Drug Resistance in Mycobacterium tuberculosis and its Impact on Modern Medicine
Article Information
Ifrah Amjad1,*, Saba Riaz2
1Institute of Microbiology and Molecular Genetics (MMG), University of the Punjab, Lahore, Pakistan
2Assistant Professor, Institute of Microbiology and Molecular Genetics (MMG), University of the Punjab, Lahore, Pakistan
*Corresponding author: Ifrah Amjad, Institute of Microbiology and Molecular Genetics (MMG), University of the Punjab, Lahore, Pakistan.
Received: 07 January 2022; Accepted: 14 January 2022; Published: 21 January 2022
Citation: Ifrah Amjad, Saba Riaz. Drug Resistance in Mycobacterium tuberculosis and its Impact on Modern Medicine. Archives of Microbiology and Immunology 6 (2022): 51-64
Share at FacebookAbstract
Throughout the history, there has been a constant war between host and microorganisms. Microorganisms have noteworthy amplitude to offer resistance to antimicrobial agents and becoming an awful public health plight. The rising probability of infections is attributed to the ability of microbes conferring drug resistance. Drug resistance is a horrible capacity of a microbe to continue growing even in the presence of drug that is normally destined to limit its growth. As a result, there is a dwindling effect on the treatment efficacy to a disease. Drug resistance is awarded at the lethal concentration to a microbe and that particular concentration should be non-lethal to a human being. Mycobacterium tuberculosis is a greatest discovery of Robert Koch and it offers resistance to first-line and second-line antibiotics. Apprehending the mode of action of resistance for tuberculosis is crucial for advancement in the field of medical microbiology and for developing death-dealing antimicrobial drugs to minimize infections and mortality. Drug resistance is a major concern as it can lead to treatment failure and adjoins burden on healthcare costs. Next-generation sequencing technologies have helped to comprehend drug resistance. In this paper, we present the attributes of drug resistance of Mycobacterium tuberculosis and its impact on public health and challenges to modern medicine along with its epidemiology, mode of action, clinical factors and multi-drug resistance.
Keywords
First-line and second-line drugs, Minimum Inhibitory concentration, Acquired and natural resistance, BCG vaccine, MDR-TB, XDR-TB
Article Details
1. Introduction
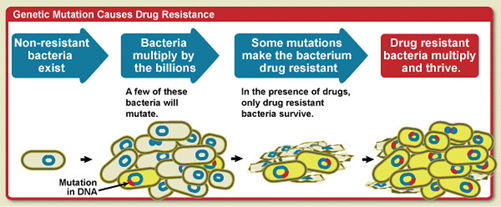
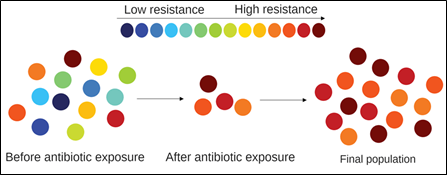
What brings home a dawn of drug resistance and major challenge to medication? Of course, it is an evolutionary change that comes to light a pressure of survival. Drug resistance is poked by every sort of living creation ranging from the field of agriculture to the domain of microbiology. If talking about molecular level, drug resistance is linked to mutations, deletions and gene amplifications. Antiquely, the challenge of drug resistance was first understood in the case of HIV-I infections. Mycobacterium is a strict aerobe, acid-fast rod1 and contains mycolic acids in cell wall. Mycobacterium tuberculosis offers 8 million cases per year which render death of 3 million people per year. Hence, it’s a need of hour to pay a high attention to its medication and resistance mechanisms. Tuberculosis is a complicated disease caused by Mycobacterium tuberculosis and it is linked to HIV infections. Immuno compromised conditions2 are developed due to HIV which increases the incidence of TB infection. Resistance simply means that a particular drug or an antibiotic is not rendering an effect on the killing of a microbe and hence a drug is no more lethal. When a drug is not killing a microbe according to normal dosage schedule, it is said to be a “resistant”. In this way, a drug falls out of minimum inhibitory concentration3 range. In my opinion:
“A smallest of the creations; a microbe achieves mastery over the noblest of the creations by being ineffective to a drug which is man-made. Hence, the creator is the most inventive.”
2. rimary or Innate Origin of Drug Resistance
Bacteria have an intrinsic property to confer resistance to drugs by different mechanisms. Salmonellae become resistant to a drug if it is infecting a host at that site where the drug cannot penetrate anymore. In gram-negative bacteria, the presence of an outer-membrane acts as a road-block for the inhibitory dose of a drug. Mycobacterium tuberculosis sometimes remain in a dormant state, where is becomes resistant to a drug phenotypically. For a drug to be effective enough, a target cell must have a specific structure. If a microbe de-orients its specific desired morphology, a drug is no more susceptible [1].
3. Acquired Genetic Drug Resistance
Bacteria exhibit drug resistance either through mutations4 or either gene transfer. In chromosome-aided resistance, a microbe becomes resistant due to two factors. It may change its structure or the transport systems are affected due to which a drug cannot reach to the site of efficacy5. Mycobacterium tuberculosis causes hindrance in the treatment of TB due to its unique feature i.e. Multi-drug resistance. Its thick and waxy cell wall containing mycolic acid calls for virulence and the affinity of degrading enzymes to the drugs serves as a barrier for drug susceptibility and hence piles up the hurdles in modern medicine and treatment of the contagious disease.
In the case of gene transfer in Mycobacterium tuberculosis, streptomycin confers resistance. Those mutants that are resistant to streptomycin are multiplied and these genes are then transferred to the progeny.
4. Biochemical Drug Resistance
Bacteria produce certain enzymes to establish drug resistance. Mycobacterium fortuitum causes chronic osteomyelitis6. However, it is resistant to many antibiotics, beta-lactam7 antibiotics and aminoglycosides. Mycobacterium fortuitum was isolated from the bone marrow of the patient. It was found that this bacterium produced beta-lactamase and aminoglycoside acyltransferase that actually conferred resistance. It can establish resistance to kanamycin as well after an enzymatic reaction of 40 minutes [3].
5. Beta-Lactam Drug Resistance
Beta-lactam drugs are resistant to most bacteria in different bacterial hosts. Eschericia coli K-12 and Salmonella typhi have R-factors that exhibit resistance to beta-lactam drugs by their beta-lactamase activity. Proteus species have less beta-lactamase activity as compared to others. Serratia marcescens falls into two groups on the basis of their resistance to beta-lactam antibiotics. Some strains are resistant to cephalosporins and other strains are resistant to penicillin. This difference is due to the production of specific types of enzymes. The former category produces cephalosporinase and the latter category produces penicillinase. The unique feature of Serratia marcescens is that it can transfer its ability to produce enzymes to E.coli and Klebsiella penumoniae [4].
6. Drug Resistance in Mycobacterium tuberculosis
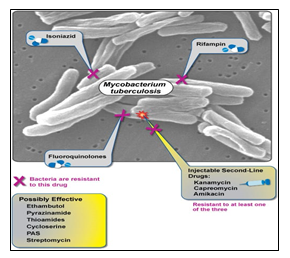
Tuberculosis has become serious and deadly disease due to the drug resistant strains. 25% of the world’s population is a victim of tuberculosis with 2 million deaths each year. Due to HIV, immune system gets weak and TB gets a greater chance to spread. Unfortunately, Mycobacterium tuberculosis has become resistant to all the first-line8 and second line antibiotics9 and hence posing a serious threat to modern medicine. In the recent cases, it has shown resistance to ethambutol (EMB), Rifampicin (RIF), Isoniazid (INH), Pyrazinamide (PZA) and Streptomycin (SM). For this reason, it is said to be MDR-TB (Multi-drug resistant-TB).
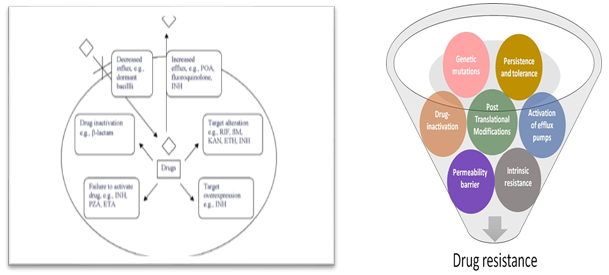
Drug resistance in Mycobacterium tuberculosis is divided into 5 groups. These mechanisms can be either primary or acquired as described above. (i) Decreased impenetrability (ii) High efflux (iii) Suppression in enzyme activity. (iv)Change in the target structure (v) Reduced pro-drug enzyme function.
Mycobacterium tuberculosis is assigned as MDR-TB (Multi-drug resistant TB) when it shows resistance to INH and RIF. Usually, it shows resistance by acquired mechanism exhibiting mutations in DNA. It has been reported that plasmids and gene transfer are not involved in conferring drug resistance [5].
6.1 Mode of Action and Mode of Resistance of First-Line Antibiotics in Mycobacterium tuberculosis
Table 1: First line antibiotics for the treatment of Mycobacterium tuberculosis along with the mode of action of drugs, their MIC and their mode of resistance.
|
Antibiotic |
Mode of Action |
Mode of Resistance |
|
Isoniazid (INH) |
MIC value for isoniazid is 0.25µg/mL. It acts only aerobic conditions at 37°C. It is activated by catalase-peroxidase enzyme (KatG) resulting in radicals that destroy target cell. The most sensitive target is InhA which directs mycolic acid synthesis. |
Resistance to the drug is acquired by the mutations (Ser315Thr mutation and mutation in InhA strucrural gene) or deletions in KatG gene. Mutations in InhA also provide resistance to ethionamide. |
|
Rifampicin (RIF) |
It has MIC of 0.5-3 µg/mL Kills log phase as well as stationary phase tuberculosis bacilli. It inhibits RNA production by adhering to RNA polymerase. It ensures treatment within 8 months. |
Resistance to the drug is due to mutation in an 81 base pair region in rpoB gene (point mutation) which encodes DNA-dependent RNA polymerase β- subunit. Mutations occur in two codons, Ser531 and His526. |
|
Pyrazinamide (PZA) |
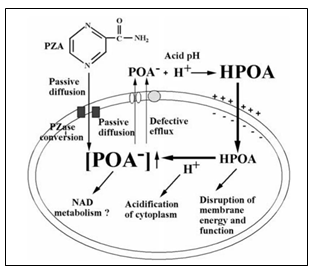
It acts only at acidic pH with MIC (Minimum inhibitory concentration) of 40µg/mL. It kills 80% of bacilli within one and half week incubation. It has no effect on mycolic acid biosynethesis. PZase converts PZA into POA upon entrance into the cell. POA is an anion that is moved outside of the cell forming HPOA which again gets an entry into the cell and disrupts the membrane and energy metabolism |
Resistance to a drug is due to mutation in pncA gene. Clustering occurs in three places of amino-acid residues i.e. 3-17, 61-85, 132-142 that inhabits the site of action for PZase. There are genetic insertions or mutations (Frameshift mutations) in pncA gene as well as His57Asp substitution. |
|
Streptomycin (STM) |
It reports for 85% drug resistance in TB infection. It has a deep impact on transcriptional as well as translational level. It causes misreading of the codons as well as inhibits protein synthesis as it caps the 30S subunit. It acts at alkaline pH with MIC (Minimum inhibitory concentration) in range of 6-7 µg/mL. |
Resistance to a drug is due to mutations in 16SrRNA as well as S12 (rpsL) which encodes ribosomal protein S12. 16SrRNA mutation unsettles a pseudoknot structure associated with S12. These call for 70% resistance in clinical isolates from patients. In rrs gene, there is an insertion of T in place of C at position 491 which accounts for misreading of the codons. |
|
Ethambutol (EMB) |
EMB acts only against the bacilli in log phase wich MIC 3 µg/mL. It hinders the formation of cell wall components formation i.e. lipoarabinomannan. It does so by inhibiting the activity of arabinosyl tranferase that converts arabinose into arabinan. |
Resistance to the drug is associated with embCAB operon encompassing point mutation, These mutations play role in inhibition of cell wall biosynthesis. [5,6,7,8,9] |
6.2 Second-Line Drug Resistance in Mycobacterium tuberculosis
6.2.1 Fluoroquinolones
Fluoroquinolones are usually associated with the inhibition of DNA synthesis by interacting with DNA gyrases and topoisomerases [10]. Fluoroquinolones are fairly active against TB bacilli with Minimum Inhibitory Concentration (MIC) of 2 µg/mL. It is a group of different antibiotics like ofloxacin, levofloxacin and ciprofloxacin. Their mode of action is such that they target DNA gyrases and suppress supercoiling of DNA. No matter how, Mycobacterium tuberculosis has shown resistance to ciproflaxin and ofloxacin due to mutation in 40th amino-acid in gyrA site. In some clinical reports, mutations in operons have also been reported. However, mutations in topoisomerase10 have not any impact on the activity of quinolone on Mycobacterium tuberculosis.
6.2.2 Ethionamide
Ethionamide (ETH) is a pro-drug that is activated upon the action of an enzyme, EthA (monooxygenase11). ETH and INH are analogous to each other. Mutations in ethA and inhA offer resistance to a drug [11]. MIC is 1-2 µg/mL. ethA inhibits inhA which interferes with the production of mycolic acid. If there is any mutation in ethA, Mycobacterium becomes resistant. Second-line therapy i.e. Thioamide ethionamide is given after TB bacilli offers resistance to INH and RIF (First-line drugs) [12].
7. Challenges to Modern Medicine Caused by Drug Resistance in Mycobacterium tuberculosis
Mycobacterium tuberculosis embraces many different mechanisms to offer drug resistance and hence provide challenges in the treatment of Tuberculosis regarding the modern medicine. It undergoes master plans to survive in different host lesions. No matter, effective medication for Mycobacterium tuberculosis is available but many patients fail to survive because of latent forms of bacilli. Due to dormancy, patients show no symptoms and are at greater risk of life-threatening infection. Cases of MDR-TB (Multi-drug resistant TB) and XDR-TB (Extremely drug resistant TB) provide challenges to modern medicine as they confer resistance to even combinations of different drugs like INH and RIF. Due to this feature, lower recovery rates have been observed and mortality rate has been elevated since 21st century. [13] The rising challenges in treatment have increased the insight into the development of more combinational drugs with a major viewpoint to lessen the time of treatment. The co-infections of HIV-TB are the significant hurdle in the treatment because of damaging drug-drug interactions [14]. In the present era, many new challenges are emerging regarding MDR-TB that poses a large burden on finances.
7.1 Analytical Framework for the Challenges and Control of Tuberculosis
The rising challenges in modern medicine have led to the development of different analytical work plans by Shiffman and Smith (2007), Clark (2000) and McAdam (1990). This framework contains four sets i.e. actor, ideas, political context, and issue traits [15].
Table 2: Shiffman and Smith analytical framework after rising challenges to modern medicine while treating tuberculosis
|
CATEGORY |
FACTORS |
|||
|
Actor |
Policy community coherence among discussants.
|
|||
|
Idea |
|
|||
|
Policy environment |
|
|||
|
Issue traits |
|
7.2 Other Challenges
Proper services related to care and facilities in treatment have also put in the major challenges in the treatment. It has been long manifested that along with multi-drug resistance by Mycobacterium tuberculosis, poverty is the utmost challenge for providing the patients with the most effective medicines due to high costs [16].
8. The Who Global Project on Drug-Resistance Surveillance
Global Project on drug resistance Surveillance was commenced in the year 1994. With the rising time, the cases of MDR-TB were increasing in different regions of the world. In 2003, World Health Organization developed a scheme to prevent the drug resistance in TB by executing the direct observed therapy strategy (DOTS) [17]. Tuberculosis is sorted as a second most infectious disease after HIV. The available BCG vaccine12 has many limitations and modification is needed [18]. Since the development of DOTS strategy, a lot of progress has been made in TB control.
9. Toxic and Inefficient Drugs for Mycobacterium tuberculosis
Apprehending the mechanism of drug resistance is very imperative to deal with drug resistance. Drug resistance is achieved after mutations that render the treatment in effective. Resistance to anti-tuberculosis drugs occurs at a frequency of 3.5×10-6 for INH and 3.1×10-8 for RIF [19].
9.1 Drug Resistance by D-Cycloserine
Cycloserine13 has an impact on the cell wall of Mycobacterium tuberculosis and it aims at alanine racemase and alanine ligase. It is regarded as group 4 bacteriostatic second-line antibiotic. Its alarming feature is that it can cause serious side effects including nausea, damage to liver, arthralgia and hypokalemia [20]. According to the study (Safety assessment) conducted in China on Cycloserine intake and its side effects, the following table is giving the summary: [20]
Table 3: Side effects of cycloserine along with the doses given to the patient
|
Patient ID |
Age |
Daily cycloserine doses |
Internal time (days) |
Side effects |
||
|
P9 |
48 |
500milligram |
119 |
Abdominal swelling |
||
|
P80 |
52 |
750milligram |
115 |
Headache |
||
|
P21 |
39 |
250milligram |
110 |
Seizure
|
9.2 Other Peptides
9.2.1 Viomycin
Viomycin belongs to a group of antibiotics known as Tuberactinomycin. It interferes with the elongation phase of Mycobacterium tuberculosis by blocking elongation factor i.e. EF-G. It hampers the ribosome for almost 40 seconds. It also causes misreading of these codons [21].
9.2.2 Capreomycin (CAP)
The extremely resistant TB bacterium confers resistance to CAP and is associated with high mortality rates. This is accompanied by mutations in rrs gene14 leading to a change from G to T change at codon 1473 [22].
10. Molecular Methods for Detection of Drug Resistance in Mycobacterium tuberculosis
DIAGNOSTICS FOR TB
Mycobacterium tuberculosis divides slowly and it is multi-drug resistant bacteria. Therefore, many different nucleic-acid based techniques have been developed to check the mutations.
10.1 Real time PCR technique
Two main perspectives are seen regarding RT-PCR. One is the use of non-specific dyes that are fluorescent and the other one is a probe that is having a specific melting temperature (Tm). If there is any change in Tm, mutations can be detected in target. Two types of probes are used in Tuberculosis. Sloppy molecular beacon probes (SMB) and Dual labeled linear probes (DLPs). SMB detects mutations in RIF resistance determining region. DLP consists of a single strand oligonucleotide that has two dyes. Both SMB and DLP target the Mycobacterium tuberculosis rrs, gyrA, rpoB genes and those mutations that are present in these genes are detected by fluorescence [23, 24].
10.2 LATE-PCR with Lights-On/Lights Off probes
To detect the substitutions in nucleotides Linear-after-the-exponential-PCR is used with lights-on and lights-off probes. Lights-on probe is labeled to a fluorescent dye and lights-off probe is labeled to a quencher dye. As a result of melting temperature, ssDNA binds to a probe with lowest Tm15 and fluorescence is emitted. If mutations are present in the target sequence, they can be detected. Interesting feature is that in LATE-PCR different colors of probes can be used that are used to detect different mutations in base pairs. [23, 25]
10.3 Sequencing
The first complete genome sequence of Mycobacterium tuberculosis was done in 1998 by Cole encoded by 4000 genes and 4.2 million base pairs. Next generation sequencing (NGS) can detect mutations, deletions and substitutions. The genes that can be amplified and sequenced for MDR-TB are rpsL, katG, inhA, embB, rpoB etc. Mutations which are present in these genes are detected by NGS. [23, 26]. Many other techniques are available for diagnostics of drug resistance in Mycobacterium tuberculosis like digital PCR, Anyplex II MTB/MDR and MTB/XDR and GENEDRIVE MTB/RIF ID KIT.
11- MDR-TB (Multi-Drug Resistant TB)
In the present era, MDR-TB has become a weighty factor that is posing a high burden on the treatment of tuberculosis. By the year 2012, there were new cases of MDR-TB with 1 lac and seventy thousands death globally.
Mycobacterium tuberculosis is assigned as multi-drug resistant if it shows resistance to isoniazid and rifampicin
MDR-TB was driven initially through hospital-acquired infections among Human Immunodeficiency Virus (HIV) patients. MDR-TB has a fear of relapsing after the treatment by first-line antibiotics. After MDR-TB patients are identified, medication should be given according to MDR- Regimen as shown in table 6.
Table 3: MDR-TB regimen in which drugs are given on the basis of efficacy on the patients [27].
|
Step 1 |
Injectable drug such as Kanamycin is given |
Drug should be chosen on the basis of patient’s history. For MDR-TB patients, Streptomycin is not given. |
|
Step 2 |
Levofloxacin or moxifloxacin is given (Fluoroquinolones) |
Before giving fluoroquinolones, patients for XDR-TB must be diagnosed. |
|
Step 3 |
Ethionamide and cycloserine should be given. |
Second-line drugs should be effective for such patients. |
|
Step 4 |
Pyrazinamide or ethambutol can be given. |
Patient should be tolerant to them. |
|
Step 5 |
Amoxillin, linezolid, ioszianid can be given. (Group 5) |
If above drugs are not effective, group 5 drugs are given. |
- XDR-TB (Extremely Drug Resistant TB)
XDR-TB is a turning out to be major public health threat. Treatment for XDR-TB patients are extended to two years. In HIV infected patients, the recovery rates are only 30-40% [28, 29].
XDR-TB is resistant to isozianid and rifampicin and to fluoroquinolones as well.
13. Conclusion
I conclude that tuberculosis has become the most deleterious disease because of its prevalence in HIV patients, high cost treatment and nosocomial transfer. The diagnosis of Tuberculosis has become difficult because of somehow asymptomatic conditions in many patients. Due to MDR- and XDR TB, it is significantly becoming more difficult to treat tuberculosis and hence posing a great threat to modern medicine. WHO has brought forward many initiatives to tackle with the disease and awareness regarding its high spread all over the world? Tuberculosis has ranked as a second most deadly disease because of its high prevalence. TB control is too necessary as TB can be ANYWHERE or Everywhere. DOTS strategy has proven to be most productive to eradicate tuberculosis but it demands the authorities’ support in terms of financial aid, medicine and personnel equipment. It’s easier spread is a reckoning hallmark whilst its treatment and due to faster spread it has become almost impossible to eradicate the disease in poor countries. But! To a satisfying version if proper medication is followed and WHO guidelines regarding exposure to the disease are regarded then I must say, “Tuberculosis is preventable as well as curable”.
[1]Resistance to de colorization by acids.
2Low immune system defenses
3Minimum concentration of a drug to inhibit bacterial growth after overnight incubation
4Change in the structure of a gene.
5Site where it can act and destroy bacteria.
6Bone destruction accompanied by inflammation.
7Having beta-lactam rings.
8The first treatment given for a disease.
9Therapy given after first-line antibiotics
10Alters the supercoiling of DNA.
11Involved to catalyze O from O2 into inorganic substrates
12Bacillus Calmette-Guerin vaccine against tuberculosis
13Analog of alanine.
14Part of 16SrRNA gene.
15Tm is the temperature at which 50% dsDNA is converted to ssDNA.
References
- Tejaswini Sastry. Drug resistance (2013).
- Rebecca Clements, David Mangus. Antibiotic resistance and bacterial evolution (2018).
- Udou T, Mizuguchi Y, Yamada T. Biochemical mechanisms of antibiotic resistance in a clinical isolate of Mycobacterium fortuitum. Presence of beta-lactamase and aminoglycoside-acetyltransferase and possible participation of altered drug transport on the resistance mechanism 133 (1986): 653-7.
- Edmund Farrar W Jr, Noel M. O'Dell, β-Lactamases and Resistance to Penicillin and Cephalosporins inSerratia marcescens,The Journal of Infectious Diseases 134 (1976): 245-251.
- Mary Margaret Wade and Ying Zhang. Frontiers in Bioscience 9 (2004): 975-994.
- Khan MT, Malik SI, Ali S,et al.Pyrazinamide resistance and mutations inpncAamong isolates ofMycobacterium tuberculosisfrom Khyber Pakhtunkhwa, Pakistan.BMC Infect Dis19 (2019):116.
- Honoré N, Cole ST. Streptomycin resistance in Mycobacterium tuberculosis. Antimicrobial Agents Chemother. 38 (1994): 238-42.
- Finken M, Kirschner P, Meier A, Wrede A, Böttger EC. Molecular basis of streptomycin resistance in Mycobacterium tuberculosis: alterations of the ribosomal protein S12 gene and point mutations within a functional 16S ribosomal RNA pseudoknot. Mol Microbiol 9 (1993): 1239-46.
- Richard T, Ellison. Determining the mechanism of action of ethambutol, Journal Watch, MD, reviewing Zhang L et al (2020).
- Hooper DC. Mode of action of fluoroquinolones. Drugs 58 (1999): 6-10.
- Glenn P. Morlock, Beverly Metchock and Robert C.Cooksey. ethA, inhHA and katG loci of Ethionamide-Resistant Mycobacterium tuberculosis Antimicrobial agents chemother 47 (2003): 3799-3805.
- Andrea E. DeBarber,Khisimuzi Mdluli,Marlein Bosman,Linda-Gail Bekker, andClifton E. Barry 97 (2000): 9677-9682.
- Adeeb Shehzad, Gauhar Rehman, Mazhar Ul-Islam, Waleed Ahmad Khattak, Young Sup Lee, Challenges in the development of drugs for the treatment of tuberculosis, The Brazilian Journal of Infectious Diseases 17 (2013): 74-81.
- Ginsberg A, Spigelman M. Challenges in tuberculosis drug research and developmentNat Med13 (2007):290-294.
- Haruka Sakamoto,Sangnim Lee,Aya Ishizuka,Eiji Hinoshita,Hiroyuki Hori,Nanao Ishibash, Kenichi Komada,Masataro Norizuki,Yasushi Katsuma, Hidechika Akashi&Kenji Shibuya. hallenges and opportunities for eliminating tuberculosis - leveraging political momentum of the UN high-level meeting on tuberculosis. BMC Public Health19 (2019):76.
- Zhang P,Xu G,Song Y,Tan J,Chen T,Deng G. 2020 Challenges Faced by Multidrug-Resistant Tuberculosis Patients in Three Financially Affluent Chinese Cities (2020).
- Rabia Johnson, Elizabeth M.Streicher, Gail E.Louw, Robin M.Warren, Paul D. van Helden and Thomas Victor. Drug Resistance in Mycobacterium tuberculosis. Current issues in Molecular Biology 8 (2006): 97-112.
- Denkinger C, kik SV, Pai M. Expert Rev. Mol. Diagn. Rapid diagnosis of Mycobacterium tuberculosis with Truenat TB: A near care approach 13 (2013):763-767.
- Sebastian M. Gygli, Sonia Borrell, Andrej Trauner, Sebastien Gagneux, Antimicrobial resistance inMycobacterium tuberculosis: mechanistic and evolutionary perspectives,FEMS Microbiology Reviews. 41 (2017): 354-373.
- Yang Li, Fei Wang, Limin Wu, Min Zhu, Guiqing He, Xinchang Chen, Feng Sun, Qihui Liu, Xiaomeng Wang, Wenhong Zhang. Cycloserine for treatment of multi-drug resistant tuberculosis 12 (2019): 721-731.
- Mikael Holm, Anneli Borg, Mans Ehrenberg and Suparna Sanyal. Molecular mechanism of viomycin inhibition of peptide elongation in bacteria 113 (2016): 978-983.
- Max R. O’Donnell, Melendhran Pillay,4Manormoney Pillay,Lise Werner,Iqbal Master,Allison Wolf, Barun Mathema, Yacoob M Coovadia, Koleka Mlisana,. Robert Horsburgh, andNesri Padayatchi. Primary capreomycin resistance is common, and associated with early mortality in extensively drug-resistant tuberculosis (XDR-TB) patients in KwaZulu-Natal, South Africa 69 (2015): 536-543.
- Thi Ngoc Anh Nguyen, Véronique Anton-Le Berre4, Anne-Laure Bañulsand?Thi Van Anh Nguyen Molecular Diagnosis of Drug-Resistant Tuberculosis; A Literature Review (2019).
- Sandy S. Roh,Laura E. Smith,Jong Seok Lee,Laura E. Via,Clifton E. Barry III,David Alland,Soumitesh Chakravorty. Comparative Evaluation of Sloppy Molecular Beacon and Dual-Labeled Probe Melting Temperature Assays to Identify Mutations inMycobacterium tuberculosisResulting in Rifampin, Fluoroquinolone and Aminoglycoside Resistance (2016).
- K.Jayalakshmi,MansiRathore,GirishPai,DattatrayJoshi. Real time polymerase chain reaction (RT-PCR) based rapid detection of multi-drug resistant (MDR) Mycobacterium tuberculosis (MTB). European Respiratory Journal38 (2011):p4394.
- Keira A. Cohen,Abigail L. Manson,Christopher A. Desjardins,Thomas Abeel&Ashlee M. Earl. Deciphering drug resistance inMycobacterium tuberculosisusing whole-genome sequencing: progress, promise, and challenges. Genome Medicine11 (2019):45.
- Kwonjune J. Seung, Salmaan Keshavjee, andMichael L. Rich. Multidrug-Resistant Tuberculosis and Extensively Drug-Resistant Tuberculosis. Cold Spring Harb Perspect Med.2015 Sep; 5(9): a017863.
- LoBue P. Extensively drug-resistant tuberculosis. Curr Opin Infect Dis 22 (2009): 167-73.
- Anthony PMol. N Sarita Shah. Extensively drug-resistant tuberculosis (XDR-TB) (2009).
- Houben, Rein MGJ, Dodd, Peter J, Metcalfe, John Z."The Global Burden of Latent Tuberculosis Infection: A Re-estimation Using MathematicalModelling".Medicine13(2016):