Development and Characterization of Alginate/Collagen Composite Bioinks for Bone Tissue Engineering
Article Information
Takaaki Arahira1,2, Kurumi Takimoto2, Mitsugu Todo3
1Faculty of Management and Information Science, Kyushu Institute of Information Sciences,6-3-1 Saifu, Dazaifu 818-0117, Japan
2School of Management and Information Science, Kyushu Institute of Information Sciences,6-3-1 Saifu, Dazaifu 818 0117, Japan
3Research Institute for Applied Mechanics, Kyushu University, 6-1 Kasuga-koen, Fukuoka 816-8580, Japan
*Corresponding author: Takaaki Arahira, Faculty of Management and Information Science, Kyushu Institute of Information Sciences, 6-3-1 Saifu, Dazaifu 818-0117, Japan.
Received: 25 April 2023; Accepted: 28 April 2023; Published: 03 May 2023
Citation: Takaaki Arahira, Kurumi Takimoto, Mitsugu Todo. Development and Characterization of Alginate/Collagen Composite Bioinks for Bone Tissue Engineering. Journal of Biotechnology and Biomedicine. 6 (2023): 160-162.
Share at FacebookAbstract
Tissue engineering aims to construct artificial organs based on cells, growth factors, and scaffold materials. Scaffolds should promote cellular activity, provide sufficient nutrition, and remove waste products. In recent years, 3D bioprinters, which can suspend cells using bioink to fabricate 3D structures, are attracting considerable attention as a technology that enables uniform cell distribution. However, few studies have examined the mechanical properties of 3D scaffold by bioink. This study aims to develop a bioink with good mechanical properties that is easy to produce. As the result, compared with commercially available bioinks, the fabricated bioink is easy to prepare by mixing Alg-Na with a collagen solution and has superior mechanical properties. Our results indicate that some material properties, such as compressive modulus and surface structure, can be controlled by varying the ratio of collagen.
Keywords
Composite bioink; Alginate; Collagen; Bioprinting; Mechanical property
Composite bioink articles Composite bioink Research articles Composite bioink review articles Composite bioink PubMed articles Composite bioink PubMed Central articles Composite bioink 2023 articles Composite bioink 2024 articles Composite bioink Scopus articles Composite bioink impact factor journals Composite bioink Scopus journals Composite bioink PubMed journals Composite bioink medical journals Composite bioink free journals Composite bioink best journals Composite bioink top journals Composite bioink free medical journals Composite bioink famous journals Composite bioink Google Scholar indexed journals Alginate articles Alginate Research articles Alginate review articles Alginate PubMed articles Alginate PubMed Central articles Alginate 2023 articles Alginate 2024 articles Alginate Scopus articles Alginate impact factor journals Alginate Scopus journals Alginate PubMed journals Alginate medical journals Alginate free journals Alginate best journals Alginate top journals Alginate free medical journals Alginate famous journals Alginate Google Scholar indexed journals Collagen articles Collagen Research articles Collagen review articles Collagen PubMed articles Collagen PubMed Central articles Collagen 2023 articles Collagen 2024 articles Collagen Scopus articles Collagen impact factor journals Collagen Scopus journals Collagen PubMed journals Collagen medical journals Collagen free journals Collagen best journals Collagen top journals Collagen free medical journals Collagen famous journals Collagen Google Scholar indexed journals Bioprinting articles Bioprinting Research articles Bioprinting review articles Bioprinting PubMed articles Bioprinting PubMed Central articles Bioprinting 2023 articles Bioprinting 2024 articles Bioprinting Scopus articles Bioprinting impact factor journals Bioprinting Scopus journals Bioprinting PubMed journals Bioprinting medical journals Bioprinting free journals Bioprinting best journals Bioprinting top journals Bioprinting free medical journals Bioprinting famous journals Bioprinting Google Scholar indexed journals Mechanical property articles Mechanical property Research articles Mechanical property review articles Mechanical property PubMed articles Mechanical property PubMed Central articles Mechanical property 2023 articles Mechanical property 2024 articles Mechanical property Scopus articles Mechanical property impact factor journals Mechanical property Scopus journals Mechanical property PubMed journals Mechanical property medical journals Mechanical property free journals Mechanical property best journals Mechanical property top journals Mechanical property free medical journals Mechanical property famous journals Mechanical property Google Scholar indexed journals nanofibers articles nanofibers Research articles nanofibers review articles nanofibers PubMed articles nanofibers PubMed Central articles nanofibers 2023 articles nanofibers 2024 articles nanofibers Scopus articles nanofibers impact factor journals nanofibers Scopus journals nanofibers PubMed journals nanofibers medical journals nanofibers free journals nanofibers best journals nanofibers top journals nanofibers free medical journals nanofibers famous journals nanofibers Google Scholar indexed journals cellulose articles cellulose Research articles cellulose review articles cellulose PubMed articles cellulose PubMed Central articles cellulose 2023 articles cellulose 2024 articles cellulose Scopus articles cellulose impact factor journals cellulose Scopus journals cellulose PubMed journals cellulose medical journals cellulose free journals cellulose best journals cellulose top journals cellulose free medical journals cellulose famous journals cellulose Google Scholar indexed journals bioink samples articles bioink samples Research articles bioink samples review articles bioink samples PubMed articles bioink samples PubMed Central articles bioink samples 2023 articles bioink samples 2024 articles bioink samples Scopus articles bioink samples impact factor journals bioink samples Scopus journals bioink samples PubMed journals bioink samples medical journals bioink samples free journals bioink samples best journals bioink samples top journals bioink samples free medical journals bioink samples famous journals bioink samples Google Scholar indexed journals organism articles organism Research articles organism review articles organism PubMed articles organism PubMed Central articles organism 2023 articles organism 2024 articles organism Scopus articles organism impact factor journals organism Scopus journals organism PubMed journals organism medical journals organism free journals organism best journals organism top journals organism free medical journals organism famous journals organism Google Scholar indexed journals artificial organs articles artificial organs Research articles artificial organs review articles artificial organs PubMed articles artificial organs PubMed Central articles artificial organs 2023 articles artificial organs 2024 articles artificial organs Scopus articles artificial organs impact factor journals artificial organs Scopus journals artificial organs PubMed journals artificial organs medical journals artificial organs free journals artificial organs best journals artificial organs top journals artificial organs free medical journals artificial organs famous journals artificial organs Google Scholar indexed journals
Article Details
Introduction
The treatment and regeneration of various organs have been realized owing to the development of regenerative medicine. In particular, research on tissue engineering, which is the foundation of bone regeneration therapy, has been increasing. Tissue engineering aims to construct artificial organs based on cells, growth factors, and scaffold materials [1]. Scaffolds promote cellular activity, provide sufficient nutrition, and remove waste products. Moreover, they have mechanical properties similar to the mechanical environment of the surrounding tissue, and are nonharmful to the organism. Additionally, several studies have demonstrated that a three-dimensional (3D) porous structure is suitable for application as a scaffold material structure. However, in cell cultures with conventional scaffolds [2], the cells are seeded on the fabricated scaffolds, resulting in a nonuniform cell distribution and inefficient organization. 3D bioprinters, which suspend cells with bioink for the fabrication of 3D structures, are attracting considerable attention as a technology that enables uniform cell distribution [3]. Currently, various organs, such as the heart [4], blood vessels [5], and skin [6], have been fabricated using 3D bioprinters. However, the use of these bioinks are limited owing to the amount and expensive cost. Moreover, few studies have evaluated the mechanical properties of 3D scaffolds prepared with bioink. The mechanical properties of scaffolds play a vital role in cell proliferation and differentiation[7]. Therefore, this study aims to develop a bioink with good mechanical properties that is easy to produce.
Material and Methods
Bioink (Bi) from CELLINK was used as a reference. Three types of original bioinks were synthesized as follows: a 4.0% sodium alginate (Alg-Na) solution (AC0) was prepared by dissolving Alg-Na powder in purified water at 37 °C for 2 days, and subsequently AC0 was mixed with a collagen solution (5.4 mg/g) at 10 vol% (AC10) and 30 vol% (AC30) (Figure. 1).

Figure 1: Preparation procedure for the bioink composites.
A 3D model (diameter, 12 mm; height, 3 mm) of the scaffold material was generated using the 3D Builder software, and converted to G-code using the Repetier-Host software. Subsequently, a 3D bioprinter (INKREDIBLE) was used to construct the 3D model, which was cross-linked using a 1.0% aqueous calcium chloride solution for 1 h (Figure.2).
Figure 2: Preparation procedure for the 3D scaffold from bioink.
Mechanical tests were performed on the fabricated scaffolds using a mechanical testing machine under a load of 500 N at 1 mm/min. Stress-strain curves were generated from the load and displacement results obtained from the mechanical tests. Stress (σ) and strain (ε) were evaluated using the following formulae:

where F is the load applied during the compressive tests, d is the scaffold diameter, L is the scaffold height, and ΔL is the displacement after loading at each time interval. The Young’s modulus was calculated from the slope of the stress-strain curve in the initial linear region (strain = 0.1–0.3). The compressive modulus were obtained by measuring four independent samples and are presented as mean values with the corresponding standard deviations (± SDs). One-way analysis of variance (ANOVA) was performed, and significant differences were calculated using the Fisher least significant difference (LSD) method (p < 0.05). The surface structure of the scaffolds were characterized by field-emission scanning electron microscopy (FE-SEM; S-4100, Hitachi Ltd., Japan). The scaffolds were mounted on aluminum stages and sputter-coated with platinum/palladium using a sputter coater (E-1030, Hitachi Ltd., Japan).
Results and Discussion
Figures 3 (a) and (b) show the stress-strain curves and compressive modulus of the scaffolds. As shown in Fig. 3 (a), the stress-strain curves exhibited the same trend for all the bioink samples. As shown in Fig. 3 (b), the compressive modulus for the Bi, AC0, AC10, and AC30 scaffolds were 105.7 ± 18.1, 136.6 ± 31.2, 151.4 ± 23.2, and 134.8 ± 10.0 kPa, respectively, which followed the trend of AC10 > AC0 > AC30 > Bi. The highest (AC10) and lowest (Bi) compressive modulus were significantly different. However, among the synthesized bioink composite samples (AC0, AC10, and AC30), AC30, which had the highest collagen content, had the lowest compressive modulus.
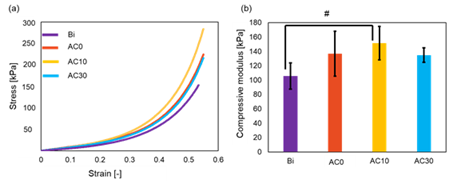
Figure 3: Mechanical properties of the scaffolds for the four bioink samples. (a) Stress-strain curves and (B) compressive modulus (# indicate p < 0.05).
Figure 4 shows FE-SEM micrographs of the scaffolds prepared with the four bioink samples. The surface structure of the scaffolds indicated the presence of fibers, which are thought to be cellulose nanofibers in the bioink. Collagen fibers were observed on the surfaces of AC10 and AC30.
In this study, the compressive modulus value was 167.8 kPa in AC0. In contrast, Kuo et al. reported that the compressive modulus of an alginate gel scaffold with a high Ca content was approximately 70 kPa [8]. Notably, the concentration of sodium alginate used in this study was effective for improving the compressive mechanical property. The surface structures of AC0 and AC30, which had similar compressive modulus, were also similar. The elastic modulus of AC0, AC10, and AC30 were identical. The concentrations of the Alg-Na solution in AC10 and AC30 were calculated to be 3.5% and 3.0%, respectively. Notably, a previous study reported that the Young's modulus of scaffolds prepared with alginic acid was proportional to the alginic acid concentration [9]. In this study, the Young's modulus did not proportionally increase with a decrease in the Alg-Na concentration in the bioink composite samples, although, AC10 (3.5% Alg-Na) had a higher Young's modulus than AC0 (4.0% Alg-Na). This may be owing to the presence of collagen fibers, which serve as a skeleton, balancing the Alg-Na concentration and maintaining the elastic modulus.
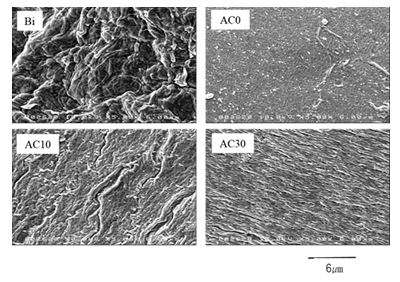
Figure 4: Field-emission scanning electron microscopy micrographs of the scaffolds for the four bioink samples.
Conclusion
The mechanical properties of scaffolds synthesized with Bi, AC0, AC10, and AC30 bioinks using a 3D bioprinter had the following trend in terms of the Young's modulus AC10 > AC0 > AC30 > Bi. Compared with commercially available bioinks, the AC30 bioink is easily prepared by mixing Alg-Na with a collagen solution and has superior mechanical properties. However, the Young's modulus of AC30 was lower than that of AC0, indicating that the use of the collagen solution resulted in a nonuniform Alg-Na concentration. The surface structures of AC10 and AC30 exhibited no collagen fibers, indicating that the collagen fiber content was insufficient. Therefore, it is necessary to improve the developed method by using solely collagen fibers as an alternative to the collagen solution. Moreover, inorganic materials, such as bioceramics, may be combined with the developed bioink in the future.
Acknowledgements
This work was supported by JSPS KAKENHI Grant Number 20K04169.
References
- Nakamura M, Ogura R, et al. Challenges in the fabrication of living organs by medical engineering: a challenging organ fabrication method Additive Organ Manufacturing. Organ Biology 24 (2017): 217-222.
- Arahira T, Todo M, Variation of mechanical behavior of β-TCP/collagen two phase composite scaffold with mesenchymal stem cell in vitro. Journal of the Mechanical Behavior of Biomedical Materials 61 (2016): 464-474.
- Caliari SR, Ramirez MA, et al. The development of collagen-GAG scaffold-membrane composites for tendon tissue engineering. Biomaterials 32 (2011): 8990-8998.
- Kazeroni ZS, Telloo M, et al. A mitral heart valve prototype using sustainable polyurethane polymer: fabricated by 3D bioprinter, tested by molecular dynamics simulation. Journal of Mechanical Engineering 5 (2012): 109-120.
- Kucukgul C, Ozler SB, et al. 3D bioprinting of biomimetic aortic vascular constructs with self-supporting cells. biotechnology and bioengineering 112 (2015): 811- 821.
- Roshangar L, Rad JS, et al. Using 3D-bioprinting scaffold loaded with adipose-derived stem cells to burns wound healing. Journal of tissue engineering and regenerative medicine 15 (2021): 546-555.
- Jiao C, Xie D, et al. Additive manufacturing of Bio-inspired ceramic bone Scaffolds: Structural Design, mechanical properties and biocompatibility. Materials & Design 217 (2022): 110610.
- Kuo CK, Ma PX. Maintaining dimensions and mechanical properties of ionically crosslinked alginate hydrogel scaffolds in vitro. Journal of Biomedical Materials Research Part A: An Official Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials 84 (2008): 899-907.
- Kaygusuz H, Evingür GA, et al. Surfactant and metal ion effects on the mechanical properties of alginate hydrogels. international journal of biological macromolecules 92 (2016): 220-224.
