Development and Validation of Stability Indicating HPLC Method for Benzalkonium Chloride in Betaxolol (0.5%) Opthalmic Solution
Article Information
Liena Alagib H Edress, Dhia Eldin Elhag*
University of Medical Sciences and Technology, UMST, Khartoum, Sudan
*Corresponding Author: Dhia Eldin Elhag, University of Medical Sciences and Technology, UMST, Khartoum, Sudan
Received: 03 March 2019; Accepted: 18 April 2019; Published: 24 April 2019
Citation: Liena Alagib H Edress, Dhia Eldin Elhag. Development and Validation of Stability Indicating HPLC Method for Benzalkonium Chloride in Betaxolol (0.5%) Opthalmic Solution. J Pharm Pharmacol Res 3 (2019): 019-027.
Share at FacebookAbstract
Background: Preservative assay in eye drops require the development of a suitable validated method for the assay.
Objective: This study was conducted to develop and validate stability indicating High Performance Liquid Chromatography (HPLC) method for assay of benzalonium chloride in betaxolol 0.5% ophthalmic solution.
Method: Method was developed validated according to International conference on harmonization (ICH) guideline. Chromatographic condition used was: L10 CN column (250 cm × 4.6 mm × 10 μm); flow rate 2.0 ml per minute; detection wavelength 245 nm; column oven 30°C; mobile phase: (0.1M sodium acetate, acetonitrile) (55:45 v/v) and injection volume 100 μL.
Results: The method was found fulfilling the ICH requirement with R2 of 0.9995 for drug, Limit of detection (LOD) of 4.53 μg/ml, and Limit of quantification (LOQ) of 13.75 for benzalkonium chloride. The mean of overall recovery % was found to be 99.96% and the repeatability results was found to be: 99.95% for benzalkonium chloride. The overall intermediate precision results were found to be 100.77%. The method proved to be accurate, precise and specific.
Conclusion: The developed method was found to be simple, sensitive and can be used for routine quality control analysis of benzalkonium chloride in betaxolol (0.5%) ophthalmic solutions.
Keywords
HPLC; Method development; Method validation; Benzalkonium chloride; Betaxolol ICH Guidelines
Article Details
1. Introduction
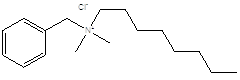
Benzalkonium chloride is a mixture of alkyl benzyl dimethyl ammonium chlorides of the general formula [C6H5CH2N(CH3)2R]Cl, where R represents a mixture of alkyls, including all or some of the group beginning with n-C8H17 and extending through higher homologs, with n-C12H25, n-C14H29, and n-C16H33 comprising the major portion. The average molecular weight of benzalkonium chloride is 360.The chemical structure is shown in Figure 1.
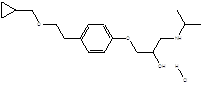
Benzalkonium chloride is used in pharmaceutical formulations as antimicrobial and in eye drops as a preservative at a concentration of 0.01-0.02% w/v [1-5]. Often it is used in combination with other preservatives or excipients, particularly 0.1% w/v disodium edetate, to enhance its antimicrobial activity against strains of Pseudomonas. In addition to this, it is used as a preservative in cosmetics [6, 7]. Betaxolol hydrochloride is a white, crystalline powder, soluble in water, with a molecular weight of 343.89. The chemical structure is present in Figure 2.
Betaxolol chloride in ophthalmic preparations has been shown to be effective in lowering intraocular pressure and is indicated in the treatment of ocular hypertension and chronic open-angle glaucoma [8, 9]. It may be used alone or in combination with other anti-glaucoma. Numerous authors described the determination of benzalkonium chloride in pharmaceutical ophthalmic preparations by using high performance liquid chromatography, HPLC/MS, capillary electrophoresis and TLC densitometry but most of their work focused on HPLC [10-15]. In this work we present a simple robust, accurate and precise method for the determination of benzalkonium chloride in betaxolol (0.5%) ophthalmic solutions.
2. Materials and Methods
2.1 Chemicals
Benzalkonium chloride working standard (100% purity) obtained from Merck, Germany. Acetonitrile and glacial acetic acid (HPLC Grade, SDFCL). Sodium acetate, disodium edetate sodium chloride (Charlo Erba) Purified water is. Ltd. Betxalol standard (99% purity) and betaxolol 0.5% ophthalmic solution samples. Obtained from Bash Pharma, Co Sudan.
2.2 Instruments
Analysis was performed on High Performance Liquid Chromatography-HPLC (SHIMADZU, JAPAN) equipped with UFLC line pump (model LC-20AB) and Prominence auto sampler (model SIL-20AC), Column L10, CN (250 mm × 4.6, 10 µm). Prominence UV/VIS Detector (model SPD-20AV), Prominence Degassing Unit (mode DGU-20A 3 R) and column oven (model CTO-20A). Mettler Toledo Balance MS model 1050 DU (Switzerland). Ultrasonicater (Model Elmasonic S80, Germany). Nylon Filter 0.45 µm. Data acquisition was made with SHIMADZU LC-Solution software.
2.3 Liquid chromatographic conditions
Injection volume 100 µL, flow rate; 2.0 mL/ minute, detection wavelength of 254 nm;column oven 30°C mobile phase (0.1M sodium acetate, acetonitrile (55:45 v/v).
2.4 Methods
2.4.1 Mobile phase preparation: Exactly 0.1M Sodium acetate buffer powder was prepared and the pH was adjusted to 5.00 with glacial acetic Acid. The mobile phase was prepared by mixing 0.1 M sodium acetate buffer and acetonitrile (45% 55%, v/v). The mixture was filtered and degassed for 10 minutes by sonication.
2.4.2 Benzalkonium chloride standard stock solution: Exactly 100 mg of benzalkonium chloride standard were accurately weighed and transferred into a 100 ml volumetric flask; the volume was completed up to mark using purified water and sonicated for 1 minute to produce a solution having a concentration of 1000 µg/ml.
2.4.3 Preparation of placebo solution: The solution containing betaxolol excipients (disodium edetate sodium chloride) excluding benzalkonium chloride is used as placebo.
3. Results and Discussion
3.1 System suitability test
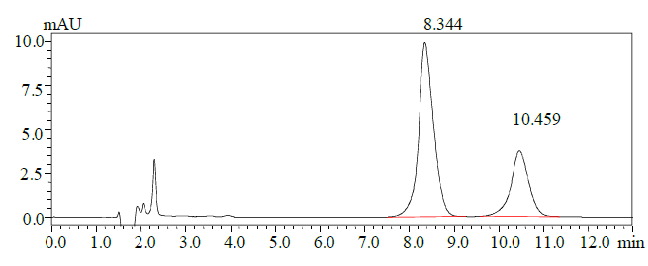
A stock solution of benzalkonium chloride of a concentration of 200 µg/ml was prepared and was injected five times and the results obtained are shown in Table 1 and the obtained chromatogram is shown in Figure 3.
|
Injection # |
Ret. Time |
Sum of Peak Area |
Theo. Plate |
Tailing Factor |
|||
|
C12 |
C14 |
C12 |
C14 |
C12 |
C14 |
||
|
1 |
8.2 |
10.3 |
209856 |
3555 |
3686 |
1.058 |
0.954 |
|
2 |
8.2 |
10.3 |
209775 |
3629 |
3859 |
1.058 |
0.954 |
|
3 |
8.2 |
10.3 |
210369 |
3620 |
3882 |
1.057 |
0.951 |
|
4 |
8.2 |
10.2 |
209576 |
3624 |
3896 |
1.058 |
0.96 |
|
5 |
8.1 |
10.2 |
209485 |
3628 |
3878 |
1.057 |
0.95 |
|
Average |
8.18 |
10.26 |
209812.2 |
3611 |
3840 |
1.058 |
0.954 |
|
STDEV |
0.04 |
0.06 |
345 |
31.62 |
87.21 |
0.001 |
0.004 |
|
RSD% |
0.55 |
0.53 |
0.16 |
0.88 |
2.3 |
0.05 |
0.41 |
Table 1: Results of System suitability test.
3.2 Stability of solution
A benzalkonium chloride solution (200ug/ml) was injected into HPLC system as fresh sample. and then was injected after 6 hours and after 24 hours. A fresh test solution was prepared and analysed after six hours and 24 hours. At each time, the sample was analyzed five times. Interady RSD% were 0.19, 33 and 33%. RSD% of sum peaks area of fresh injected working standard of benzalkonium chloride, after 6 hours and after 24 hours was calculated and found to be 0.28%.
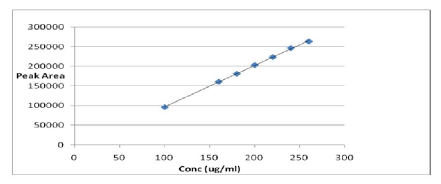
3.3 Linearity
A series of seven concentrating levels (100.15-160.24, 180.27, 200.30, 220.33, 240.36 and 260.39 µg/ml were prepared from the stock solution (1000 µg/ml) and the solutions were measured and the calibration curve was plotted. The regression equation obtained was:
Y=1051.6x - 8405.4, R2=0.9995
3.4 Limit of detection and limit of quantification
The limit of detection (LOD) and the limit of quantification (LOQ) of the benzalkonium chloride were calculated
according to ICH guidelines. The values obtained were 4.53 µg/ml and 13.75 µg/ml, respectively.
3.5 Specificity
Placebo of the betaxolol (0.5%) ophthalmic solution, equivalent to the sample volume was taken and solution prepared and analysed. No interferents peaks were shown in the obtained chromatogram.
3.6 Accuracy
Three different quantities (low, medium and high i.e. 80%, 100% and 130% of the standard test solution) of the authentic standard were prepared and injected in triplicate for each spike level. The results obtained were acceptable (acceptance recovery criteria % is 98%-102 %). See Table 2.
|
Conc. |
80% |
100% |
130% |
|
Avg. assay.(n=3) |
79.95% |
100.23% |
129.61% |
|
Avg. recovery |
99.94% |
100.23% |
99.70% |
|
RSD% |
1.57 |
1.56 |
0.73 |
Table 2: Results of the recovery study.
3.7 Precision
Study of the precision of the assay was determined by repeatability (intra-day) and intermediate precision (inter-day) in triplicate. Repeatability was evaluated by assaying of eight determinations at 100% of the test concentrations. Intermediate precision was assessed by comparing the assay of eight determinations at 100% of the test concentrations on different days (3 days) prepared in the same manner for repeatability. The RSD% obtained was <2.
3.7.1 Intra-day precision:
|
Day 1 |
AverageAssay (n=3) |
STDEV |
RSD% |
|
99.95 |
0.398 |
0.40 |
|
|
Day 2 |
100.39% |
0.328 |
0.33 |
|
Day 3 |
101.96% |
0.594 |
0.58 |
Table 3: Benzalkonium chloride results of the intra-day precision test (repeatability).
3.7.2 Intermediate precision: The overall intermediate precision of the method is shown in Table 4. The results were within the acceptable range, i.e. RSD ≤ 2.
|
Days |
Avg. Assay |
STDEV |
RSD% |
|
Day 1 |
99.95 |
1.056 |
1.05 |
|
Day 2 |
100.39 |
||
|
Day 3 |
101.96 |
||
|
Overall average |
100.77 |
Table 4: Overall intermediate precision results.
3.8 Robustness
The Robustness was determined by injecting triplicate injections of standard solution. The parameters tested were, flow rate, column oven temperature and detection wavelength mobile phase pH. A summary of Robustness Parameters is given in Table 5.
|
Flow rate |
Flow rate 1.8 |
Flow rate 2.2 |
||
|
C12 |
C14 |
C12 |
C14 |
|
|
Average area |
224248 |
180432.5 |
||
|
Retention time |
9.6 |
12.11 |
8.3 |
10.5 |
|
Tailing factor |
1.01 |
0.9 |
0.94 |
0.87 |
|
Resolution |
- |
3.34 |
- |
3.34 |
|
Theoretical plate |
3477.16 |
3727.16 |
3192.26 |
3457.44 |
|
Average Assay |
100.14 |
98.94 |
||
|
RSD% |
0.01 |
0.41 |
||
|
Mobile phase |
Mobile phase pH 5.2 |
Mobile phase pH 4.8 |
||
|
C12 |
C14 |
C12 |
C14 |
|
|
Average area |
201732 |
202461 |
||
|
Retention time |
8.7 |
10.8 |
8.3 |
10.4 |
|
Tailing factor |
1.0 |
0.9 |
1.0 |
0.9 |
|
Resolution |
- |
3.37 |
- |
3.48 |
|
Theoretical plate |
3723.39 |
3937.23 |
3689.8 |
3906.75 |
|
Assay |
102.4 |
102.86 |
||
|
RSD% |
0.83 |
0.97 |
||
|
Oven Temp |
Column oven 28°C |
Column oven 32°C |
||
|
C12 |
C14 |
C12 |
C14 |
|
|
Average area |
206112 |
221596 |
||
|
Retention time |
9.0 |
11.5 |
8.6 |
10.8 |
|
Tailing factor |
0.9 |
0.8 |
0.9 |
0.8 |
|
Resolution |
- |
4.0 |
- |
4.0 |
|
Theoretical plate |
24337 |
25904 |
26178 |
28189 |
|
Assay |
100.8 |
100.4 |
||
|
RSD% |
0.07 |
0.21 |
||
|
Wavelength |
Wavelength 252 nm |
Wavelength 256 nm |
||
|
C12 |
C14 |
C12 |
C14 |
|
|
Average area |
184293 |
231035 |
||
|
Retention time |
8.6 |
10.9 |
8.6 |
10.9 |
|
Tailing factor |
0.9 |
0.8 |
0.9 |
0.8 |
|
Resolution |
- |
4.0 |
- |
4.0 |
|
Theoretical plate |
24587 |
26305 |
24529 |
26170 |
|
Assay |
101.5 |
101.3 |
||
|
RSD% |
0.56 |
0.42 |
||
Table 5: Summary of robustness test results.
3.9 Forced degradation studies
Forced degradation was studied by a deliberate degradation through exposure of betaxolol eye drops 0.5% to acid hydrolysis, base hydrolysis, photo degradation (UV, 254nm), heat and oxidation as follows; 5 ml of betaxolol eye drops solution was transferred to 100-ml volumetric flask; 5 ml of 1M HCl was added, mixed and allowed to stand for 60 minutes at room temperature. The volume was completed up to mark using placebo and sonicated for 1 minute, the solution mixed, filtered and injected into the HPLC system. The same procedure was repeated with the addition of 5 ml of 0.01M NaOH for base hydrolysis, or 5 ml of 30% H2O2 (oxidation) or exposure to UV radiation for photo-degradation study.
For the effect of heat study, 5 ml of betaxolol 0.5% ophthalmic solution was transferred to 100-ml volumetric flask and exposed for dry heat (oven) at 25, 35, 45, 55 and 65°C. Then the volume was completed up to mark using placebo and sonicated for 1 minute, mixed, filtered and injected into the HPLC system. The assay of the drug was 99.32%. The results of the drug assay after subjection the drug to the above mentioned stress conditions are shown in Table 6.
|
Stress condition |
Acid |
Base |
Oxidation |
Thermal |
|
Assay |
95.46 |
47.44 |
97.6 |
99.11 |
Table 6: Summary of Forced degradation Study results.
4. ConclusionA simple, sensitive, cost- effective method for the determination of benzalkonium chloride in betaxolol 0.5%
ophthalmic solution was developed and validated. The developed method meets the ICH guidelines for method validation.
References
- Saisyo A, Shimono R, Oie S, Kimura K, Furukawa H. The Risk of Microbial Contamination in Multiple-Dose Preservative-Free Ophthalmic Preparations. Biol Pharm Bull 40 (2017): 182-186.
- Datta S, Baudouin C, Brignole-Baudouin F, Denoyer A, Cortopassi GA. The Eye Drops Preservative Benzalkonium Chloride Potently Induces Mitochondrial Dysfunction and Preferentially Affects LHON Mutant Cells. Invest Ophthalmol Vis Sci 58 (2017): 2406-2412.
- Chan Hew Wai A, Becasse P, Tworski S, Pradeau D, Planas V. Optimising concentrations of antimicrobial agents in pharmaceutical preparations: Case of an oral solution of glycerol and an ophthalmic solution containing cysteamine. Ann Pharm Fr 72 (2014): 415-421.
- Ryan G, Fain JM, Lovelace C, Gelotte KM. Effectiveness of ophthalmic solution preservatives: a comparison of latanoprost with 0.02% benzalkonium chloride and travoprost with the sofZia preservative system. BMC Ophthalmol 11 (2011): 8.
- Rasmussen CA, Kaufman PL, Kiland JA. Benzalkonium chloride and glaucoma. J Ocul Pharmacol Ther Off J Assoc Ocul Pharmacol Ther 30 (2014): 163-169.
- Choi SM, Roh TH, Lim DS, Kacew S, Kim HS, Lee B-M. Risk assessment of benzalkonium chloride in cosmetic products. J Toxicol Environ Health B Crit Rev 21 (2018): 8-23.
- Shah N, Naseby DC. Efficacy of benzalkonium chloride against bioluminescent P. aeruginosa ATCC9027 constructs. Biosens Bioelectron 97 (2017): 8-15.
- Huang W, Zhang N, Hua H, Liu T, Tang Y, Fu L, et al. Preparation, pharmacokinetics and pharmacodynamics of ophthalmic thermosensitive in situ hydrogel of betaxolol hydrochloride. Biomed Pharmacother 83 (2016): 107-113.
- Denis P, Demailly P, Saraux H. Clinical evaluation of betaxolol in ophthalmic suspension with or without preservative agent in patients with glaucoma or ocular hypertension. J Fr Ophtalmol 16 (1993): 297-303.
- Liu F, Xiao KP, Rustum AM. Determination of individual homologues and total content of benzalkonium chloride by reversed-phase high-performance liquid chromatography using a short butyl column. J AOAC Int 92 (2009): 1644-1651.
- Hou Y-H, Wu C-Y, Ding W-H. Development and validation of a capillary zone electrophoresis method for the determination of benzalkonium chlorides in ophthalmic solutions. J Chromatogr A 976 (2002): 207-213.
- Gomez-Gomar A, Gonzalez-Aubert MM, Garces-Torrents J, Costa-Segarra J. Determination of benzalkonium chloride in aqueous ophthalmic preparations by high-performance liquid chromatography. J Pharm Biomed Anal 8 (1990): 871-876.
- Kwiecień A, Krzek J, Walczak M, Mazur M. Development and validation of stability-indicating TLC-densitometric method for determination of betaxolol with LC-ESI/MS analysis of degradation product. Acta Pol Pharm 70 (2013): 643-652.
- Liu J, Deng W, Yu M, Wen R, Yao S, Chen B. Rapid analysis of benzalkonium chloride using paper spray mass spectrometry. J Pharm Biomed Anal 145 (2017): 151-157.
- Labranche L-P, Dumont SN, Levesque S, Carrier A. Rapid determination of total benzalkonium chloride content in ophthalmic formulation. J Pharm Biomed Anal 43 (2007): 989-993.