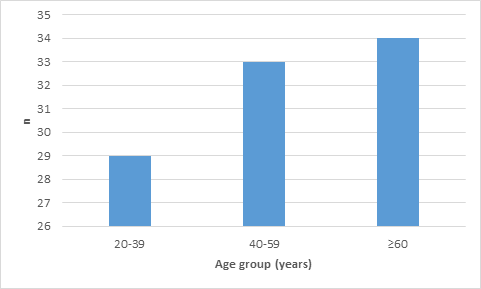Clinical Characteristics of Patients Newly Diagnosed with Primary Open Angle Glaucoma at the Yaoundé Central Hospital-Cameroon
Article Information
Viola A Dohvoma1,2*, Stève R Ebana Mvogo1, Marina Bidjogo1, Caroline T Mvilongo2, Marie E Akono2, Marie B Nguena2, Emilienne Epee1,2, Côme Ebana Mvogo1,2
1Faculty of Medicine and Biomedical Sciences, University of Yaoundé I, Yaoundé-Cameroon
2Yaoundé Central Hospital, Yaoundé-Cameroon
*Corresponding Author: Viola Andin Dohvoma, Faculty of Medicine and Biomedical Sciences, University of Yaoundé I, P.O. Box 3851 Messa, Yaoundé, Cameroon
Received: 09 December 2019; Accepted: 18 December 2019; Published: 17 January 2020
Citation: Viola A Dohvoma, Stève R Ebana Mvogo, Marina Bidjogo, Caroline T Mvilongo, Marie E Akono, Marie B Nguena, Emilienne Epee, Côme Ebana Mvogo. Clinical Characteristics of Patients Newly Diagnosed with Primary Open Angle Glaucoma at The Yaoundé Central Hospital-Cameroon. Journal of Ophthalmology and Research 3 (2020):1-7.
Share at FacebookAbstract
Objective: To describe the clinical characteristics of patients newly diagnosed with primary open angle glaucoma (POAG) in a tertiary hospital.
Methods: A review of the medical records of patients newly diagnosed with POAG between 1st January 2016 and 31st January 2018 was done. POAG diagnosis was based on the presence of any two of the following: elevated intraocular pressure (IOP), glaucomatous optic nerve changes and characteristic visual field defects in the presence of a normal angle on gonioscopy. Blindness was defined as corrected distant visual acuity (CDVA) of < 0.05. Severe visual impairment was defined as CDVA <0.3 but >0.05.
Results: Amongst the 10 400 new patients seen during the study period, 96 were diagnosed with POAG (0.92%). The male: female sex ratio was 1.5. The mean age was 49.1 ± 19.9 years. Family history of glaucoma was present in 11 patients (11.46%). The mean cup/disc ratio was 0.6 ± 0.2. Glaucomarelated blindness was unilateral in 5.2% of patients (n=5). No patient had bilateral blindness. Unilateral J Ophthalmol Res 2019; 2 (3): 050-056 DOI: 10.26502/fjor.2644-00240011 Journal of Ophthalmology and Research 2 and bilateral severe visual impairment were present respectively in 10.42% and 33.33% of cases.
Conclusion: Late presentation is common in POAG with severe cupping and moderate to advanced visual field defects. Unilateral blindness or severe visual impairment is present in 38.5% of cases. Information and screening should be put in place to encourage early diagnosis which will reduce the burden of glaucoma-related blindness.
Keywords
Primary open angle glaucoma; Blindness; Severe visual impairment
Article Details
1. Introduction
Glaucoma is the third cause of blindness worldwide after cataract and uncorrected refractive error. It was responsible for blindness in 8% amongst the 36 million blind people in 2015 [1]. Africa has the highest prevalence of blindness, with glaucoma accounting for 15% [2]. Primary open angle glaucoma (POAG) which is the most frequent form of glaucoma has a prevalence of 4.16% in Africa. When compared to other World Health Organization sub-regions, Africa presents the highest prevalence of POAG [3]. In Cameroon, hospital-based studies have reported the prevalence of POAG to be between 2.2 and 4.4% [4, 5]. Besides a high prevalence, it has an earlier onset and progresses more rapidly than in Caucasians [6, 7]. The lack of symptoms at the onset of POAG, socio-economic deprivation, ignorance and lack of screening all contribute to late presentation. For many patients, diagnosis is made at a stage of advanced disease with blindness or severe visual impairment [8]. Severe visual impairment and blindness at presentation is indicative of late diagnosis and such data can help in the planification of eye health services. Early diagnosis may play a role in the fight against blindness which will go a long way to improve the socio-economic status of both the individual and the community. The aim of this study was to determine the prevalence of severe visual impairment and blindness amongst patients newly diagnosed with primary open angle glaucoma at the eye unit of a tertiary hospital.
2. Patients and Methods
We carried out a descriptive study in which we reviewed the medical records of all new patients seen between 2nd January 2016 and 31st January 2018 at the ophthalmic unit of a tertiary hospital. Records of adult patients (≥ 20 years) diagnosed with POAG were included. Diagnosis of POAG was based on the presence of glaucomatous optic nerve changes and characteristic visual field defects with an open angle on gonioscopy. Ethical clearance was obtained from the institutional ethical clearance committee and anonymity was respected. Variables studied included age, sex, family history of glaucoma, corrected distant visual acuity, intraocular pressure (IOP), central corneal thickness (CCT), vertical cup/disc ratio and the mean defect on automated visual field. The severity of visual defects was graded using the Hodapp, Parish and Anderson’s grading scale (early defect for MD no worse than -6dB; moderate for MD worse than -6dB but no worse than -12dB; severe for MD worse than -12dB) [9]. Blindness was defined as best-corrected distant visual acuity (BCDVA) of < 0.05 and severe visual impairment was defined BCDVA <0.3 but >0.05. Blindness and severe visual impairment were defined for each eye. These were considered glaucoma-related in the absence of any other blinding condition in each eye.
3. Results
Amongst the 10 400 new patients seen during the study period, 96 were diagnosed with POAG (0.92%). The male: female sex ratio was 1.5. The mean age was 49.1 ± 19.95 years and 30.2% of the study population were aged below 40 (Figure 1).
Ten patients (10.4%) came for voluntary screening for glaucoma, amongst whom 4 had a positive family history of glaucoma. A total of 11 patients (11.46%) had a family history of glaucoma. Eighteen patients (18.8%) were diabetic and 16 (16.7%) were hypertensive.
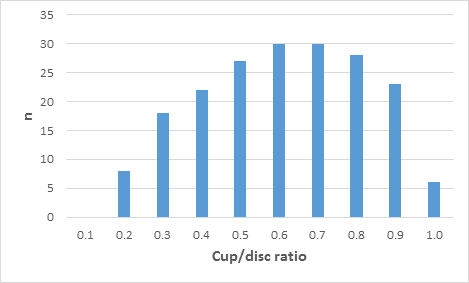
The IOP was >21mmHg in 26 patients (26.8%). Central corneal thickness was measured in 64 eyes of 34 patients (35.42%). No case of elevated IOP was adjusted to normal IOP and no case of normal IOP was adjusted to elevated IOP. The mean cup/disc ratio was 0.6 ± 0.2 and there was asymmetry in 34 patients (35.42%). The distribution of the cup/disc ratio is shown in (Figure 2).
Considering the mean defect (MD), 29.7% of eyes had advanced glaucoma. The classification of the severity of glaucoma with respect to the MD is shown in (Table 1).
|
Number of eyes |
Percentage (%) |
|
|
Mild (MD < 6db) |
92 |
47.9 |
|
Moderate (MD: 6-12db) |
43 |
22.4 |
|
Severe (MD> 12db) |
57 |
29.7 |
|
Total |
192 |
100 |
MD: mean defect; db: decibel
Table 1: Severity of glaucoma with respect to mean defect.
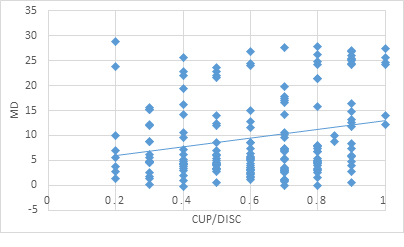
There was a positive correlation (r=0.23; p=0.0013) between the cup/disc ratio and the MD as shown in (Figure 3).
Glaucoma-related blindness was unilateral in 5.2% of patients (n=5). No patient had bilateral blindness. Unilateral and bilateral severe visual impairment were present respectively in 10.42% and 33.33% of cases.
4. Discussion
New cases of POAG represented 0.9% of all new patients seen in this study. This is smaller than the 3.8% reported by Olushola et al in the university teaching hospital of Ibadan [10]. The ratio of new cases in this hospital-based study might be smaller than expected in the general population, because POAG is usually asymptomatic and patients might not seek care in the early stages of the disease. This is supported by the high frequency of new cases in population studies. Studies from Cameroon and Ghana have reported frequencies of new POAG cases of 8.2% and 8.5% following the screening of 635 and 1483 people respectively [11, 12]. In the Barbados Eye Study, a larger black population of 4709 was screened and 7% of new POAG cases were detected [13]. Based on these population studies, there are possibly many more undiagnosed cases in our general population. Early diagnosis and management of POAG which play a role in the prevention of visual impairment and blindness should be encouraged, by putting in place measures such as voluntary screening and the screening of family members of patients with POAG. In this study, 10.4% of cases came for voluntary screening, amongst whom 40% had a family history of glaucoma. There were more males than females in this study. This male predominance has been reported by some authors [10, 14], though others found no difference [12]. The mean age in our study was 49.1 years. This corroborates several African authors who report the mean age of diagnosis of POAG to be between 45 and 55 years [12, 14, 15].
Low-tension glaucoma was common in this study. However, pachymetry was done only in 34 patients (35.4%). Though the pachymetry did not influence the outcomes of the IOP for these patients, the influence of the central corneal thickness cannot be underestimated as up to 11.3% of new cases of low-tension glaucoma have been shown to have elevated IOP following adjustments after pachymetry [16]. Several studies have reported the frequency of low-tension glaucoma to be about 50% of all POAG [17, 18, 19]. In Japanese patients, low-tension glaucoma could represent as high as 70 % of POAG cases [17]. The mean vertical cup/disc ratio was 0.6 ± 0.2. Eyes with a cup/disc ratio ≥ 0.7 represented 45.3% of cases (n=87/192). This is lower than that reported by other authors for newly diagnosed POAG. In the series of Abdull et al, the mean cup/disc ratio was 0.8 [18]. Olushola et al reported a cup/disc ratio ≥ 0.7 in 72% of eyes while [10]; Osaki et al reported a cup/disc ratio ≥ 0.8 in 67.7% of eyes [19]. Severe cupping indicates advanced glaucoma which increases the risk of visual impairment. This ties with the positive correlation found between cup/disc ratio and the MD.
The prevalence of blindness in this study is lower than those reported by other authors. Unilateral blindness was found in 5.2% of patients and there was no case of bilateral blindness. Olushola et al reported 37% and 28% respectively of unilateral and bilateral blindness [10], while Olawoye et al reported 44.6% and 29.7% respectively [20]. The low prevalence of blindness in our study could be due to the study site. This study was done in a general ophthalmology unit whereas those of previously cited authors were done in glaucoma units, where many cases were referred from either optometrists or general ophthalmologists. However, there is a possibility that our prevalence underestimates the reality of glaucoma-related blindness at diagnosis given its hospital-based nature. Patients with bilateral blindness may not present to hospital services. There is a general suboptimal utilization of health care services in our setting for reasons such as ignorance, beliefs, inaccessibility and poverty.
5. Conclusion
With a third of new POAG patients presenting with bilateral severe visual impairment in this study, efforts need to be put in place to inform and screen patients for glaucoma. A national population-based study will better evaluate the scale and the impact of glaucoma.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Flaxman SR, Bourne RRA, Resnikoff S, et al. Global causes of blindness and distance vision impairment 1990-2020: a systematic review and meta-analysis. Lancet Glob Health 5 (2017): e1221-e1234.
- Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ 82 (2004): 844-851.
- Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020 90 Br J Ophthalmol 90 (2006): 262-267.
- Eballé AO, Owono D, Bella AL, et al. Caractéristiques cliniques et épidémiologiques du glaucome chronique à angle ouvert. Cahiers d’études et de recherches francophones / Santé 18 (2008): 19-23.
- Ellong A, Mvogo CE, Bella-Hiag AL, et al. La prévalence des glaucomes dans une population de Noirs camerounais. Cahiers d’études et de recherches francophones / Santé 16 (2006): 83-88.
- Tielsch JM, Sommer A, Katz J, et al. Racial variations in the prevalence of primary open-angle glaucoma. The Baltimore Eye Survey. JAMA 266 (1991): 369-374.
- Ostermann J, Sloan FA, Herndon L, et al. Racial differences in glaucoma care: the longitudinal pattern of care. Arch Ophthalmol 12 (2005): 1693-1698.
- Ashaye AO. Clinical Features of Primary Glaucoma in Ibadan. Niger J Ophthalmol 11 (2003): 70-75.
- Hodapp E, Parish R, Anderson D. Clinical decisions in glaucoma. St Louis: Mo-Mosby, (1993).
- Olushola O, Oluwatoni O, Omodele J, et al. Spectrum of Glaucoma Presentation in a Suburban Teaching Hospital in South Western Nigeria Health Science J 10 (2016): 466.
- Preussner P-R, Grossmann A, Ngounou F, et al. Glaucoma screening in Western Cameroon. Graefes Arch Clin Exp Ophthalmol 247 (2009): 1671-1675.
- Ntim-Amponsah CT, Amoaku WMK, Ofosu-Amaah S, et al. Prevalence of glaucoma in an African population. Eye 18 (2004): 491-497.
- Leske MC, Connell AM, Schachat AP, et al. The Barbados Eye Study. Prevalence of open angle glaucoma. Arch Ophthalmol 112 (1994): 821829.
- Yehouessi L, Doutetien C, Sounouvou I, et al. Dépistage du glaucome primitif à angle ouvert au centre national hospitalier et universitaire de Cotonou, Bénin. J Fr Ophtalmol 32 (2009): 20-24.
- Awoyesuku EA, Ejimadu CS. Visual disability in newly diagnosed primary open angle glaucoma (POAG) patients in a tertiary hospital in Nigeria. Niger J Med 21 (2012): 78-80.
- Dohvoma V, Nchifor A, Ebana Mvogo S, et al. Central Corneal Thickness in Newly Diagnosed Cameroonian Glaucoma Patients with “Normal” Intraocular Pressures. J Ophth Dis 1 (2015): 103.
- Shiose Y, Kitazawa Y, Tsukahara S, et al. Epidemiology of glaucoma in Japan- a nationwide glaucoma survey. Jpn J Ophthalmol 35 (1991): 133-155.
- Abdull MM, Gilbert CC, Evans J. Primary open angle glaucoma in northern Nigeria: stage at presentation and acceptance of treatment. BMC Ophthalmol 15 (2015): 111.
- Osaki TH, Kasahara N, Della Paolera M, et al. Presentation of glaucoma in an urban tertiary care hospital in South America: legal blindness and prevalence. Int Ophthalmol 30 (2010): 361-366.
- Olawoye O, Tarella S. Spectrum of glaucoma presentation in a Nigerian tertiary hospital. Niger J Ophthalmol 22 (2014): 11-15.