Bornavirus Antigens in Psittaciformes Infected with Psittaciform Bornavirus and their Use in Diagnostic Procedures
Article Information
Josephine M McHugh, Siwo R de Kloet*
Animal Genetics Inc., 3382 Capital Circle NE, Tallahassee FL 32308, USA
*Corresponding Author: Dr. Siwo R. de Kloet, Animal Genetics Inc., 3382 Capital Circle NE, Tallahassee FL 32308 USA
Received: 28 September 2020; Accepted: 05 October 2020; Published: 03 November 2020
Citation: Josephine M McHugh, Siwo R de Kloet. Bornavirus Antigens in Psittaciformes Infected with Psittaciform bornavirus and their Use in Diagnostic Procedures. Archives of Veterinary Science and Medicine 3 (2020): 83-108.
Share at FacebookAbstract
This study describes the occurrence of psittaciform bornavirus antigens and antibodies in tissues of birds persistently infected with psittaciform bornavirus. Analysis of serum of infected birds shows that antibodies against the P40 nucleoprotein occur most commonly, whereas antibodies to the glycoprotein are virtually completely absent. Classical Western blotting using rabbit antibodies showed that the P40 nucleoprotein is the only antigen which can be reliably detected in tissues as diverse as brain, liver, feather calami and material such as cloacal swabs. The sensitivity of the latter approach is about 1 µg. Using automated Western blotting (ProteinSimple, WES) improved this sensitivity to about 200 pg. Rabbit antibodies against the P24 phosphoprotein detect a cross-reacting protein of the same molecular size in a classical Western blot of tissue homogenates of uninfected birds. Rabbit antibodies against the P16 matrix protein do not detect the protein in a Western blot, classical or automated, possibly through shielding by other components in the tissue homogenates or prevention of the transfer of the protein from the gel to the PDVF membrane during the blotting, although such antibodies can be used as detection antibodies in a sandwich ELISA analysis where the capture antibodies are of avian origin. The latter approach allows the detection of the P24 phosphoprotein and the P16 matrix protein in feather calami and cloacal swabs at a sensitivity of approximately 200 pg.
Keywords
Avian bornavirus; P40 nucleoprotein; P24 phosphoprotein; P16 matrix protein; ELISA (enzyme-linked immunoassay); sandwich ELISA; Classical Western blotting; Automated Western blotting
Avian bornavirus articles; P40 nucleoprotein articles; P24 phosphoprotein articles; P16 matrix protein articles; ELISA (enzyme-linked immunoassay) articles; sandwich ELISA articles; Classical Western blotting articles; Automated Western blotting articles
Article Details
Abbreviations
ABG = avian bornaviral ganglioneuritis
ABV = avian bornavirus
BSA = bovine serum albumen
EDTA = ethylene diamino tetraacetic acid
Elisa = enzyme-linked immunosorbent assay
HRP = horse radish peroxidase
MBP = maltose binding protein
MWD = macaw wasting disease
PBS = 0.15 M NaCl in 0.02 M sodium phosphate buffer pH 7.4
PDD = proventricular dilatation disease
PMSF = phenylmethylsulfonyl fluoride
PsBV = Psittaciform orthobornavirus
PVDF = polyvinylidene difluoride
TBST = 0.15 M NaCl containing 0.02 M tris-HCl buffer pH 7.0 and 0.05% Tween 20
TBSTM = TBST containing 5% fat free dried milk
TMB = 3,3′,5,5′-tetramethylbenzidine
VSV = vesicular stomatitis virus
1. Introduction
Psittaciform orthobornavirus (PsBV) (order Mononegavirales, family Bornaviridae, genus Orthobornavirus, [1, 2] is associated with proventricular dilatation disease (PDD), also called avian bornaviral ganglioneuritis (ABG) or macaw wasting disease (MWD), an often fatal disorder of parrots and parakeets [3, 4]. 2 species of PsBV (PsBV 1 and 2) consisting of 8 strains are currently recognized [1]. PsBV is primarily a noncytolytic, neurotropic virus giving rise to neurological symptoms [5] such as unstable gait and feather picking, but the virus can also infect many other organs such as feather calami and the proventriculus [6, 7, 8] causing intestinal blockage leading to starvation. PDD is therefore often first recognized by the appearance of undigested seed in fecal matter. Nevertheless, many birds, infected with bornavirus, can live a long, symptom-free life [9, 10].
Bornavirus infection in psittaciformes, an avian family which contains many endangered species, is primarily a disease of captive birds and may affect species such as the Spix macaw (Cyanopsitta spixii) which currently only survives in captivity (e.g. [11]) or the Kakapo (Strigops habloptilus) which has a very limited geographical distribution in New Zealand [12, 13]. Infection of psittaciform birds in their natural habitat or among feral birds has not been established with certainty [14-17] and more field studies should be conducted. The mode by which birds in captivity transmit the infection is not known, but fecal-oral transmission is suspected [18, 19]. Experimental infections are done by injection of infected tissue homogenates or cell cultures. The virus can also be vertically transmitted through eggs [20,
21] and suspected cases of “spontaneous infection” may well be the result of birds having asymptomatic parents [22].
Psittaciform bornavirus is a negative-stranded RNA virus, with a genome of approximately 8900 nts, carrying six genes coding for 1) P40, a nucleoprotein, 2) P10, a cofactor for transcription, 3) P24, a phosphoprotein, 4) P16, a matrix protein, 5) P57, a glycosylated capsid protein and 6) a RNA dependent RNA polymerase [23, 24]. Virus-like structures have been observed in birds infected with bornavirus [25] and psittaciform bornavirus can be cultured from infected tissue segments in Japanese quail cells [26, 27] or duck embryo cells [10]. However, the physical purification of psittaciform bornavirus has not been described and although the purification of mammalian bornavirus by CsCl density gradient centrifugation [28], affinity chromatography with “virobeads” [29] and tandem affinity purification [30] has been reported, the actual quantitative molecular composition of the particles of this virus has however also not been described [31].
At present antemortem detection of infection of birds with PsBV is routinely done by RT-PCR of material derived from cloacal swabs, fecal matter and feather calami or by serological means through detection by immunohistochemistry of biopsies and Elisa or Western blotting using anti-avian bornavirus antibodies [9, 32-34]. The use of RNA as diagnostic material suffers from its chemical instability, although procedures for stabilization with [e.g “RNA-later” (Lader, US patent 8,178,296 B2)] or with guaninidium thiocyanate [35] are known. Furthermore, use of fecal matter as a source of bornaviral RNA suffers from the fact that the infected bird may be an “intermittent fecal shedder” [36], requiring the collection of several samples at different times, which can be a major problem in an investigation involving wild birds. In addition, the virus in fecal matter may just be “passing through” without truly infecting the bird [36, 37]. Calami of dried feathers are therefore more useful, for even after storage at ambient temperature for several months, RNA usable for diagnostic purposes can still be obtained [38, 39]. Proteins are in general more stable than RNA [40] and may therefore in certain circumstances be more useful than RNA. In this report we further describe therefore which bornaviral proteins accumulate in detectable amounts in feather calami and cloacal material of parrots and parakeets infected with psittaciform bornavirus and how such proteins can be used in establishing useful diagnostic procedures for the detection of an psittaciform bornavirus infection in Psittaciformes, using automated Western blotting and sandwich ELISA.
2. Materials and Methods
2.1 Bird maintenance and determination of infection with avian bornavirus
Most sun conures (Aratinga solstitialis) and other birds used in this investigation were maintained in the private aviaries of the principal investigator (S.R. de K.) as described previously [32]. Samples of other birds were obtained from customers of Animal Genetics. Infection with bornavirus was detected by RT-PCR of feather calamus RNA as described in an earlier publication using primers that amplify the P10 gene and a segment of the polymerase gene [32]. Tissues were obtained from birds which had died from PDD or from other causes, such as fighting, predation, accidents, etc. No birds were euthanized for this investigation. Feathers (contour and wing cover) were obtained by plucking from dead, as well as living birds. Blood was collected from the brachial vein. No anesthesia was used. Tissue of a mouse (Mus musculus) was obtained from a colleague at the University of Florida. Additional bird tissues, feathers and blood were obtained from other, local, breeders.
2.2 Elisa for the detection of anti-bornaviral antibodies
Blood was collected from the brachial vein using 20 µl EDTA coated glass capillaries (Cat # 19.447, Sarstedt, Newton, NC, USA). For routine ELISA, the contents of the capillaries were immediately diluted 10 fold in 180 µl of 0.15 M NaCl containing 0.025 M Na2-EDTA pH 8.0 (buffer A) and centrifuged for 10 min in an Eppendorf minifuge at 2000xg at 4oC to obtain the diluted plasma.
Indirect ELISA, using recombinant avian bornavirus proteins conjugated to maltose binding protein (MBP, New England Biolabs, Ipswich, Massachusetts, USA) was carried out as described earlier [20, 32, 38]. The MBP gen (Genbank accession number AAB86559.1) of the vector pMAL-c2X was modified by replacing the sequence coding for N10 by a sequence coding for HIS8 and the sequence coding for the factor Xa recognition site by a sequence coding for thrombin proteolytic site. The nucleotide sequences of the genes coding for the proteins have been entered into the Genbank database with accession numbers: FJ792853 (P40); EU781967 (P27, the N-terminal half of the glycoprotein); JF894121 (P29, the C-terminal half of the glycoprotein); HM369502 (P24); HM369503 (P16); JF894120 (P10) and JF894122 (BPOL), a fragment of the psittaciform bornaviral RNA polymerase. One µg recombinant psittaciform bornavirus antigen-MBP conjugate (in 50 µl 0.05M Na2CO3 pH 9.5) was used per well of a microtiter plate (Brandplates, immunoGradeTM, Brand GMBH, Wertheim, Germany) and incubated with shaking for 60 minutes at room temperature. MBP and empty wells were used as controls. After washing with 0.15 M NaCl containing 0.02 M tris-HCl buffer pH 7.0 and 0.05% Tween 20 (TBST) the plates were blocked for one hour at 40C with 5% fat free dried milk in TBST (TBSTM). Prior to the ELISA the plasma samples were further mixed with TBSTM for a final dilution of 400 fold. Fifty µl of this solution was applied to the wells of the microtiter plate. After one hour shaking at room temperature the plates were washed again with TBST and subsequently incubated for one hour with 2000 fold diluted goat anti-bird antibody conjugated to horseradish peroxidase (HRP) (Cat# A140-110P, Bethyl, Montgomery, TX, USA) in TBSTM as the secondary antibody. After washing with TBST, 50 μl of a non-precipitating, stabilized TMB solution (Cat# TM1999, Scytek Laboratories, Logan, UT, USA) was applied to the wells, and the plates were incubated for 15 min at room temperature, approximately 250C. The reaction was stopped with 50 µl 0.5 N HCl, and the optical density was measured at 450 nm, using a microplate reader (Tecan, Mannedorf, Switzerland). As cut-off value for identifying positive sera the mean absorbance value of 3 standard deviations above the mean absorbance value of sera from a group of 8 psittaciform bornavirus-negative African grey parrots was taken [32].
2.3 Polyclonal rabbit antibodies against avian bornavirus P40, P29, P24, P16, P10 and bornavirus RNA polymerase antigensAffinity purified polyclonal antibodies against psittaciform bornavirus P40 (nucleoprotein), P29 (C-terminal segment of glycoprotein), P24 (phosphoprotein), P16 (matrix protein), P10 (replication cofactor) and a fragment of the RNA polymerase, conjugated to keyhole limpet hemocyanin (KLH) were generated in rabbits for us by Genscript (Piscataway, NJ, USA). The Genbank accession numbers of the proteins used by Genscript are the same as mentioned above for the ELISA with the exception of the fragment used for the psittaciform bornavirus RNA dependent RNA polymerase (amino acids 543-716 Genbank accession nr NC_028106.1) and which was recommended to us by Genscript. The titers of the antibody preparations were, as reported by the manufacturer and confirmed by us, approximately 100,000.
2.4 Classical western blotting
For classical Western blotting of feather calamus extracts and tissue homogenates, using polyacrylamide gel electrophoresis [42] and electro-transfer to PVDF membranes, four 3 mm feather calami (from contour- or small wing cover feathers) were cut off and immersed in 50 µl loading buffer (1% sodium dodecyl sulphate in 0.05 M Tris-HCl buffer pH 8.0, 0.1 M EDTA, 0.5% mercaptoethanol 0.005% bromophenol blue and 0.5 mM phenylmethylsulfonyl fluoride (PMSF) and immersed in a boiling waterbath for 5 minutes. For brain or liver, 50 mg tissue was homogenized in 500 µl of 2% SDS and 5 µl protease inhibitor cocktail (Cell Signaling Technology, Danvers, MA, USA) in a mini Dounce homogenizer [38]. Fifty µl of the homogenate was mixed with 50 µl of 2% sodium dodecyl sulphate in 0.1 M Tris-HCl buffer pH 8, 0.2 M EDTA, 0.01% bromophenol blue and 1% mercaptoethanol and heated for 5 min in a boiling waterbath. Ten µl of the feather extracts or tissue lysates were loaded onto a 12% polyacrylamide gel made in tricine buffer [42]. Electrophoresis as well as electrotransfer to PDVF membranes was carried out using standard procedures [42-45] as described before [38]. After blocking in TBSTM, blots were incubated for 1 hr at room temperature with the primary antibody diluted 2000 fold in TBSTM, washed with TBST and incubated for 1 hr with goat anti rabbit antibody conjugated to horseradish peroxidase (2000 fold diluted in TBSTM) as the secondary antibody. After washing again with TBST, the blots were developed with the TMB “precipitating” (Cat#TM5999, Scytek Laboratories, Logan, UT, USA) solution.
2.5 Automated western blotting
Automated Western blotting was carried out using WES (ProteinSimple, San Jose, CA, USA). For tissue homogenates 0.25 µl tissue homogenate, prepared as described above, were mixed with 1 µl ProteinSimple 5x fluorescent mastermix and 3.75 µl 0.1 x sample buffer and immersed in a boiling waterbath for 5 min. For feather calami, four 3 mm long, dried wing cover feather calami (approximate combined weight approximately 1 mg) were suspended in 25 µl 2 x loading buffer loading buffer diluted with 25 µl 0.01 M EDTA pH 8.0 and heated for 5 min at 1000C. Again 0.25 µl of the extract, 1 µl ProteinSimple fluorescent mastermix and 3.75 µl of 0.1 x Protein Simple sample buffer were combined, placed in the WES plate and the experiment carried out following the directions of the manufacturer.
2.6 Induction in chickens and isolation of polyclonal anti-avian bornavirus antibodies from egg yolk
One year old Bantam chickens were immunized [46] with 100 µg P40-, P29-, P24- and P16 psittaciform bornavirus protein conjugated to maltose binding protein (MBP), prepared as described above, in 100 µl TBS (0.15 M NaCl, containing 0.02 M tris-HCl buffer pH 7.0) and 100 µl complete Freunds adjuvant (Cat# 77140,Thermofisher Scientific) by injection at two different places in the breast muscle. The immunization was repeated after 2 weeks using incomplete Freunds adjuvant (Cat# 77145, Thermofisher Scientific). Eggs were collected beginning at 2 weeks subsequent to the last immunization. Polyclonal antibodies were isolated from chicken eggs by the polyethylene glycol (PEG) procedure [47, 48], followed by affinity purification using thiophilic gel chromatography [49]. The procedure was as follows: Egg yolk was separated from egg white by careful breaking of the egg shell, followed by decanting of the egg white. After washing the yolk with distilled water to remove residual egg white, the yolk sack was punctured and the yolk collected. The yolk was diluted with an equal volume of buffer A (0.1 M NaCl, 0.02 M Tris-HCl buffer pH 7.4) and mixed with two yolk volumes of 7% polyethylene glycol (MW6000). After storage at 4oC overnight the mixture was centrifuged for 30 min at 14.000 x g at 40C. The supernatant was filtered through Whatman 3MM filter paper. Solid polyethylene glycol (PEG6000) was added to a final concentration of 12 per cent, the mixture was stored overnight at 4oC and the precipitated IgY collected by centrifugation at 14,000 x g. The precipitate was redissolved in a volume of buffer A equal to the original volume of the egg yolk and the IgY was reprecipitated by the addition of an equal volume of 24% PEG in buffer A for a final concentration of 12% PEG and collected by centrifugation for 15 min at 14.000 x g. Subsequently, the IgY was dissolved in half the original yolk volume of buffer A, and an equal volume of 1.8 M (NH4)2SO4 pH 8.0 was added for a final (NH4)2SO4 concentration of 0.9 M. The solution was loaded on a T-gel column (Pierce Biotechnology, Rockford, IL, USA) equilibrated with 0.9M (NH4)2SO4, the column was washed with 0.9 M (NH4)2SO4 until the OD280 reached zero, and the IgY eluted with 0.02 M Tris HCl buffer pH 7.4. The concentration of the IgY was measured by the Bradford assay [50], using purified bovine IgG as the standard and the solution diluted or concentrated by ultrafiltration (Centricon YM 10, Millipore, Burlington, MA, USA) until a final concentration of 2 mg per ml was reached. The titers of the antibody preparations were: Anti-P40 = 2500; anti-P29 = 2500; anti-P24 = 5000 and anti-P16 = 10.000.
2.7 Sandwich ELISA
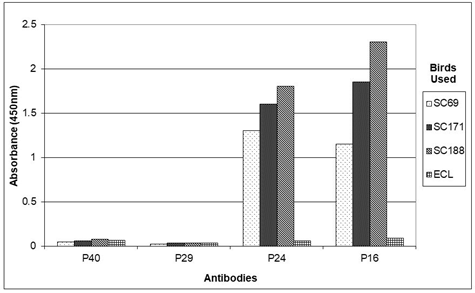
Sandwich ELISA assays [41, 51] for the presence of psittaciform borna virus P40, P29, P24, P16, P10 and RNA polymerase antigens in tissue homogenate, cloacal swabs or feather calami were carried out using the protocol provided by Abcam (Cambridge, Mass, USA) with some modifications. Wells of an ELISA plate were filled with 50 µl sun conure plasma, 100 fold diluted in 0.01 M NaHCO3, pH 9.0 from a bird with antibodies against e.g. P40, P24, P16 and P10(e.g. SuCo 188), as determined by ELISA (Figure 1) or with 100 fold diluted chicken anti-avian borna P40-, P29-, P24- or P16 IgY, prepared as described above, as the capture antibody. The titers of the sun conure serum the respective antibodies were estimated by serial dilution as described earlier [32] and were (e.g. for SuCo 188) P40 = 4000; P24 = 4000, P16 = 4000 and P10 = 1000. The titers of the chicken antibodies are given above. Plates were shaken for an hour at room temperature, subsequently washed with TBST, blocked for an hour with 100 µl TBSTM and washed again with TBST. Subsequently the wells were filled with 50 µl TBSTM containing tissue homogenate, feather calamus extract or cloacal swab material. Tissue homogenates were prepared by homogenizing 50 mg of tissue in 500 µl of cold (40C) PBS in a mini-Dounce homogenizer [20, 38]. The homogenates were centrifuged for 5 min at 10,000 x g at 40C and the supernatant was mixed with 700 µl of TBSTM. The feather calamus extract was prepared by vortexing three 4 mm long wing cover feather calami for 10 min in 250 µl TE, and mixing with 350 µl TBSTM. The cloacal material was obtained in a similar way by vortexing a single swab in 250 µl TE for an hour at room temperature and mixing with 350 µl TBSTM. 50 µl of the tissue homogenate, the feather calamus or cloacal swab material was used per well of the microtiter plate containing the capture antibody and shaken for an hour in a rotary shaker at room temperature. After washing again with TBST, 50 µl of detecting rabbit anti-P40, -P29, -P24, -P16, -P10 or POL antibody, diluted 2000 fold in TBSTM, was added and shaking continued for another hour. The plates were washed with TBST, incubated for an hour with 50 µl 2000 fold diluted goat anti-rabbit antibody conjugated to horse radish peroxidase (Cat# A140-110P, Bethyl, Montgomery, TX, USA), washed again with TBST and finally incubated at room temperature (approx. 220C) for 15 min with 50 µl of non-precipitating, stabilized TMB solution (Cat# TM1999, Scytek Laboratories, Logan, UT, USA). The reaction was stopped with 50 µl 0.5 N HCl, and the optical density was measured at 450 nm, using a microplate reader (Tecan, Mannedorf, Switzerland) as described above for a regular ELISA. Photographs of the plates were made prior to the addition of the 0.5N HCl.
3. Results
3.1 Presence of anti-avian bornavirus antibodies in psittacines naturally infected with psittaciform bornavirus
Psittaciform bornavirus 2 and 4 are the strains most often found among captive psittacines [16, 52]. Polyclonal antibodies, generated in birds or rabbits in response to the antigens of one strain (e.g. PsBV2) recognize the homologous form of these antigens in parrot-PsBV4 as well. A review of the Genbank database showed that the amino acid sequence differences are rather small and range from 2% for the P16 matrix protein to approximately 5% for the P40 nucleoprotein, the P24 phosphoprotein, the polymerase and the glycoprotein. These observations agree with those made earlier by other investigators [52]. Only the P10 protein shows a higher amino acid sequence divergence between the two strains at approximately 20%.
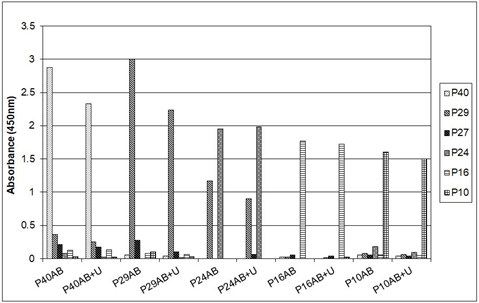
Plasma analysis by ELISA of psittacines naturally infected with psittaciform bornavirus (as determined by bornavirus specific RT-PCR of feather calami or cloacal swabs) shows that antibodies against borna virus antigens (as determined in an ELISA using recombinant antigens) occur in highly variable amounts [32, 38]. Antibodies against the P40 nucleoprotein are almost always found, antibodies which react with the recombinant P24 phosphoprotein, the P16 matrix protein and the P10 protein are frequently found, sometimes in a very high concentration, but sometimes these antibodies are virtually absent. Antibodies against the capsid protein (measured as antibodies against the recombinant N-terminal P27 or C-terminal P29 segments) are mostly undetectable, or are only present in very low quantity. Antibodies against a fragment of the bornavirus RNA polymerase can be found, but since a recombinant form of the entire protein was not available, extensive studies of this component are not included in this investigation. Previous studies [32, 38] have shown that the titers of these antibodies were very variable and could sometimes be very high, e.g. 16.000 for the P24 protein. A time course investigation showed that the specific anti-bornavirus antibody profile of a particular infected bird could be maintained for a very long time, as long as the infected bird was alive, which could be 10 or more years [38]. Finally, although heat denaturation (5 min at 1000C) of the P40, P24 and P10 antigens or washing of the ELISA with 6 M urea had some quantitative effect on the recognition by their respective serum antibodies as determined by ELISA, heat treatment of especially the P16 protein abolished the reaction in some cases almost completely (Figure 1).
Figure 1: Response of anti-bornavirus antibody ELISA profiles of different sun conures to heat denaturation of the recombinant bornaviral antigens. Nrs (333, 188, 189 and 506) are identification numbers of the sun conures used; Ecl is an uninfected Eclectus parrot. D = reaction with heat denatured (5 min at 1000C) antigen.
3.2 Detection of infection with psittaciform bornavirus antigens by Western blotting using rabbit antibodies. Specificity of the rabbit antibodies against individual recombinant bornavirus proteins in an ELISA
In our earlier studies [32, 38], antibodies isolated from egg yolk, obtained from sun conures, naturally infected with psittaciform bornavirus, were used. Such antibodies gave in an analysis by Western blotting a considerably lower background than whole serum. Since the supply of sun conure egg yolk antibodies is dependent on the birds laying eggs and is therefore rather limited, rabbit anti-bornavirus antibodies against the recombinant form of the bornavirus antigens, which can be made on order, were used.
Figure 2 shows the specificity of the rabbit antibodies against the different recombinant psittaciform bornavirus antigens. In an ELISA all antibodies react primarily with their specific antigens, only the P24 antibody reacted also in a substantial way with the P29 antigen. The antibody to the P29 segment did however not react with the P24 protein. Washing with 6 M urea or denaturation by heating (not shown) had little to no effect on the interaction of the antigens with the antibodies.The anti-psittaciform bornavirus polymerase antibody could not be analyzed in depth, since no complete antigen was available and the rabbit antibody was generated against only a fragment.
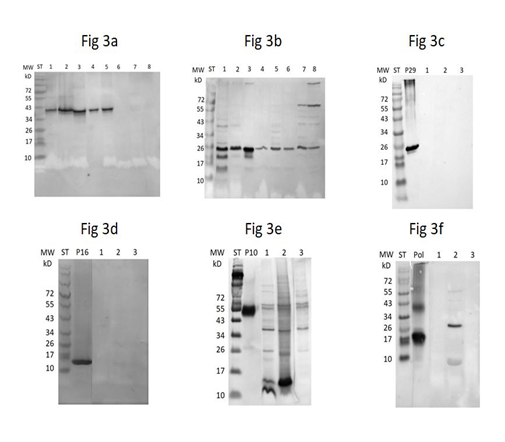
Probing of classical Western blots of brain or liver homogenates derived from parrots or parakeets persistently infected with psittaciform bornavirus [32, 38] with rabbit antibodies generated in response to individual recombinant psittaciform bornavirus proteins made in E. coli can detect a number of psittaciform bornaviral proteins in such tissues. In a classical Western blot rabbit anti-P29 matrix protein (Figure 3c) and anti-P16 glycoprotein antibodies (Figure 3d) do not produce a visible signal, although in an ELISA these antibodies recognize purified recombinant P16 and P29. Although the rabbit anti-P10 antibody reacts primarily with P10 (Figure 3e), in many birds it also cross-reacts with a number of host proteins. The rabbit anti-RNA polymerase antibody does not detect material of approximately 200 kD, the expected molecular weight of the RNA polymerase in the brain homogenate, but instead detects material of approximately 30 kD (Figure 3f) in the liver of infected birds, but not in the brain homogenates displayed in the figure. The data show therefore that only P40 nucleoprotein-like (Figure 3a) and P24 phosphoprotein-like material (Figure 3b) of the correct molecular weight, detectable by the classical Western procedure size, are formed in infected birds. A further examination of the Western blots, showed however that the rabbit anti-P24 antibody detects also material of about 24 kD in tissues of uninfected birds and even in a mouse (Mus musculus) (Figure 3b). These latter results differ from the earlier observations where egg yolk antibodies from a sun conure infected with avian bornavirus did not detect P24-like protein in tissues of uninfected birds [38]. The results show therefore that only the rabbit anti-P40 nucleoprotein antibody identifies a component of approximately 40 kD, which occurs only in birds infected with avian bornavirus. The data show also that these components can be identified in different tissues including feather calami (Figure 5).
The rabbit anti-recombinant matrix P16 antibody does not detect P16-like material in tissue homogenates or extracts of birds infected with bornavirus by the Western blot procedure, although the ELISA analysis using plasma from infected birds (Figure 1) suggests that many of these birds contain antibodies against this bornaviral component. A negative result in the Western blot is usually caused by the absence of the relevant antigen. However the results obtained with the sandwich ELISA procedure where P16 has to be recognized by two different antibodies (mammalian and avian, see below) suggest strongly that the bornavirus P16 matrix protein is present in persistently infected birds. The absence of the P16 component in the classical Western blot can also be the result of the inability of the antibodies to interact with the antigen, as a result of the destruction of relevant (discontinuous) epitopes by the Western blotting procedure [43, 44]. Further investigations [45] have shown however that discontinuous epitopes, which have been destroyed by heating in loading buffer, can often be regenerated during the Western blotting procedure. Since recombinant P16 by itself can readily be detected after Western blotting (Ln P16 in Figure 3d), destruction of discontinuous epitopes can be ruled out as a factor. Aggregation as a result of heating in SDS containing solutions [53] and consequently failure of transfer from the gel to the PVDF membrane has also been shown to interfere with Western blotting. Doing the Western blotting without heating the sample was shown to overcome this problem with Sars-corona virus membrane protein [53], but our attempts to apply this procedure to bornavirus P16 matrix protein failed (data not shown). Addition of recombinant P16 to a tissue homogenate in a Western blot may result in a signal at approximately 16kD, the strength of which (Figure 4a) is dependent on the relative amounts of brain homogenate of an infected bird and recombinant P16 used, suggesting that factors in the homogenate have an effect on the recognition of P16 by the antibodies, or that such factors interfere in an unknown way with the migration of the P16 out of the gel, or interfere with the binding of the P16 to the PVDF membrane and the positive outcome of the Western blot.
Similar to P16, probing of a Western blot of homogenates or extracts of tissues from a bird infected with bornavirus using rabbit anti-P29 glycoprotein antibody (Figure 3c) does not detect the glycoprotein in these homogenates using the current procedure, although the antibody recognizes recombinant P29 glycoprotein by itself again very well. Since the ELISA (Figure 1) showed that infected birds also do not contain detectable anti P29 antibodies, these results are most readily explained to be the consequence of the absence of the bornavirus glycoprotein in infected birds. This conclusion is further supported by the data in Fig 5b which show, that contrary to what has been found for P16, the presence of the brain homogenate in a Western blot does not interfere with the detection of added recombinant P29.
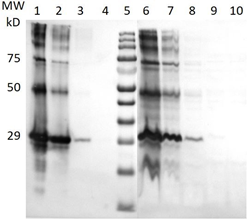
Figure 3: Western blots of a sun conure tissue homogenates after probing with rabbit antibodies against individual recombinant psittaciform bornavirus antigens. 3a = with anti P40; 3b = with anti-P24; 3c = with anti-P29; 3d = with anti-P16; 3e = with anti-P10; 3f = with anti-RNA polymerase. ST = MW standard; ln 0 = antigens used for antibody induction (in fig 3e P10-MBP, MW 53 kD is used instead of P10 (MW 10 kD)); ln 1 = brain sample of infected sunconure 442; ln 2 = liver of infected sun conure 188; ln 3 = brain of infected sun conure 188; ln 4 = brain of infected African grey parrot (Psittacus erithacus); ln 5 = brain of infected great green macaw (Ara ambiguous); ln 6 = brain of uninfected Eclectus parrot (Eclectus roratus); ln 7 = brain of Japanese quail (Coturnix coturnix japanicus); ln 8 = brain of a mouse (Mus musculus).
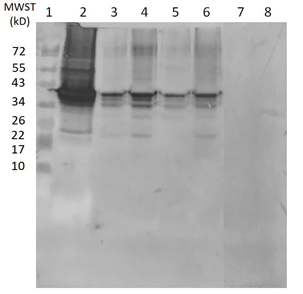
Figure 4a: Effect of the addition of recombinant P16 matrix protein to a Western blot of a sun conure brain homogenate. ln 1 = Protein MW standard; ln 2 = recombinant P16 protein ; ln 3 = rec. P16 protein + 2 µl SuCo 188 brain homogenate; ln 4 = 10 µl SuCo 188 homogenate; ln 5 = rec. P16 + 10 µl SuCo 188 homogenate; ln 6 = 10 µl Eclectus brain homogenate; ln 7 = rec. P16 + 10 µl Eclectus brain homogenate.
Figure 4b: Effect of the addition of recombinant P29 glycoprotein to a Western blot of a sun conure (SuCo188) brain homogenate. ln 1: 1000 ng rec. P29; ln 2: 100 ng rec. P29; ln 3; rec. 10 ng P29; ln 4: 1 ng P29; ln 5 = MW standard. ln 6 = 10 µl brain homogenate of SuCo188 + 1000 ng P29; ln 7 = as ln 6 but with 100 ng P29; ln 8 as ln 6 but with 10 ng P29; ln 9, as ln 6 but with 1 ng P29; ln 10 = 10 µl brain homogenate without addition of recombinant P29.
3.3 Detection of bornavirus P40 nucleoprotein in feather calami of naturally infected birds by classical western blotting
While the serological detection of anti-bornaviral antibodies is routinely applied for the diagnosis of an infection with bornavirus, it is an indirect procedure and does not directly indicate the actual presence of the antigens. Detection of bornaviral RNA by RT-PCR or protein by Western blotting is a direct method for the diagnosis of an infection. The relatively noninvasive procedures to obtain RNA from individual birds by using feathers, cloacal swabs or fecal material as a source of RNA, have therefore made RT-PCR a favoured procedure for the detection of bornavirus in parrots. The instability of RNA upon storage and handling as well as the intermittent shedding of the virus in the intestinal tract requiring repeated sampling [10] can present problems, in particular when it concerns collecting samples from wild birds in their native habitat. Although such problems can be overcome in part by the use of special solutions like “RNA-later” and dried feather stems where RNA can remain stable for several months [38], proteins are more stable than nucleic acids [40] and detection of bornavirus protein might therefore be advantageous as a diagnostic procedure. Therefore we also investigated the utility of protein analysis of feather calami for the detection of a bornavirus infection (Figure 5).
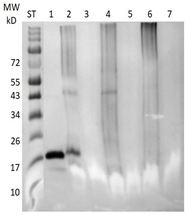
Figure 5: Classical Western blot of feather calami extracts after probing with a rabbit antibody against the psittacine bornavirus P40 nucleoprotein. ln 1 = Molecular weight standard; ln 2 = infected sun conure 171 brain homogenate; ln 3 = infected sun conure 5024 feathers; ln 4 = infected sun conure 171 feathers; ln 5 = infected sun conure 442 feathers; ln 6 = infected sun conure 188 feathers; ln 7 = uninfected African Grey parrot feathers; ln 8 uninfected lovebird feathers.
A semiquantitative investigation using a concentration series of recombinant P40 nucleoprotein showed that the classical Western procedure using this rabbit antibody preparation could detect a minimum of 1 µg of P40 nucleoprotein. As was mentioned before [38] using sun conure antibodies isolated from egg yolk, the P40 nucleoprotein could be detected in feathers that had been stored at room temperature for several years with a much greater sensitivity (3ng). Therefore, in order to improve the detection sensitivity of the identification of the P40 nucleoprotein with the rabbit antibody the samples were also analyzed using the automated Western system WES of ProteinSimple which uses chemiluminescent detection.
3.4 Detection of bornavirus P40 nucleoprotein in feather calami by automated western blotting
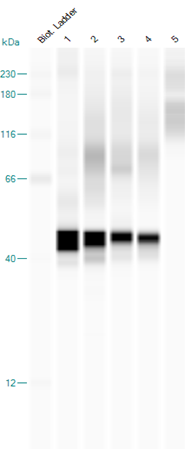
The results in Fig 6 show that this procedure also detects a protein of approximate 42 kD in feathers from birds infected with bornavirus, which had been stored for seven years, the maximum length of time feathers were available. A semiquantitative study, using a concentration series of recombinant P40 nucleoprotein showed that, using rabbit antibodies and chemiluminescent detection, the sensitivity was approximately 200 pg for P40, or 5,000 times more sensitive than a classical Western blot using horse radish peroxidase and rabbit antibodies and 12 times more sensitive than using sun conure antibodies.
Figure 6: Identification of P40 nucleoprotein in feather calami extracts of different psittaciformes by automated Western blotting. ln 1= infected sun conure 171 brain; ln 2 = infected sun conure 171 feathers; ln 3 = infected sun conure 442 feathers; ln 4 = infected sun conure 8 feathers; ln 5 = uninfected Eclectus parrot feathers.
3.5 Detection of bornaviral proteins by sandwich ELISA
Detection of viral antigens by Western blotting, manual or automatic, is still a rather complicated and labor intensive procedure. Sandwich ELISA or antigen capture ELISA [41, 51] has therefore become a standard procedure for the detection of pathogenic organisms, because it is technically relatively simple and has been used for the development of some rapid point of care diagnostic procedures.
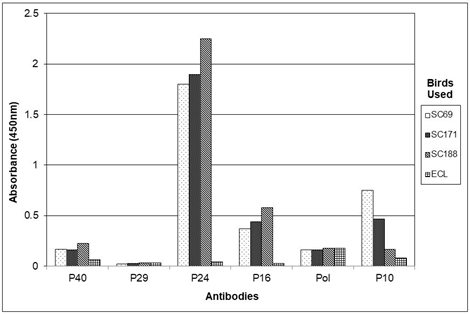
Figures 7a, 7b and 7c show the results of experiments in which the bornaviral P40, P29, P24 P16, P10 and RNA polymerase content of brain homogenates from a number of sun conures infected (SuCo 69, 171 and 188) and an Eclectus parrot (Eclectus roratus), not infected with bornavirus, were analyzed by sandwich ELISA using serum from a sun conure (SuCo188) infected with bornavirus (Figure 7a) or antibodies obtained from egg yolks of chickens immunized with recombinant psittaciform bornavirus antigens conjugated to MBP (P40, P29, P24 and P16) (Figure 7b) as the capture antibody. Figure 7c shows a photograph of the ELISA plate prior to the addition of the stop solution. The data show that whereas with both capture antibody sources (sun conure as well as chicken) the P24 and P16 antigens give clear signals with the infected birds, no P29 and almost no P40 antigens can be detected by the procedure used in these studies. Since in an ELISA, avian as well as rabbit antibodies recognize recombinant P40 very well this might indicate that the avian antibodies used for capture and the rabbit antibodies subsequently used for detection, recognize the same epitopes in the P40 nucleoprotein, rendering the rabbit antibodies unable to produce a signal, since the relevant epitopes were already occupied by the avian antibodies [41].The failure to detect the P29 glycoprotein antigen by the sandwich procedure agrees with the results obtained from the Western blots which indicated that the P29 antigen is not formed in a detectable quantity. In these studies the P10 and the polymerase antigen could only be investigated using the whole sun conure serum as source of the capture antibody since chicken antibodies against these two antigens were not available. The results show however that also the P10 protein produces a signal in birds infected with psittaciform bornavirus and not in noninfected birds. Further investigations showed that in addition to brain homogenates similar results can be obtained using other tissues or diagnostic materials, such as fecal matter, cloacal swabs, liver homogenates, feather calami etc (Figure 8).
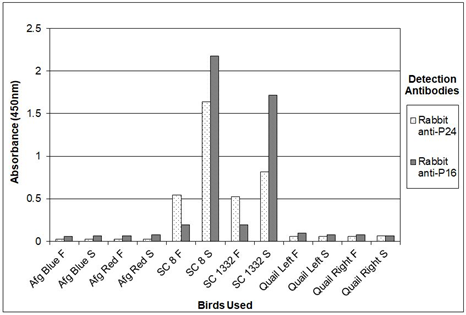
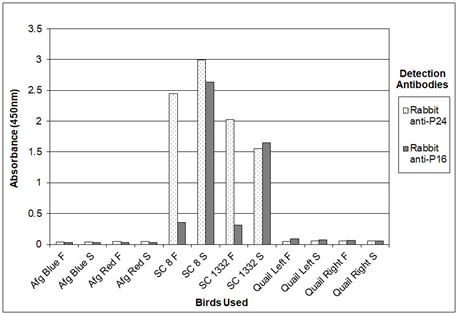
Figure 8 shows therefore the results of experiments where sandwich ELISA was used to detect the presence of psittaciform bornavirus among the birds in one of our aviaries. In these experiments feather calami or cloacal swabs extracts, prepared by simple vortexing in TE of the calami or swabs of two sun conures (SuCo8 and SuCo3225) persistently infected with psittaciform bornavirus 2 (as determined by RT-PCR of feather calami) were analyzed, and compared with material of two uninfected African grey parrots, a species which is susceptible to psittaciform bornavirus and two Japanese quail, a species which is not considered susceptible to the psittacine disease, but which supports the growth of psittaciform bornavirus in vitro in cell cultures [26, 27]. These birds had been housed in the same enclosure for five years. The sun conures were offspring of infected birds, the African grey parrots were offspring of uninfected parents. All birds were part of our own collection. The results show again that, using serum of an infected sun conure (SuCo188) or chicken antibodies against P40, P24 and P16 as the source of capture antibodies, and specific rabbit antibodies for P40, P24 and P16 as the detection antibodies, the P24 as well as the P16 antibodies give a clear signal using material of the infected sun conures, as opposed to the samples obtained from the uninfected African Grey parrots and Japanese quail which produced no or almost no signal. A quantitative analysis using a dilution series of recombinant P24 protein showed that the procedure, using horseradish peroxidase and TMB could detect minimally approximately 200 pg of P24 antigen. Repeating the same experiment with chicken anti P24-MBP antibody instead of sun conure serum revealed the same sensitivity. The P16 antibody produced a variable signal which makes this antigen therefore less suitable for a quantitative analysis. Storage of fecal or feather samples for a week at room temperature only marginally affected the results, and suggests that transportation at ambient temperature should cause no problems in the analysis. The experiment described in Figure 8, although done with only two infected sun conures and two uninfected African grey parrots shows that psittaciform bornavirus is not easily transmitted between psittacines through contact, as has been concluded also by other investigators [18, 19].
Figure 7: Analysis by sandwich ELISA of brain homogenates of 3 sun conures (SC69, SC171 and SC188) infected with psittacine bornavirus as compared to a brain homogenate of an uninfected Eclectus parrot (ECL). Figure 7a with serum of SuCo 188 (SCs) as the source of the capture antibodies; Figure 7b with chicken (CHI) anti-P40, -P29, -P24 and -P16 IgY as the capture antibody; Figure 7c is a photograph of the plate before the addition of the stop solution (HCl).
Figure 7c rows AB = SuCo69; Figure 7c rows CD = SuCo 171; Figure 7c rows EF = SuCo188; Figure 7c rows GH = Eclectus parrot. ln 1 – 6 with SuCo 188 serum as the source of the capture antibodies; ln 7 – 10 = with chicken anti-P40, -P29, -P24 or -P16 as the capture antibody resp. ln 11 – 12 = blank (no antibodies). Relevant rabbit antibodies were used as the detecting antibodies.
Figure 8: Analysis of feather calami (F) and cloacal swabs (S) of sun conures (SC 8 and SC 1332), infected with psittacine bornavirus and uninfected African Grey parrots (Afg Red and Afg Blue) or Japanese quail (Quail Left and Quail Right) for the presence of bornavirus P16 and P24 antigens by sandwich ELISA. Figure 8a with CHIgY P16 or CHIgY 24 as the capture antibody; Fig 8b with SuCo188 serum as the source of capture antibody against P16 and P24.
4. Discussion
The experiments described in this study show that of the six proteins, encoded in the psittaciform bornaviral genome, only one, the bornavirus P40 nucleoprotein can be reliably identified serologically in persistently infected birds by Western blotting, using rabbit antibodies raised against the recombinant bornaviral antigens. The glycoprotein (P29) does not occur or occurs in a quantity, undetectable by the Western blotting procedure used, resembling the results obtained earlier in rats, where bornaviral glycoprotein expression was also highly restricted in persistently infected animals [54], although the bornaviral glycoprotein is considered to be an important mediator in the cell to cell spread of bornavirus in infected animals [54-56]. The P16 matrix protein cannot be detected by Western blotting, probably because of intervening factors in the tissue homogenate, causing aggregation and failure to transfer from the gel to the PDVF membrane during the Western blotting procedure.
In Western blots the anti-P24 phosphoprotein antibody gives also positive results in uninfected birds and even in a mouse. The cross-reactivity of low avidity antibodies against human bornavirus P24 phosphoprotein with other host antigens had also been reported in the initial studies on the occurrence of bornaviral infection in psychiatric human patients [57-59], casting doubt on the involvement of bornavirus as an agent in these conditions. As in humans, the nature of the avian proteins, cross reacting with the rabbit anti-P24 antibody still has to be resolved. The rabbit anti-P24 antibody also detected in some infected birds material of a lower (<24kD) molecular weight that may be comparable to material found earlier by others in gerbil brain infected with bornavirus and which was the result of translation initiation at an internal AUG codon of the P24 gene [60]. Finally we have not studied the P10 and the bornaviral RNA polymerase in detail because attempts to form antibodies against these proteins in chickens failed.
Although the rabbit anti-recombinant matrix P16 antibody also does not detect P16-like material in Western blots of tissue homogenates or extracts of infected birds, the indirect ELISA (Figure 1) and the sandwich ELISA (Figures 7, 8) show that many infected birds contain anti-P16 antibodies as well as the P16 bornaviral protein. In the sandwich ELISA the latter component occurs in an amount roughly the same as the P24 phosphoprotein (Figure 7) and should therefore be, like the P24 protein, relatively easily detectable in the Western blot. The matrix protein is considered to be an important organizing, structural element in negative stranded RNA viruses [61], but it’s gene, like that of the glycoprotein, can nevertheless be deleted without problems [62-66] in persistently infected cells and tissues. Although a negative result in a Western blot is usually caused by the absence of the relevant antigen, it can however also be the result of the inability of the antibodies to interact with the antigen, as a result of the destruction of relevant (discontinuous) epitopes by the Western blotting procedure [43-45], shielding of epitopes by e.g. glycosylation [67] or inability of the protein to be transferred from the gel to the membrane as a result of aggregation [53]. Investigations by e.g. Zhou et al. [45] have shown however that discontinuous epitopes, which should have been destroyed by heating in SDS containing loading buffer, may be regenerated during the Western blotting procedure. Since recombinant P16 by itself can be readily detected after Western blotting (Figure 3d ln P16), permanent destruction of discontinuous epitopes can therefore be ruled out as a factor. Furthermore, the bornaviral matrix protein is not glycosylated [68] which rules out a role of shielding by glycosylation as an explanation for the negative result in the Western blot. Addition of a constant amount of recombinant P16 to different amounts of tissue homogenate in a Western blot may result in a signal at 16kD, the strength of which (Figure 4a) is dependent on the relative amounts of brain homogenate of an infected bird and recombinant P16 used. This may result in the appearance of a well-defined component of approximately 16 kD, when the relative amount of P16 is above a certain threshold. This suggests that factors in the homogenate have an effect on the recognition of P16 by the antibodies, or that such factors interfere with the binding of the P16 to the PVDF membrane, the migration of P16 from the gel to the membrane or the binding of the P16 to the antibody. The absence of natural anti-P16 antibodies in the brain of infected birds (de Kloet, unpubl. results) rules such antibodies out as a factor. The addition of recombinant P16 to a tissue homogenate may also result in a smear in the blot of material of a high molecular weight. This observation might therefore also indicate aggregation of the bornaviral P16 with unidentified cellular components, comparable with what has been found for other viruses by others [53]. The failure to detect the P16 component in the Western blot is specific for P16 and is not observed for P29, as the presence of brain homogenate in a Western blot fails to have an effect on the detection of this antigen (Figure 4b).
Like the previous study [38] the results described in the present investigation demonstrate that direct analysis of biological samples for the presence of a relevant viral component, is still preferable over the identification of the products of the immune system of the infected host such as antibodies. After all, aggregation of serum components, as a result of adverse conditions during sample collection and transportation, or cross reaction with antibodies of host components may produce false positive results (McHugh and de Kloet, manuscript in prep.) and birds infected with avian bornavirus may not produce anti-bornavirus antibodies at all [38].
The current study shows that the P40 nucleoprotein and the P24 phosphoprotein are the bornaviral protein antigens which may be most useful in a practical way for the diagnosis of a bornavirus infection through protein analysis, albeit using different technical approaches. A semiquantitative investigation using a concentration series of recombinant P40 nucleoprotein showed that the classical Western procedure using a rabbit antibody preparation, horse peroxidase labeled secondary antibody and TMB could detect minimally 1 µg of P40 nucleoprotein. The detection sensitivity of the identification of the P40 nucleoprotein with the rabbit antibody could be improved to 200 pg using the automated Western system WES of ProteinSimple which uses chemiluminescent detection. This latter procedure has been used a number of times before e.g. for the quantification of protein kinase C [69]. Taking the molecular weight of avian bornavirus P40 as 40,000, 200 pg P40 corresponds to approximately 3.0 x 109 molecules. The exact molecular composition of avian bornavirus is not known [31] but an analysis of another negatively stranded unsegmented RNA virus, vesicular stomatitis virus (VSV) [70] showed that this virus contains approximately 1200 nucleoprotein molecules per viral particle. Assuming that the relative molecular composition of bornavirus quantitatively resembles that of VSV, the detection limit of 200 pg P40 nucleoprotein corresponds then to approximately 3 x 106 viral particles, which is much less sensitive than RT-PCR detection of avian bornavirus which can detect 10 to 100 molecules of bornavirus RNA [71]. The greater stability of protein compared to RNA may however well outweigh this disadvantage. In addition, the RT-PCR may overestimate the quantity of virus because in most cases no distinction is made between viral genomic RNA and messenger RNA. Use of P24 in combination with chemoluminescent detection may well improve the detection limit further.
The detection of avian bornavirus P40 nucleoprotein or P24 phosphoprotein in feather stems, even after the feathers have been stored for several years, might also be helpful with the study of the history of bornavirus infection in psittaciformes, for it might eventually allow the analysis of e.g. museum or mummified specimens. Avian bornavirus infection of psittacines has been described already several years ago as “macaw wasting disease” [72] but molecular analysis of bornavirus-like sequences in the DNA of mammals [73, 74] and reptiles [75] suggests that borna virus may have infected these latter animals and their ancestors already more than 80 million years ago. Unfortunately the virtual absence of paleoviral sequences in the avian genome [76] presently prevents a similar analysis of bird viruses. The finding that the gene for RNA dependent RNA polymerase has entered the bat genome already almost 12 million years ago and is most closely related to its homologue in parrot bornavirus 2 suggests that avian bornavirus also has existed at least that long [74].
5. Conclusion
Bornavirus infection in psittaciform bird can be diagnosed by Western blot analysis of feather calami extracts, using the P40 nucleoprotein and chemoluminescent detection or the P24 phosphoprotein using the P24 phosphoprotein and sandwich ELISA horseradish peroxidase labeled secondary antibodies. Both procedures have a sensitivity of approximately 200 pg.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Amarasinghe GK, Aréchiga Ceballos NG, Banyard AC, et al. Taxonomy of the order Mononegavirales: update 2018. Arch Virol 163 (2018): 2283-2294.
- Kuhn JH, Dürrwald R, Bào Y, et al. Taxonomic reorganization of the family Bornaviridae. Arch.Virol 160 (2015): 621-632.
- HonkavuoriKS, Shivaprasad HL,Williams BL, et al. Novel bornavirus in psittacine birds with proventricular dilatation disease. Emerg Infect Dis 14 (2008): 1883-1886.
- KistlerAL,GanczA, Clubb, et al. Recovery of divergent avian bornaviruses from cases of proventricular dilatation disease: identification of a candidate etiologic agent. Virol J 5 (2008): 88.
- Fluck A,Enderlein D,Piepenbring A, et al. Correlation ofavianbornavirus-specific antibodies and viral ribonucleic acid shedding with neurologicalsignsand feather-damaging behaviour in psittacinebirds. Vet. Rec 184 (2019): 476.
- Horie M. Parrotbornavirusinfection: correlation with neurological signs andfeatherpicking? Vet Rec. 184 (2019): 473-475.
- Payne S,Shivaprasad HL,Mirhosseini N,et al. Unusual and Severe Lesions of Proventricular Dilatation Disease in Cockatiels (Nymphicus hollandicus) Acting as Healthy Carriers of Avian Bornavirus (ABV) and Subsequently Infected With a Virulent Strain of ABV. Avian Pathol 40 (2011): 15-22.
- Rinder M, Ackermann A, Kempf H, et al. Broad tissue and cell tropism of avianbornavirusin parrots with proventricular dilatation disease. J Virol 83 (2009): 5401-5407.
- de KloetSR, DorresteinGM. Presence of avian bornavirus RNA and anti-avian bornavirus antibodies in apparently healthy macaws. Avian Dis 53 (2009): 568-573.
- Hoppes SM, Tizard I, Shivaprasad HL. Proventricular dilatation disease; Diagnosis, Pathology, Prevalence, and control. The Veterinary Clinics of North America Exotic Animal Practice 16 (2013): 339-355.
- Fischer D, Neumann D, Purchase C, et al. The use of semen evaluation and assisted reproduction in Spix's macaws in terms of species conservation. Zoo Biol 33 (2014): 234-244.
- Alley MR, Gartrell BD. Wildlife dieases in New Zealand: recent findings and future challenges. N Z Vet J 67 (2019): 1-11.
- Clout MN.A celebration of kakapo progress in the conservation of an enigmatic parrot.Notornis53 (2006): 1-2.
- Encinas-Nagel N,Enderlein D,Piepenbring A, et al. Avian bornavirus in free ranging psittacine birds, Brazil. Emerg Infect Dis 20 (2014): 2103-2106.
- Kessler S, Heenemann K, Krause T, et al. Monitoring of free-ranging and captive Psittacula populations in Western Europe for avian bornaviruses, circoviruses and polyomaviruses. Avian Pathol 49 (2020): 119-130.
- Sassa Y, Bui VN, Saitoh K, et al. Parrot bornavirus-2 and -4 RNA detected inwildbird samples in Japan are phylogenetically adjacent to those found in petbirdsin Japan. Virus Genes 51 (2015): 234-243.
- VillanuevaI,GrayP,MirhosseiniN, et al. The diagnosis of proventricular dilatation disease: use of a Western blot assay to detect antibodies against avian Borna virus. Vet Microbiol 143 (2010): 196-201.
- Heckmann J, Enderlein D, Piepenbring AK, et al. Investigation of Different Infection Routes of ParrotBornavirusin Cockatiels. Avian Dis 61 (2017): 90-95.
- Rubbenstroth D, Brosinski K, Rinder M, et al. No contact transmission of avianbornavirusin experimentally infected cockatiels (Nymphicus hollandicus) and domestic canaries (Serinus canaria forma domestica). Vet Microbiol 172 (2014): 146-156.
- KerskiA, de Kloet AH, de Kloet SR. Vertical transmission of avian bornavirus in Psittaciformes: avian bornavirus RNA and anti-avian bornavirus antibodies in eggs, embryos, and hatchlings obtained from infected sun conures (Aratinga solstitialis). Avian Dis 56 (2012): 471-478.
- Lierz M, Piepenbring A, Herden C, et al. Verticaltransmissionofavianbornavirusin psittacines. Emerg Infect Dis 17 (2011): 2390-2391.
- Ogawa H, Sanada Y,Sanada N, et al. Proventricular Dilatation Disease Associated With Avian Bornavirus Infection in a Citron-crested Cockatoo That Was Born and Hand-Reared in Japan. J Vet Med Sci 73 (2011): 837-840.
- Briese T, Schneemann A, Lewis AJ, et al. Genomic organization of Borna disease virus. Proc Natl Acad Sci USA 91 (1994): 4362-4366.
- Schwardt M, Mayer D, Frank R, et al. The negative regulator of Borna disease virus polymerase is a non-structural protein. J Gen Virol 86 (2005): 3163-3169.
- Gough RE, Drury SE, Harcourt-Brown N, et al. Virus-like particles associated with macaw wasting disease.Vet Rec 139 (1996): 24.
- Horie M, Sassa Y, Iki H, Ebisawa K, et al. Isolation of avian bornaviruses from psittacine birds using QT6 quail cells in Japan. J Vet Med Sci 78 (2016): 305-308.
- Rubbenstroth D, Rinder M, Kaspers B, et al. Efficient isolation of avian bornaviruses (ABV) from naturally infected psittacine birds and identification of a new ABV genotype from a salmon-crested cockatoo (Cacatua moluccensis).Vet Microbiol 161 (2012): 36-42.
- Zimmermann W,Breter H,Rudolph M, et al. Borna disease virus: immunoelectron microscopic characterization of cell-free virus and further information about the genome. J Virol68 (1994): 6755-6758.
- SakudoA, Tanaka Y, Ikuta K. Capture of infectious borna disease virus using anionic polymer-coated magnetic beads. Neurosci Lett 494 (2011): 237-239.
- Mayer D,Baginsky S,Schwemmle Isolation of viral ribonucleoprotein complexes from infected cells by tandem affinity purification. Proteomics 5 (2005): 4483-4487.
- Schneider U, Naegele M, Staeheli P, et al. Active borna disease virus polymerase complex requires a distinct nucleoprotein-to-phosphoprotein ratio but no viral X protein. J Virol 77 (2003): 11781-11789.
- de Kloet AH, Kerski A, de Kloet SR. Diagnosis of avian Bornavirus infection in psittaciformes by serum antibody detection and reverse transcription assay using feather calami. J Vet Diagn Invest 23 (2011): 421-429.
- Guo J, Payne S, Zhang S, et al. Avian bornaviruses: diagnosis, isolation, and genotyping. Curr Protoc Microbiol 34-15I.1. (2014): 1-33.
- Ouyang N,Storts R,Tian Y, et al. Histopathology and thedetectionofavianbornavirusin the nervous system ofbirdsdiagnosed with proventricular dilatation disease. AvianPathol 38 (2009): 393-401.
- ChomczynskiP, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162 (1987): 156-159.
- Murray O, Turner D, Streeter K, et al. Apparent resolution of parrot bornavirus infection in cockatiels (Nymphicus hollandicus). Vet Med (Auckl) 8 (2017): 31-36.
- Berg M,Johansson M,Montell H, et al. Wild birds as a possible natural reservoir ofBornadiseasevirus. Epidemiol Infect 127 (2001): 173-178.
- McHugh JM, de Kloet SR. Discrepancy in the diagnosis of avianbornavirus infection of psittaciformes by protein analysis of feathers calami and enzyme-limked immunosorbent assay of serum antibodies. J Vet Diagn Invest 27 (2015): 150-158.
- Shearer PL, Sharp M, Bonne N, et al. A quantitative, real-time polymerase chain reaction assay for beak and feather disease virus. J Virol Methods 159 (2009): 98-104.
- Service RF. Researchers close in on ancientdinosaurproteins. Science 355 (2017): 441-442.
- Crowther JR. The ELISA Guidebook. Springer Protocols, Methods in Molecular Biology 2nd edition (2009). Humana Press, Totowa NJ, Chapter 2, 3.
- Schägger H. Tricine-SDS-Page. Nature Protocols 1 (2006): 16-32.
- Gorr TA, Vogel J. Western blotting revisited: Critical perusal of unappreciated technical issues. Proteomics Clinical Applications 9 (2015): 396-409.
- Kurien BT, Scofield RH. Western blotting: An Introduction. Methods in Molecular Biology 1312 (2015): 17-30.
- Zhou Y-H, Chen Z, Purcell RH, et al. Positive reactions on Western blots do not necessarily indicate the epitopes on antigens are continuous. Immunol Cell Biol 85 (2007): 73-78.
- Carlander D,Stålberg J,Larsson Chicken Antibodies: A Clinical Chemistry Perspective. Ups J Med Sci 104 (1999): 179-189.
- Gassmann M, Thömmes P, Weiser T, et al. Efficient production of chicken egg yolk antibodies against a conserved mammalian protein. FASEB J 4 (1990): 2528-2532.
- Polson A, von Wechmar MB, van Regenmortel MH. Isolation of viral IgY antibodies from yolks of immunized hens. Immunol Commun 9 (1980): 475-493.
- Porath J, Maisano F, Belew M. Thiophilic adsorption – a new method for protein fractionation. FEBS Lett 185 (1985): 306-310.
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72 (1976): 248- 254.
- Zrein M, Obert G, van Regenmortel MHV. Use of egg-yolk antibody for the detection of respiratory Syncytial virus in nasal secretions by ELISA. Arch Virol 90 (1986): 197-206.
- Zimmermann V, Rinder M, Kaspers B, et al. Impact of antigenic diversity on laboratory diagnosis of Avian bornavirus infections in birds. J Vet Diagn Invest 26 (2014): 769-777.
- Yi-Nung Lee, Li-Kuang Chen, Hsin-Chieh Ma, et al. Thermal aggregation of SARS-CoV membrane protein. J Virol Methods 129 (2005): 152-161.
- Werner-Keiss N,Garten W,Richt JA,et al. Restricted Expression of Borna Disease Virus Glycoprotein in Brains of Experimentally Infected Lewis Rats. Neuropathol Appl Neurobiol 34 (2008): 590-602.
- Lennartz F, Bayer K, Czerwonka N, et al. Surfaceglycoproteinof Borna disease virus mediates virus spread from cell to cell. Cell Microbiol 18 (2016): 340-354.
- Gonzalez-Dunia D, Cubitt B, Grasser FA, et al. Characterization of Borna disease virus p56 protein, a surfaceglycoproteininvolved in virus entry. J Virol 71 (1997): 3208-3218.
- Allmang U, Hofer M, Herzog S, et al. Low avidity of human serum antibodies for Borna disease antigens questions their diagnostic value. Mol Psychiatry 6 (2001): 329-333.
- Bechter K,Herzog S,Richt JA, et al. Pathogenicity of Borna disease virus in psychiatric and neurologic disorders of humans. Current status of research and critical comments. Nervenarzt 68 (1997): 425-430.
- Billich C,Sauder C, Frank R, et al. High-avidity human serum antibodies recognizing linear epitopes of Borna disease virus proteins. Biol Psychiatry 51 (2002): 979-987.
- Kobayashi T,Watanabe M,Kamitani W, et al. Translation initiation of a bicistronic mRNA of Borna disease virus: a 16-kDa phosphoprotein is initiated at an internal start codon. Virology 277 (2000): 296-305.
- Liljeroos L, Butcher SJ. Matrix proteins as centralized organizers of negative strand-sense RNA virions. Front Biosci 18 (2013): 696-715.
- Cathomen T, Mrkic B, Spehner D, et al. A matrix-less measles virus is infectious and elicits extensive cell fusion: consequences for propagation in the brain. EMBO J 17 (1998): 3899-3908.
- Daito T, Fujino K, Honda T, et al. A novel borna disease virus vector system that stably expresses foreign proteins from an intercistronic noncoding region. J Virol 85 (2011): 12170-12178.
- Fujino K, Yamamoto Y, Daito T, et al. Generation of a non-transmissive Borna disease virus vector lacking both matrix and glycoprotein genes. Microbiol Immunol 61 (2017): 380-386.
- InoueM,TokusumiY,BanH,et al. A new Sendai virus vector deficient in the matrix gene does not form virus particles and shows extensive cell-to-cell spreading. J Virol 77 (2003): 6419-6429.
- Yoshizaki M, Hironaka T, Iwasaki H, et al. Naked Sendai virus vector lacking all of the envelope-related genes: Reduced cytopathogenicity and immunogenicity. J Gene Med 8 (2006): 1151-1159.
- Seabright GE, Doores KJ, Burton DR, et al. Protein and Glycan Mimicry in HIV Vaccine Design. J Mol Biol 431 (2019): 2223-2247.
- Kraus I, Eickmann M, Kiermayer S, et al. Open reading frame III of borna disease virus encodes a nonglycosylated matrix protein. J Virol 75 (2001): 12098-12104.
- Chen J-Q, Heldman MR, Herrmann MA, et al. Absolute quantitation of endogenous proteins with precision and accuracy using a capillary Western system. Anal Biochem 442 (2013): 97-103.
- Thomas D,Newcomb WW,Brown JC,et al. Mass and molecular composition of vesicular stomatitis virus: a scanning transmission electron microscopy analysis. J Virol 54 (1985): 598–607.
- Wensman JJ,Thorén P,Hakhverdyan M,et al. Development of a real-timeRT-PCRassay for improveddetectionof Borna disease virus. J Virol Methods 143 (2007): 1-10.
- Gregory C, Latimer KS, Niagro F, et al. A review of proventricular dilatation syndrome.J Assoc Avian Vet 8 (1994): 69-75.
- Horie M, Honda T, Suzuki Y, et al. Endogenous non-retroviral RNA virus elements in mammalian genomes. Nature 463 (2010): 84-87.
- HorieM,KobayashiY,HondaT,et al. An RNA-dependent RNA polymerase gene in bat genomes derived from an ancient negative-strand RNA virus. Sci Rep 6 (2016): 25873.
- Hyndman TH, Shilton CM, Stenglein MD, et al. Divergent bornaviruses from Australian carpet pythons with neurological disease date the origin of extant Bornaviridaeprior to the end-Cretaceous extinction. PLoS Pathog 14 (2018): e1006881.
- Cui J, Zhao W, Huang Z, et al. Low frequency of paleoviral infiltration across the avian phylogeny. Genome Biology 15 (2014): 539-552.