Fecal Microbiome Diversity in Irritable Bowel Syndrome (IBS) Clinical Subtypes
Article Information
Jasmin Ramic1*, Irina Milovac2, Zoran Mavija3, Naida Lojo-Kadric1, Maida Hadzic1, Stojko Vidovic2, Beate Niesler4, Nikolas Dovrolis5, Maria Gazouli6, Naris Pojskic1, Lejla Pojskic1
1University of Sarajevo, Institute for Genetic Engineering and Biotechnology, Sarajevo, Bosnia and Herzegovina
2Medical Faculty, University of Banja Luka, Banja Luka, Bosnia and Herzegovina
3Department of Gastroenterology, Clinical Center of Republic of Srpska, Banja Luka, Bosnia and Herzegovina
4Institute of Human Genetics, Department of Human Molecular Genetics, University of Heidelberg, Germany
5Laboratory of Medical Biology, Department of Medicine, Democritus University of Thrace, Alexandroupolis, Greece
6Laboratory of Biology, Medical School, National and Kapodistrian University of Athens, Athens, Greece
*Corresponding Author: Jasmin Ramic, University of Sarajevo, Institute for Genetic Engineering and Biotechnology, Sarajevo, Bosnia and Herzegovina
Received: 09 November 2020; Accepted: 16 November 2020; Published: 05 February 2021
Citation:
Jasmin Ramic, Irina Milovac, Zoran Mavija, Naida Lojo-Kadric, Maida Hadzic, Stojko Vidovic, Beate Niesler, Nikolas Dovrolis, Maria Gazouli, Naris Pojskic, Lejla Pojskic. Fecal Microbiome Diversity in Irritable Bowel Syndrome (IBS) Clinical Subtypes. Archives of Clinical and Biomedical Research 5 (2021): 125-136.
Share at FacebookAbstract
Irritable bowel syndrome (IBS) is a functional gut brain gastrointestinal (GI) disorder, typically accompanied by constipation or diarrhea, usually without any organic evidence. The prevalence of IBS is rather high of about 10-15% (10, 1 % according to Rome III and 4, 1% according to Rome IV, Enck P. et al 2016, Sperber A.D. et al 2020, Black C.J. et al 2020) in the working population. Quality of life in patients with IBS is reduced and therefore a major obstacle to the normal physical and social wellbeing.
In intensified clinical research worldwide new pathogenic mechanisms of IBS are suggested, including intestinal dysbiois one of the critical contributing factors to onset or further development of IBS. Intestinal microbiome represents a real ecosystem of microorganisms and human GI tract lining cells. The diversity and composition of the GI microbiome may differ significantly inter- and intra-individually, depending on sex, age or physiological conditions (pregnancy, disease, etc).
Intestinal microbiome composition frequently changes in association with IBS symptoms, and the purpose of this study was to investigate if there is a clear relationship in microbial composition and relative abundance of microbial taxa in feces of persons diagnosed with IBS. Fecal microbiota profiling was done in a group of nine clinically confirmed IBS patients and 6 corresponding healthy controls, based on species specific 16s RNA gene. No statistically significant differences in Alpha and Beta diversity indices were found.
Keywords
IBS; Fecal microbiome; 16s rRNA gene; Dysbiosis
IBS articles; Fecal microbiome articles; 16s rRNA gene articles; Dysbiosis articles
Article Details
Introduction
Recent evidence has shown that irritable bowel syndrome (IBS) is not just a functional gastrointestinal (GI) disorder, but rather represents a prototypical gut-brain disorder [1]. IBS affects 10.1 % (Rome III) and 4.1 % (Rome IV) worldwide, and 70–75% of affected individuals are female [2-4]. Typical symptoms involve chronic gut distress and abdominal pain with altered bowel habits without evident organic cause [2,5]. Diagnostics of IBS is based on a patient symptom reporting according to the standardized criteria [1,6]. Clinical stratification and management of IBS is based on specific defecation pattern into the following clinical subtypes: diarrhea-predominant (also referred to as ‘IBS with diarrhea’; IBS-D), constipation-predominant (or ‘IBS with constipation’; IBS-C), a mixed subtype (IBS-M) and unsubtyped IBS (IBS-U) [2]. Anxiety and depression are common co-morbidities in IBS, reflecting the due to complex relationship between altered gut physiology, visceral sensation and psychological perceptions that are mediated via the brain–gut axis patients with IBS additionally experience somatic pain [7] as well as other somatic pain syndromes such as migraine, fibromyalgia and chronic fatigue syndrome are over-represented [3].
Environmental (extrinsic) factors such as psychological distress [8], diet, smoking habits, infectious gastroenteritis that results in post-infectious IBS (PI-IBS) [9] and measurable alterations in the gut microbiota produce a complex interaction with genetic variants dispersed in the human genome (SNP’s, CNV’s) (intrinsic factors) and lead to individual epigenetic prints. These factors might contribute to central and peripheral (neurobiological) intermediate phenotypes (affecting brain and enteric nervous system function), influence the brain–gut axis and consequently manifest as central, behavioral and gastrointestinal intermediate phenotypes, thereby predisposing to IBS and its co morbid conditions [10-13].
Culture-free, PCR based methods using 16S rRNA gene-specific primers have provided insight in the gut lining microbiome composition of in patients with various clinical subtypes of IBS. Dysbiosis in different functional gastrointestinal disorders (including IBS) as compared to healthy individuals is reflected in overall microbial density decrease and relative abundance of major clades – bacteriodetes and firmicutes. High-throughput sequencing techniques have made it possible to identify detected DNA sequences in the gut microbiome, which is a more direct approach than PCR and provides higher resolution taxonomic classification. The aim of our study, was to use the culture-independent and high resolution 16S rRNA of the fecal microbiomes of patients with IBS and of disease free volunteers from Bosnia and Herzegovina to detect IBS specific signatures.
Material and Methods
Stool sampling and Genomic DNA extraction.
Stool samples from six healthy volunteers and nine patients with IBS were taken according to Helsinki declaration and samples were stored at -80°C before genomic DNA was isolated with the DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA) and mechanical lysis. Fecal samples were incubated in ATL buffer with proteinase K, mechanically lysed with a FastPrep FP120 (MP Biomedical, Solon, OH), treated with RNaseA, and purified on Qiagen columns. One tube containing 10 µl of sterile water was extracted in parallel to detect possible contamination.
16S rRNA gene sequencing
Amplicon libraries for pyrosequencing were constructed using a forward primer containing the 454 primer B and the 515F 16S sequences and a reverse primer containing the 454 primer A and the 806R 16S sequences. Each 50 µl PCR reaction mix contained 50 ng template DNA, 1 U Platinum Taq High Fidelity polymerase and buffers (Invitrogen, Carlsbad, CA), 400 nM primer, 2 mM MgSO4, 0.2 mM dNTP mix and was amplified by 30 cycles of touch-down PCR (37). Duplicate PCR amplicons for each sample were pooled and purified with Agencourt AMPure XP (BeckmanCoulter Genomics, Danvers, MA). Barcoded amplicons were combined in equimolar ratios for pyrosequencing on a 454-FLX-Titanium Genome Sequencer (Roche 454 Life Sciences, Branford, CT). QIIME2 v.2020.2.0 [14] was used to cluster, quality control was done using the DADA2 algorithm [15] and assign taxonomies based on the SILVA r132 database [16] to the raw sequences.
Statistical analyses of microbiome data
All analyses of the taxonomized sequences were performed on the MicrobiomeAnalyst [17] online platform. Total Sum Scaling (TSS) was applied to the data but no further transformations before the downstream analysis. Alpha-diversity was calculated using the CHAO1 index and ANOVA on the OTU level. Beta-diversity was calculated and visualized using Non-metric multidimensional scaling (NMDS) and Principal Coordinate Analysis (PCoA) on the OTU level. Correlation analysis was carried out using Spearman’s correlation algorithm. Mann-Whitney U test was used to compare differences between IBS and healthy controls and variance in taxonomic distribution between clinical subtypes (Med Calc v.19.0.4).
Results
Identification of the bacterial DNA was carried in a way that will amplify species-specific parts of bacterial DNA using array technique. Quantity of extracted total DNA varied form 2-21 ng/µl. Average of 780 190 to 1 063 311 sequence reads at genus level were detected from stool samples of IBS patients and healthy controls, respectively.
The number and proportion of successful readings at the genus level were given for each individual sample. Number of identified bacterial species per sample and Shannon species diversity index for each sample were similar in both studied groups and they are shown in Table 1.
Table1: Results of reading classification at genus level and values of Shannon species diversity index
|
Sample No. |
Sample ID |
Number Reads PF |
% Reads Classified to Genus |
Number of Species Identified |
Shannon Species Diversity index |
|
1 |
P1 |
989772 |
95.70 % |
504 |
2.176 |
|
2 |
P2 |
1041379 |
95.58 % |
591 |
2.029 |
|
3 |
P3 |
838990 |
93.27 % |
529 |
1.843 |
|
4 |
P4 |
963706 |
96.81 % |
516 |
0.810 |
|
5 |
P5 |
283370 |
97.39 % |
522 |
1.521 |
|
6 |
P6 |
917349 |
94.64 % |
639 |
2.052 |
|
7 |
P7 |
236754 |
97.58 % |
734 |
2.622 |
|
8 |
P8 |
968109 |
96.36 % |
540 |
2.349 |
|
9 |
P9 |
782277 |
95.87 % |
531 |
2.498 |
|
10 |
K1 |
1057237 |
97.46 % |
594 |
2.383 |
|
11 |
K2 |
1272946 |
95.77 % |
424 |
1.397 |
|
12 |
K3 |
884924 |
93.91 % |
646 |
2.386 |
|
13 |
K4 |
1031704 |
97.78 % |
424 |
2.464 |
|
14 |
K5 |
1072739 |
96.32 % |
618 |
2.164 |
|
15 |
K6 |
1060313 |
90.88 % |
583 |
2.481 |
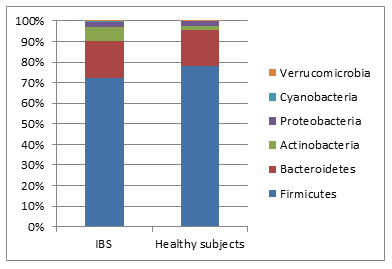
Based on diversity estimates (Shannon and Chao diversity indices) the overall microbiome diversity was reduced in IBS as compared to healthy controls (Graph 1).
Graph 1- Relative proportion of major phyla in IBS and healthy individuals (Mann-Whitney U test between groups P>0.05)
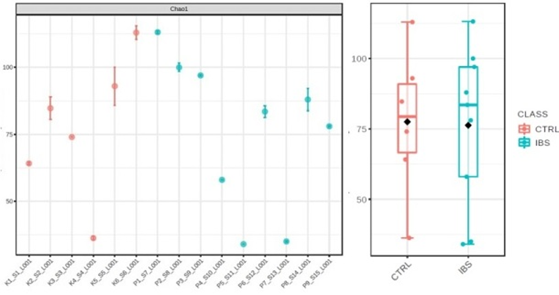
Alpha diversity index, which represent the average species diversity in individual samples (Figure 1a) or group (Figure 1b) did not shown significant difference on either level.
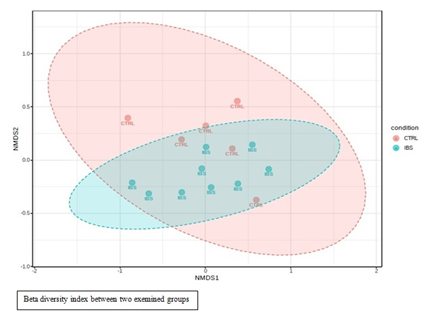
Beta diversity index which refers to the ratio of determined species between IBS group and control group is pictured on Figure 2. No statistically significant difference was observed in microbiota composition between IBS-patients and healthy control group.
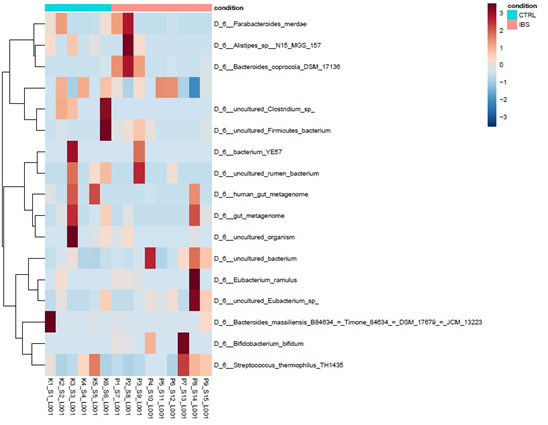
Grouping after correlation analysis of bacterial composition of two examined group, where we also didn’t find significant correlation, is represented on Figure 3.
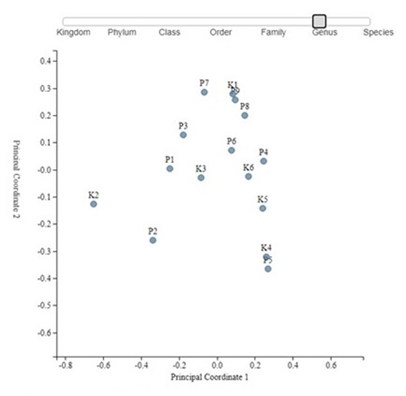
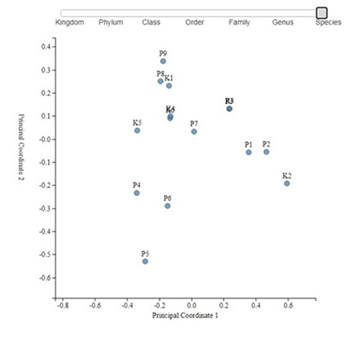
Principal Coordinate Analysis (PCoA) which is used for measuring of differences in the distribution of taxonomic classifications between samples, up to a fixed taxonomic level also didn't show any clustering of samples from different group neither on genus of species level (Figure 4 and 5).
Most frequently found genus’s in control groups were Blautia, Faecalibacterium, Coprococcus, and Ruminococcus and in the IBS patient group Blautia, Faecalibacterium, Bifidobacterium and Ruminococcus.
The observed difference in taxonomic distributionbetween IBS and healthy controls nor between clinical subtypes at level of phylum was not statistically significant (Mann Whitney test P>0.05) (Graph 1).
Discussion
The gut microbiome represents a specific ecosystem with a large number of different bacteria, fungi and archaea interacting in a more or less complicated manner. Those relations depends, like in every other ecosystem, of multiple intrinsic and external factors such are diet, lifestyle, smoking and alcohol consumption, use of drugs (probiotics, antibiotics, etc.), genetic factors, physiological conditions (age, pregnancy, surgery etc.) and interrelationships of microorganisms which habituate gut. Major lifetime event in GI physiology is the microbial colonization of human gastrointetinal tract, which is considered as basis for later normal developement and growth of other bacterial, fungal and archea species [18].
Alpha (average species diversity in given ecosystem) and beta diversity (refers to the ratio of all determined species) which are measure of species diversity, represent well established system for monitoring state of ecosystems. But, specific relations between bacterial species, physiological condition of human gut, and their interrelationships maybe can better be explained through ratio of bacterial species which are represents of specific condition [19]. In our study a slightly higher number of bacterial species was found in the patient group compared to the control group, although when the average diversity of bacterial species was observed, no statistically significant difference was found between the two groups.
The mechanism of emergence of IBS can in great part be explained by the changes and imbalance in microbiome composition (dysbiosis), in both composition and number of microo rganisms, that lead to activation of the immune system and eventually, inflammation [20;21]. The results of our study did not reveal any plausible link between stool microbiome and generalized IBS symptoms, nor specific IBS subtypes. Having in mind literature about changes in intestinal disorders [22-24], it was expected that there was a greater possibility of a statistically significant difference in microbiome composition between patients with IBS and the control group of subjects.
Our results did not confirm the expected thesis that there will be a difference in both the number and type, of bacterial organisms detected in the fecal product of subjects from the two groups. The abundance of individual bacterial families, genus's and species is in accordance with the literature [25], however, at no classification level was found a statistically significant difference between the observed groups. The obvious conclusion for such a result is that there is no difference in the quantitative and qualitative composition of the microbiome of patients with IBS compared to healthy subjects from our samples what maybe can be attributed to small sample size. However, results like this are also not unknown in the literature dealing with the relevant field. In their work Hughert W.L et al., 2019, didn't find distinct microbial signature differences in patient with IBS and randomly taken control samples in Swedish population. Also in recent Meta studies is found that the diversity of the microbiota was either decreased or not different in IBS patients compared with controls [26].
Also, other factors can modulate composition of gut microbiome in IBS patient. It is suggested that environmental factors such as nutritional habits, antibiotic usage, and lifestyle (stress, smoking habits, and inactivity) probably have a greater influence on the gut microbiome than genetics [27]. In our study we did not have data about IBS patient therapy, but it is well known that antibiotic usage may also influenced composition and numbers of species in gut microbiome.
Besides that there is a growing evidence of the involvement of fungal biome (mycobiome) alterations in IBS patients. Authors also conclude that development of visceral hypersensitivity indicates fungi dysbiosis may have indispensable role in IBS pathogenesis [28]. One of the possible reasons for the absence of a statistically significant difference in the number and type of microorganisms present in the collected fecal sample of the two examined groups, and in our opinion the most likely, may be the small sample size. This project is conceived as a study in which appropriate methods of fecal sampling, isolation and quantification of isolated DNA, its amplification, and detection of bacterial species present in the microbiome at the quantitative and qualitative level, would ultimately be reliably selected.
The method of DNA isolation from the fecal sample itself was successful, despite the fact that it showed considerable inconsistency on the quantitative level in terms of the final DNA concentration. The difference in the initial amount of the DNA template of individual samples, which enters the reaction, can affect the final results. Mentioning the latter, it should be noted that inequality in DNA isolation results was equally present in both groups. DNA purification and amplification, and sequencing of specific 16s regions of individual bacterial organisms at the quantitative and qualitative level have proven to be a very good and reliable method for microbiome analysis. The previously listed methods that have proven successful should be applied to future research that would be conducted on a larger number of respondents, which would certainly mean greater statistical reliability of the results.
Conclusions
A number of studies of GI tract microbiome in healthy and non-healthy individuals suggest both quality and quantity of microbiome content play important role in normal physiology of GI tract and overall individual health. The diversity and quantitative content of gut microbiome is changed in IBS, which is necessary in understanding complex IBS etiology and potential new target in treatment of IBS symptoms. In our study a no significant difference was found in composition or in number of individual bacterial species in gut microbiome between two examined groups. However, our results indicate that geographical distribution and local nutritional habits might play an important role in overall predisposition to the effects microbiota content changes and IBS symptoms.
Acknowledgements: This work is partly supported by a research grant of Federal Ministry of Science and Education of Bosnia and Herzegovina No. 05-39-2579-1/18. This manuscript results in part from collaboration and network activities promoted under the frame of the international network GENIEUR (Genes in Irritable Bowel Syndrome Research Network Europe), which has been funded by the COST program (BM1106, www.GENIEUR.eu) and is currently supported by the European Society of Neurogastroenterology and Motility (ESNM, www.ESNM.eu).
References:
- Lacy BE, Mearin F, Chang L, et al. Bowel disorders. Gastroenterology 150 (2016): 1393-1407.
- Enck P, Aziz Q, Barbara G, et al. Irritable bowel syndrome. Nature Reviews Disease Primers 2 (2016): 16014.
- Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome Foundation global study. Gastroenterology 160 (2020): 99-114.
- Black CJ, Ford AC. Global burden of irritable bowel syndrome: trends, predictions and risk factors. Nature Reviews Gastroenterology & Hepatology (2020): 1-4.
- Azpiroz F, Bouin M, Camilleri M, et al. Mechanisms of hypersensitivity in IBS and functional disorders. Neurogastroenterology & Motility 19 (2007): 62-88.
- Longstreth GF, Thompson WG, Chey WD, et al. Functional bowel disorders. Gastroenterology 130 (2006): 1480-1491.
- Spiller RC, Humes DJ, Campbell E, et al. The Patient Health Questionnaire 12 Somatic Symptom scale as a predictor of symptom severity and consulting behaviour in patients with irritable bowel syndrome and symptomatic diverticular disease. Alimentary Pharmacology & Therapeutics 32 (2010): 811-820.
- Larauche M, Mulak A, Taché Y. Stress and visceral pain: from animal models to clinical therapies. Experimental Neurology 233 (2012): 49-67.
- Spiller RC. Role of infection in irritable bowel syndrome. Journal of Gastroenterology 42 (2007): 41-47.
- Mayer EA. Gut feelings: the emerging biology of gut–brain communication. Nature Reviews Neuroscience 12 (2011): 453-466.
- Öhman L, Törnblom H, Simrén M. Crosstalk at the mucosal border: importance of the gut microenvironment in IBS. Nature Reviews Gastroenterology & Hepatology 12 (2015): 36-49.
- Fukudo S, Kanazawa M. Gene, environment, and brain-gut interactions in irritable bowel syndrome. Journal of Gastroenterology and Hepatology 26 (2011): 110-115.
- Gazouli M, Wouters MM, Kapur-Pojskic L, et al. Lessons learned—resolving the enigma of genetic factors in IBS. Nature Reviews Gastroenterology & Hepatology 13 (2016): 77-87.
- Bolyen E, Rideout JR, Dillon MR, et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nature Biotechnology 37 (2019): 852-857.
- Callahan BJ, McMurdie PJ, Rosen MJ, et al. DADA2: high-resolution sample inference from Illumina amplicon data. Nature Methods 13 (2016): 581-583.
- Quast C, Pruesse E, Yilmaz P, et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Research 41 (2012): D590-D596.
- Dhariwal A, Chong J, Habib S, et al. MicrobiomeAnalyst: a web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Research 45 (2017): W180-W188.
- Xiao L, Ding G, Ding Y, et al. Effect of probiotics on digestibility and immunity in infants: A study protocol for a randomized controlled trial. Medicine 96 (2017).
- Kristensen NB, Bryrup T, Allin KH, et al. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: a systematic review of randomized controlled trials. Genome Medicine 8 (2016): 52.
- Öhman L, Simrén M. Pathogenesis of IBS: role of inflammation, immunity and neuroimmune interactions. Nature reviews Gastroenterology & Hepatology 7 (2010): 163.
- Rajilic-Stojanovic M, Jonkers DM, Salonen A, et al. Intestinal microbiota and diet in IBS: causes, consequences, or epiphenomena?. The American Journal of Gastroenterology 110 (2015): 278.
- Distrutti E, Monaldi L, Ricci P, et al. Gut microbiota role in irritable bowel syndrome: New therapeutic strategies. World Journal of Gastroenterology 22 (2016): 2219.
- Halvorson HA, Schlett CD, Riddle MS. Postinfectious irritable bowel syndrome—a meta-analysis. American Journal of Gastroenterology 101 (2006): 1894-1899.
- Labus JS, Hollister EB, Jacobs J, et al. Differences in gut microbial composition correlate with regional brain volumes in irritable bowel syndrome. Microbiome 5 (2017): 49.
- Almeida A, Mitchell AL, Boland M, et al. A new genomic blueprint of the human gut microbiota. Nature 568 (2019): 499-504.
- Pittayanon R, Lau JT, Yuan Y, et al. Gut microbiota in patients with irritable bowel syndrome—a systematic review. Gastroenterology 157 (2019): 97-108.
- Rothschild D, Weissbrod O, Barkan E, et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555 (2018): 210-215.
- Botschuijver S, Roeselers G, Levin E, et al. Intestinal fungal dysbiosis is associated with visceral hypersensitivity in patients with irritable bowel syndrome and rats. Gastroenterology 153 (2017): 1026-1039.
- Drossman DA, Camilleri M, Mayer EA, et al. AGA technical review on irritable bowel syndrome. Gastroenterology 123 (2002): 2108-2131.
- North CS, Downs D, Clouse RE, et al. The presentation of irritable bowel syndrome in the context of somatization disorder. Clinical Gastroenterology and Hepatology 2 (2004): 787-795.
- Frissora CL, Koch KL. Symptom overlap and comorbidity of irritable bowel syndrome with other conditions. Current Gastroenterology Reports 7 (2005): 264-271.
- Cole JA, Rothman KJ, Cabral HJ, et al. Migraine, fibromyalgia, and depression among people with IBS: a prevalence study. BMC Gastroenterology 6 (2006): 26.
- Boeckxstaens GE, Drug V, Dumitrascu D, et al. Phenotyping of subjects for large scale studies on patients with IBS. Neurogastroenterology & Motility 28 (2016): 1134-1147.
- Hugerth LW, Andreasson A, Talley NJ, et al. No distinct microbiome signature of irritable bowel syndrome found in a Swedish random population. Gut 69 (2020): 1076-1084.