Dermal Sinus Track with Intraspinal Dumbbell Mature Cystic Teratoma in an Infant Presenting as Recurrent Meningitis: A Case Report
Article Information
Ghanem Al Sulaiti1, Muhammad Mohsin Khan2, Ghaya Al rumaihi1, Ahmed EL Beltagi3, Issam El Bozom3, Doaa Mohammed Taha2, Samar Magboul5, Kazim Mohammed4*
1Senior Consultant Neurosurgery, Hamad Medical Corporation, Doha, Qatar
2Resident Neurosurgery, Hamad Medical Corporation, Doha, Qatar
3Department of Pathology, Hamad Medical Corporation, Doha, Qatar
4Specialist Neurosurgery, Hamad Medical Corporation, Doha, Qatar
5Resident Pediatric, Hamad Medical Corporation, Doha, Qatar
*Corresponding Author: Kazim Mohammed, Specialist neurosurgery, Hamad Medical Corporation, Doha, Qatar
Received: 26 July 2021; Accepted: 03 August 2021; Published: 31 August 2021
Citation:
Ghanem Al Sulaiti, Muhammad Mohsin Khan, Ghaya Al rumaihi, Ahmed EL Beltagi, Issam El Bozom, Doaa Mohammed Taha, Samar Magboul, Kazim Mohammed. Dermal Sinus Track with Intraspinal Dumbbell Mature Cystic Teratoma in an Infant Presenting as Recurrent Meningitis: A Case Report. Journal of Spine Research and Surgery 3 (2021): 099-105.
Share at FacebookAbstract
Cystic mature teratomas presenting in the spine are rare, comprising less than 0.1% of all spinal tumors. They arise from all three germ cell layers and usually accompany other congenital abnormalities. Spinal teratomas are more common in children as compared to adults and are usually associated with spinal dysraphism. Dermal Sinus is a closed type of Spinal dysraphsim, lined with epithelium and are mostly associated with Dermoid, Epidermoid and Posterior arch defect [1]. We present a case of an infant with recurrent meningitis. His examination was remarkable for a lumbar dimple. Imaging revealed a dermal sinus track continuous with a dumbbell appearance of extradural and intradural nodules, while the histopathology confirmed mature cystic Teratoma.
Keywords
Cystic Mature Teratomas, Infant
Cystic Mature Teratomas articles Cystic Mature Teratomas Research articles Cystic Mature Teratomas review articles Cystic Mature Teratomas PubMed articles Cystic Mature Teratomas PubMed Central articles Cystic Mature Teratomas 2023 articles Cystic Mature Teratomas 2024 articles Cystic Mature Teratomas Scopus articles Cystic Mature Teratomas impact factor journals Cystic Mature Teratomas Scopus journals Cystic Mature Teratomas PubMed journals Cystic Mature Teratomas medical journals Cystic Mature Teratomas free journals Cystic Mature Teratomas best journals Cystic Mature Teratomas top journals Cystic Mature Teratomas free medical journals Cystic Mature Teratomas famous journals Cystic Mature Teratomas Google Scholar indexed journals Infant articles Infant Research articles Infant review articles Infant PubMed articles Infant PubMed Central articles Infant 2023 articles Infant 2024 articles Infant Scopus articles Infant impact factor journals Infant Scopus journals Infant PubMed journals Infant medical journals Infant free journals Infant best journals Infant top journals Infant free medical journals Infant famous journals Infant Google Scholar indexed journals spinal dysraphism articles spinal dysraphism Research articles spinal dysraphism review articles spinal dysraphism PubMed articles spinal dysraphism PubMed Central articles spinal dysraphism 2023 articles spinal dysraphism 2024 articles spinal dysraphism Scopus articles spinal dysraphism impact factor journals spinal dysraphism Scopus journals spinal dysraphism PubMed journals spinal dysraphism medical journals spinal dysraphism free journals spinal dysraphism best journals spinal dysraphism top journals spinal dysraphism free medical journals spinal dysraphism famous journals spinal dysraphism Google Scholar indexed journals Dermal Sinus articles Dermal Sinus Research articles Dermal Sinus review articles Dermal Sinus PubMed articles Dermal Sinus PubMed Central articles Dermal Sinus 2023 articles Dermal Sinus 2024 articles Dermal Sinus Scopus articles Dermal Sinus impact factor journals Dermal Sinus Scopus journals Dermal Sinus PubMed journals Dermal Sinus medical journals Dermal Sinus free journals Dermal Sinus best journals Dermal Sinus top journals Dermal Sinus free medical journals Dermal Sinus famous journals Dermal Sinus Google Scholar indexed journals Intraspinal articles Intraspinal Research articles Intraspinal review articles Intraspinal PubMed articles Intraspinal PubMed Central articles Intraspinal 2023 articles Intraspinal 2024 articles Intraspinal Scopus articles Intraspinal impact factor journals Intraspinal Scopus journals Intraspinal PubMed journals Intraspinal medical journals Intraspinal free journals Intraspinal best journals Intraspinal top journals Intraspinal free medical journals Intraspinal famous journals Intraspinal Google Scholar indexed journals Meningitis articles Meningitis Research articles Meningitis review articles Meningitis PubMed articles Meningitis PubMed Central articles Meningitis 2023 articles Meningitis 2024 articles Meningitis Scopus articles Meningitis impact factor journals Meningitis Scopus journals Meningitis PubMed journals Meningitis medical journals Meningitis free journals Meningitis best journals Meningitis top journals Meningitis free medical journals Meningitis famous journals Meningitis Google Scholar indexed journals Neurosurgery articles Neurosurgery Research articles Neurosurgery review articles Neurosurgery PubMed articles Neurosurgery PubMed Central articles Neurosurgery 2023 articles Neurosurgery 2024 articles Neurosurgery Scopus articles Neurosurgery impact factor journals Neurosurgery Scopus journals Neurosurgery PubMed journals Neurosurgery medical journals Neurosurgery free journals Neurosurgery best journals Neurosurgery top journals Neurosurgery free medical journals Neurosurgery famous journals Neurosurgery Google Scholar indexed journals neuromotor deficits articles neuromotor deficits Research articles neuromotor deficits review articles neuromotor deficits PubMed articles neuromotor deficits PubMed Central articles neuromotor deficits 2023 articles neuromotor deficits 2024 articles neuromotor deficits Scopus articles neuromotor deficits impact factor journals neuromotor deficits Scopus journals neuromotor deficits PubMed journals neuromotor deficits medical journals neuromotor deficits free journals neuromotor deficits best journals neuromotor deficits top journals neuromotor deficits free medical journals neuromotor deficits famous journals neuromotor deficits Google Scholar indexed journals lumbosacral spine articles lumbosacral spine Research articles lumbosacral spine review articles lumbosacral spine PubMed articles lumbosacral spine PubMed Central articles lumbosacral spine 2023 articles lumbosacral spine 2024 articles lumbosacral spine Scopus articles lumbosacral spine impact factor journals lumbosacral spine Scopus journals lumbosacral spine PubMed journals lumbosacral spine medical journals lumbosacral spine free journals lumbosacral spine best journals lumbosacral spine top journals lumbosacral spine free medical journals lumbosacral spine famous journals lumbosacral spine Google Scholar indexed journals mid-lumbar articles mid-lumbar Research articles mid-lumbar review articles mid-lumbar PubMed articles mid-lumbar PubMed Central articles mid-lumbar 2023 articles mid-lumbar 2024 articles mid-lumbar Scopus articles mid-lumbar impact factor journals mid-lumbar Scopus journals mid-lumbar PubMed journals mid-lumbar medical journals mid-lumbar free journals mid-lumbar best journals mid-lumbar top journals mid-lumbar free medical journals mid-lumbar famous journals mid-lumbar Google Scholar indexed journals
Article Details
1. Case Presentation
A 7 months old child was a product of full 38 weeks of gestation and cesarian section (CS) delivery. Clinically the child was active, with expected growth milestones, moving all limbs, no neuromotor deficits. He had two dimples on his back, at S1/2 and another at the S4 area, with no other abnormality noticed. The patient had a history of left inguinal abscess at the age of 2 months, which was treated with drainage and IV antibiotics (Augmentin). At the age of 4 months, he presented with fever and decreased activity and was diagnosed with Klebsiella aerogenes meningitis sensitive to gentamicin and Meropenem, for which he was treated with 21 days IV Meropenem. The immunology workup was unremarkable and was declared immunocompetent. After two days of discharge, he was readmitted to Hospital, and CSF cultures grew Klebsiella aerogenes. The pediatric infection team continued him on IV Meropenem for six weeks and Amikacin as synergistic for two weeks. Repeat CSF after two weeks turned negative. However, while on IV antibiotics at the 4th week, he had a fever spike, and repeat CSF regrew the same organism. MRI head, dorsal, and Lumbosacral spine was done. MRI head showed meningeal thickening and enhancement, and MRI spine showed thickening and enhancement of cauda equina nerve roots. Further targeted 3D T2 series MRI (3D-T2-Space) with multiplanar reformats displayed to advantage a subcutaneous dermal sinus tract connecting the cutaneous dimple to a rounded extradural lesion which was connected by a tubular tract which continued into the intradural compartment coursing cranially to end in a similar-appearing intradural rounded lesion giving an intraspinal dumbbell appearance (Figure 1 and 2).
Inview of the recurrent infections, MRI evidence of spinal dysraphism with intraspinal extra- and intra-dural lesion, and after discussion with the parents, it was decided to excise the dermal sinus tract, which was believed to be the culprit of repeated meningitis. Other than two dimples at the S1/2 and S4 regions, no obvious deformity was noted on the back. Intraoperatively, elliptical skin incision was made at S1/2 region, the dermal sinus tract was identified, which had a small (about 8mm) extradural mass, the sinus tract was further followed which eventually became intradural at the S1 level, the sinus tract was identified running cranially to end at the filum terminale-CONUS junction as a second small (about 8mm) intradural nodule, Thus given a Dumbell shaped appearance.The sinus tract was carefully dissected from surrounding Nerve roots, blood vessels, and Filum terminale to be removed as single piece under continuous neuromonitoring. In total, it was about 8cm, and under a microscope, a few patchy hairs were noticed along the length of the dermal track (Figure 3). Samples were taken for pathology that showed skin excision with underlying fibro adipose tissue. There is a sinus tract that keeps dilation and forms a cyst (Figure 3A). The tract and the cyst are lined by keratinized stratified squamous epithelium with numerous keratin flakes in the lumen. The cyst wall has skin adnexal structures including sebaceous glands, hair follicles (Figure 3B), and eccrine and apocrine sweat glands. Focally there is mature glial brain tissue adherent to the skin (Figure 3c). Post-operatively the child had an uneventful smooth recovery. A follow-up MRI showed total resection of the sinus tract and intraspinal lesion and resolution of the previously seen meningeal enhancement and was eventually discharged after completion of his antibiotic course.
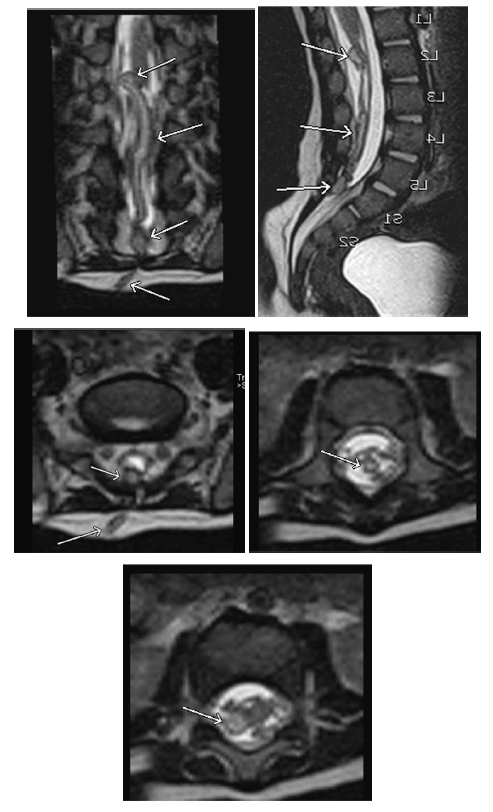
Figure 1: MRI of the lumbosacral spine, 3 D T2 weighted sequence 1 mm thickness (SPACE), coronal and sagittal reconstruction images (a), and (b) respectively showing the full display of the different components of the abnormality, from inferior to superior a subcutaneous sinus tract coursing to a 5 mm extradural nodular lesion at sacral S1-2 level, continuous with an intradural sinus tract, ending in an intradural 5 mm nodular lesion at L1-2 level (arrows from inferior to superior in a, and b). Axial sections at different levels, (b) at sacral S1-2, (c) at mid-lumbar, and (d) at lumbar L1-2 levels, showing the subcutaneous sinus tract coursing to the extradural nodular lesion with bright signal intensity center and low signal intensity rim (long and short arrows respectively in a), cross-section of the intradural tract surrounded by cauda equina nerve roots (arrow in c), and the upper intradural nodular lesion (arrow in d).
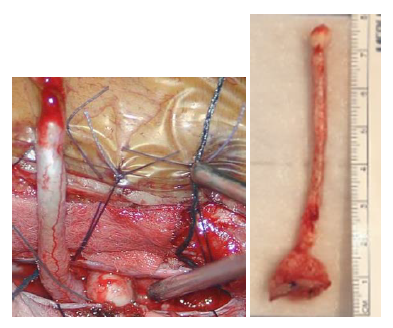
Figure 2: Intra-operative photograph (a) showing the dermal sinus tract and the second nodule seen intradural. (b) Total excision of the tract along with the nodules.
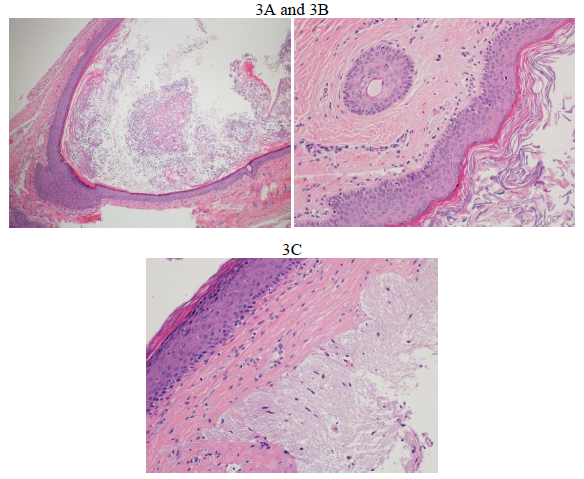
Figure 3: A) Sections show dilated sinus forming a cyst (Hematoxylin and Eosin ×40); B) Sections show stratified squamous epithelium lining the sinus/cyst wall containing hair follicle (left side of the figure) (Hematoxylin and Eosin ×200); C) Sections show adherent glial brain tissue to the squamous epithelium (right side of the figure) (hematoxylin and eosin ×200).
2. Discussion
The spine's spinal tumors can be broadly classified into two categories extradural or intradural, with the intradural tumors further subdivided into intra- or extramedullary tumors. About 55 % of all primary spinal tumors are intra-dural extramedullary [2], with the two most common tumors of the spine being the nerve sheath tumors (neurofibromas and schwannomas) and spinal meningiomas [3]. Spinal teratomas are rare tumors encountered more commonly in children, usually associated with spinal dysraphism and a rare entity in adults where it can present as intramedullary tumor [4]. The first case of intraspinal Teratoma was reported in 1863 by Virchow [5]. Teratomas are embryonal tumors that normally arise from germ cells. They are defined as being composed either of tissues that are foreign to the area in which they form, or of tissues that derive from all three of the germ layers. Malignant teratomas are known as teratocarcinomas; these cancerous growths have played a pivotal role in the discovery of stem cells. “Teratoma” is Greek for “monstrous tumor”; these tumors were so named because they sometimes contain hair, teeth, bone, neurons, and even eyes.
A literature review described a broad spectrum of the association, including diastematomyelia, myelomen-ingocele, spina bifida, partial sacral agenesis, and hemivertebrae to Spinal Teratomas [6]. On the other hand, some cases have been described with preceding history of trauma or prior procedure like lumbar punctures [7]. There are two proposed theories for intraspinal teratomas, the germ cell theory that postulates that these tumors arise from germ cells that migrate during embryogenesis, the other theory postulates that these tumors arise from the misfolding of germ cells in the lateral mesoderm causing entrapment in various parts of the nervous system during secondary neurulation (non-dis-junction) [8]. Other than that, there have been case reports in the literature indicating that preceding history of trauma and intragenic intervention have also been implicated [9]. After the repair of myelomeningocele in patients, it has also been reported due to some dermal and epidermal tissue remains after the closure or after spinal epidural injection [10]. Most of the closed Dermal Sinus tracks present with midline cutaneous dimple overlying the spine and are associated with other cutaneous abnormalities, such as hemangiomas or tuft of hairs, discolouration [1]. The orifice of the sinus may be so small that it escapes detection except on close inspection. Hair may be seen protruding from the opening. The sinus tract begins at the skin dimple and tracks cephalad through the soft tissues to traverse the dorsal dura. Our Patient except for 2 Dimple didn’t have any associated abnormality. Most of dermal sinuses clinically manifest within the 1st decade, usually before the age of 5, because of their tendency to become infected [11]. Authors have recommended inspection of the midline skin carefully in a child sufferings repeated episodes of unexplained meningitis [12].
The first case report of intraspinal cystic Teratoma revealed by recurrent episodes of chemical meningitis was in 1980 by Albert Larbrissea et al. [13]. Extensive Review of Literature by Tebruegge M et al., on epidemiology and pathogenesis of recurrent bacterial meningitis, described 24 cases of recurrent bacterial meningitis associated with dermoid and epidermoid cysts, with the most case having an abnormal connection to the CSF space in the form of a dermal sinus tract which was the causation for meningitis in early months of Life [14]. In his single center-based case series over 15 years, Sharma et al. reported 10 cases of spinal Teratoma, with age ranging from 10 months to 51 years (mean 23.5 years) with male predominance, and most common clinical presentation was the weakness of lower limbs, backache and urinary bladder symptoms [15]. Our patient presented with history of recurrent bacterial meningitis- with no focal neurological deficit or neurological manifestations - despite prolonged antibiotic coverage, Sharma et al, also stressed the importance of total surgical resection as the treatment of choice. They had one case of recurrence after nine years of operation. Total lesion biopsy should be done ,not only a sample biopsy; as many isolated tumors shows mixed histopathological patterns with immature cells, whole mass rather than just small biopsy specimens should be done as many isolated tumors shows mixed histopathological patterns with immature cells.
3. Conclusion
Even though Spinal Teratoma can present in various ways, ranging from recurrent meningitis to paralysis and spinal compression symptoms, a lower threshold for Complete Spinal imaging should be maintained in recurrent infection, especially in the early age of Life immunocompetent patients. Although a rare entity, Mature cystic Teratoma should be included as the Differential diagnosis alongwith Dermoid and Epidermoid, especially when it presents the Dermal sinus tract. Complete surgical excision is the mainstay of treatment and to avoid further recurrent infections.
References
- Mishra SS, Panigrahi S. Thoracic congenital dermal sinus associated with intramedullary spinal dermoid cyst. J Pediatr Neurosci 9 (2014): 30-32.
- Zimmerman RA, Bilaniuk LT. Imaging of tum-ors of the spinal canal and cord. Radiol Clin North Am 26 (1988): 965-1000.
- Gebauer GP, Farjoodi P, Sciubba DM, et al. Magnetic resonance imaging of spine tumors: Classification, differential diagnosis, and spectrum of disease. J Bone Joint Surg Am 90 (2008): 146-162.
- Sung KS, Sung SK, Choi HJ, et al. Spinal intradural extramedullary mature cystic Teratoma in an adult. J Korean Neurosurg Soc 44 (2008): 334-337.
- Virchow R. Die krankhaften Geschwulste. Dreissing Vorlesungen gehalten wahrend des Wintersemesters 1862-1867 an der Universitat zu Berlin. Berlin, Germany: A. Hirschwald (1863).
- Nonomura Y, Miyamoto K, Wada E, et al. Intramedullary Teratoma of the spine: Report of two adult cases. Spinal Cord 40 (2002): 40-43.
- Cavazzani P, Ruelle A, Michelozzi G, et al. Spinal dermoid cysts originating intracranial fat drops causing obstructive hydrocephalus: Case reports. Surg Neurol 43 (1995): 466-469.
- Echevarria ME, Fangusaro J, Goldman S. Pediatric central nervous system germ cell tumors. A review Oncologist 13 (2008): 690-699.
- Stevens QE, Kattner KA, Chen YH, et al. Intradural extramedullary mature cystic Teratoma: Not only a childhood disease. J Spinal Disord Tech 19 (2006): 213-216.
- Kudo N, Hasegawa K, Ogose A, et al. Malignant transformation of a lumbar intradural dermoid cyst. J Orthop Sci 12 (2007): 300-302.
- Morandi X, Mercier P, Fournier HD, et al. Dermal sinus and intramedullary spinal cord abscess. Report of two cases and review of the literature. Childs Nerv Syst 15 (1999): 202-208.
- Wang KC, Yang HJ, Oh CW, et al. Spinal congenital dermal sinus- experience of 5 cases over a period of 10 years. J Korean Med Sci 8 (1993): 341-347.
- Larbrisseau A, Renevey F, Brochu P, et al. Recurrent chemical meningitis due to an intraspinal cystic teratoma. Journal of Neurosurgery 52 (1980): 715-717.
- Tebruegge M, Curtis N. Epidemiology, etiology, pathogenesis, and diagnosis of recurrent bacterial meningitis. Clinical microbiology reviews 21 (2008): 519-537.
- Sharma M C, Aggarwal M, Ralte A M, et al. Clinicopathological study of spinal teratomas. Journal of Neurosurgical Sciences; Torino 47 (2003): 95-100.
