childhood though may be diagnosed in adults and has no gender predilection. Exact incidence of GLA remains unknown owing to its rarity. The imaging features are mainly based on case reports due to relative rarity of this disease.However, imaging can be suggestive of diagnosis as biopsy may not always be possible in these cases.
The presentation is variable and the clinical course varies according to site of involvement and extent of disease. Different sites involved includes the thorax, liver, spleen, retroperitoneum and the skeletal system. Thoracic involvement has been linked to a poorer prognosis when compared to other organ involvement. [3]
The thoracic manifestations of GLA seen on imaging include a soft tissue thickening/low attenuation soft tissue mediastinal mass. Peribronchovascular septal thickening, diffuse or segmental having a variable extension to subpleural surface has also been reported. Pleural effusion of variable nature is another observed finding. Osseous involvement in GLA is seen to favor the appendicular skeleton with multiple sites involved in a non-contiguous manner. These have been described as non-contiguous punched out lytic lesions without sclerotic rim with preservation of cortex, features which were seen in our case.
Abdominal involvement may manifest as cysts within the viscera such as pancreas, spleen, liver or kidney with or without associated organomegaly. These may well be demonstrated on sonography. Our case however did not reveal any abdominal imaging manifestations.
These imaging findings of GLA are often indistinguishable from KLA and distinction is based on a more aggressive clinical picture of hemoptysis, hemorrhagic effusions,thrombocytopenia and high mortality seen in KLA. KLA is considered to be a more aggressive disease presenting early in childhood and thought to be arising from GLA. Thoracic involvement is much more common in KLA in comparison to GLA. In patients with hypofibrinogenemia, significant thrombocytopenia, hemoptysis and hemorrhagic effusions, KLA should be suspected over GLA. Diagnosis may be inferred clinically if biopsy is unsafe. Recent studies are investigating the role of genetics in these anomalies and it was found that somatic NRAS p.Q61R variant has a role in pathogenesis of KLA.[4]Though the genetic testing and histopathology was not done in our case, mild clinical course with no pleural /pericardial involvement and non-significant thrombocytopenia suggested the possibility of GLA over KLA.
Another complex lymphatic anomaly associated with osseous and soft tissue involvement is Gorham stout disease. Clinical presentation is often with pain and bone fractures. The osseous lesions in GSD are characterised by progressive osteolysis and cortical destruction (also called as “vanishing bone disease”) and peri osseous infiltrating soft tissue in contrast to GLA where the cortex is preserved and there is no periosteal soft tissue.[5] The lesions can be appreciated well, both on plain radiography or CT. Greater number of bones are involved in GLA when compared to GSD. Appendicular skeleton involvement is also more commonly observed with GLA (A comparative study between GLA and GSD has reported that ribs are most frequently involved in both the diseases. However, skull, clavicle, and cervical spine are more commonly involved in GSD whereas thoracic spine, humerus and femur are more commonly involved in GLA). [6]Punched out lytic lesions were observed in our case with involvement of scapula, ribs, thoracic vertebrae, humerus, femur, tibia and fibula with extension to the cortex, however there was no cortical resorption. Biopsy from the rib lesions for diagnosis is discouraged because of the risk of development of refractory pleural effusions post biopsy. Recently, imaging findings have been considered to differentiate between GLA and GSD.[3,6]
Central conducting lymphatic anomaly (CCLA) is another complex anomaly wherein imaging manifestations are poorly described owing to its rarity. It results due to failure to drain the lymph into venous system due to abnormality of central channels like thoracic duct or cisterna chyli. Depending on the site of the anomaly, manifestions such as chylothorax, pulmonary lymphangiectasia, chylous ascites, protein-losing enteropathy, cutaneous vesicles, or superficial chylous leaks may be seen.[1]Osseous changes can also be seen in this entity as focal areas of hyperlucency due to dilated intraosseous channels or a permeative appearance. Ectatic lymphatics along the course of central lymphatics seen on MRI should raise the suspicion of this anomaly which is confirmed by lymphangiography.
Treatment in complex lymphatic anomalies is based on management of symptoms and controlling the growth of abnormal lymph vessels. Treatment options may include surgical procedures to drain excess fluid, medications to help control bleeding, and chemotherapy to help stabilize the condition. Current therapy is supportive with sirolimus as single agent or in combination with bisphosphonates.
[7] Sirolimus is an mTOR inhibitor which can prevent lymphangiogenesis and decrease lymphatic endothelial cell activity. [8]
Conclusion
GLA is a complex lymphatic anomaly having multiorgan involvement .The presence of characteristic osseous lesions along with blood parameters may help to distinguish these from other complex lymphatic anomalies such as KLA, GSD and CCLA. Histopathology is important for diagnosis however often it is not feasible due to patient refusal of invasive procedure or the risks of refractory effusions. In such settings the characteristic imaging appearances in the correct clinical scenario can be highly suggestive of this entity.
List of abbreviations
GLA: Generalized lymphatic anomaly; KLA: Kaposiform lymphangiomatosis; GSD: Gorham stout disease; CCLA: central conducting lymphatic anomaly; CT: Computed tomography; MRI: Magnetic resonance imaging; mTOR: Mammalian target of rapamycin
References
- Trenor C, Chaudry G Complex lymphatic anomalies. Semin Pediatr Surg 23 (2014): 186-190.
- Ricci K, Iacobas I How we approach the diagnosis and management of complex lymphatic anomalies. Pediatr Blood Cancer.
- Ozeki M, Fujino A, Matsuoka K, Nosaka S, Kuroda T et al Clinical features and prognosis of generalized lymphatic anomaly, kaposiform lymphangiomatosis, and Gorham- Stout disease. Pediatr Blood Cancer 63 (2021): 832-838.
- Barclay S, Inman K, Luks V, McIntyre J, Al-Ibraheemi A et al A somatic activating NRAS variant associated with kaposiform lymphangiomatosis. Genet Med 21 (2019): 1517-1524.
- Chaudry G, Trenor C, Dickie B Lymphatic In: Trenor C, Adams D (eds) Vascular Anomalies. Springer Cham.
- Lala S, Mulliken J, Alomari A, Fishman S, Kozakewich H et al Gorham-Stout disease and generalized lymphatic anomaly-clinical, radiologic, and histologic differentiation. Skeletal Radiol 42 (2013): 917-924.
- Adams D, Ricci K Vascular anomalies: Diagnosis of complicated anomalies and new medical treatment options. Hematol Oncol Clin North Am 33 (2019): 455-470.
- Ozeki M, Asada R, Saito A, Hashimoto H, Fujimura T et al Efficacy and safety of sirolimus treatment for intractable lymphatic anomalies: a study protocol for an open-label, single-arm, multicenter, prospective study (SILA). Regen therapy 10: 84-91.
