Application of Atomic Absorption Spectroscopy to determine Mineral and Heavy Metal distribution level of Medicinal Plants
Article Information
Bushra Hina1*, Ghazala –H- Rizwani2, Uzma Naseeb3, Ambreen Huma4, Zeeshan Hyder5
1Department of Pharmacognosy, Institute of Pharmaceutical Sciences, Jinnah Sindh Medical University
2Director Hafiz Ilyas institute of Pharmacology and Herbal Sciences, Hamdard University, Pakistan
3 Department of Biochemistry, Sindh Medical College, Jinnah Sindh Medical University
4Department of Pharmacognosy, Faculty of pharmacy, Ziauddin University
5Department of Pharmacognosy, University of Karachi
*Corresponding Author: Bushra Hina, Department of Pharmacognosy, Institute of Pharmaceutical Sciences, Jinnah Sindh Medical University.
Received: 01 December 2023; Accepted: 11 December2023; Published: 28 December 2023.
Citation: Bushra Hina, Ghazala –H- Rizwani, Uzma Naseeb, Ambreen Huma, Zeeshan Hyder. Application of Atomic Absorption Spectroscopy to determine Mineral and Heavy Metal distribution level of Medicinal Plant. Journal of Analytical Techniques and Research 5 (2023): 26-32.
Share at FacebookAbstract
This study was done to apply the technique of atomic absorption spectrophotometery in the field of pharmacy to ensure the quality and purity of herbal plant material. In this regard samples of Justicia adhatoda were collected, dried, weighted, digested and analyzed using Atomic Absorption Spectrophotometer for their mineral contents (Copper, Chromium, Zinc, Cobalt, and Iron) as well as toxic heavy metal (Lead and Cadmium) contamination. A comparative study of two different sample preparation techniques i.e. dry ashing and wet digestion protocols was carried out in order to observe the effects of sample pre treatment on the qualitative and quantitative analysis prior to atomic absorption spectroscopic analysis. After spectroscopic analysis statistical evaluation of the data was performed using. On the basis of statistical evaluation using t-test (p<0.05), it was concluded that wet digestion method was found more significant for the analysis of Pb, Cu, Zn, Ni, and Fe. Moreover another comparison was also performed among 3 different samples collected from three various sites of Karachi city to detect changes in metallic composition. Application of one way ANOVA followed by Duncan test indicated no significant difference in Pb, Cd, and Zn concentrations collected from all three zones(p<0.05). It can be concluded that atomic absorption spectroscopy can successfully utilized to monitor toxic metal contamination as well as to determine essential metal profile in herbs.
Keywords
medicinal plants, Heavy metal toxicity, quality control, atomic absorption spectrophotometer.
medicinal plants articles; Heavy metal toxicity articles; quality control articles; atomic absorption spectrophotometer articles.
medicinal plants articles medicinal plants Research articles medicinal plants review articles medicinal plants PubMed articles medicinal plants PubMed Central articles medicinal plants 2023 articles medicinal plants 2024 articles medicinal plants Scopus articles medicinal plants impact factor journals medicinal plants Scopus journals medicinal plants PubMed journals medicinal plants medical journals medicinal plants free journals medicinal plants best journals medicinal plants top journals medicinal plants free medical journals medicinal plants famous journals medicinal plants Google Scholar indexed journals Heavy metal toxicity articles Heavy metal toxicity Research articles Heavy metal toxicity review articles Heavy metal toxicity PubMed articles Heavy metal toxicity PubMed Central articles Heavy metal toxicity 2023 articles Heavy metal toxicity 2024 articles Heavy metal toxicity Scopus articles Heavy metal toxicity impact factor journals Heavy metal toxicity Scopus journals Heavy metal toxicity PubMed journals Heavy metal toxicity medical journals Heavy metal toxicity free journals Heavy metal toxicity best journals Heavy metal toxicity top journals Heavy metal toxicity free medical journals Heavy metal toxicity famous journals Heavy metal toxicity Google Scholar indexed journals quality control articles quality control Research articles quality control review articles quality control PubMed articles quality control PubMed Central articles quality control 2023 articles quality control 2024 articles quality control Scopus articles quality control impact factor journals quality control Scopus journals quality control PubMed journals quality control medical journals quality control free journals quality control best journals quality control top journals quality control free medical journals quality control famous journals quality control Google Scholar indexed journals atomic absorption spectrophotometer articles atomic absorption spectrophotometer Research articles atomic absorption spectrophotometer review articles atomic absorption spectrophotometer PubMed articles atomic absorption spectrophotometer PubMed Central articles atomic absorption spectrophotometer 2023 articles atomic absorption spectrophotometer 2024 articles atomic absorption spectrophotometer Scopus articles atomic absorption spectrophotometer impact factor journals atomic absorption spectrophotometer Scopus journals atomic absorption spectrophotometer PubMed journals atomic absorption spectrophotometer medical journals atomic absorption spectrophotometer free journals atomic absorption spectrophotometer best journals atomic absorption spectrophotometer top journals atomic absorption spectrophotometer free medical journals atomic absorption spectrophotometer famous journals atomic absorption spectrophotometer Google Scholar indexed journals analytical techniques articles analytical techniques Research articles analytical techniques review articles analytical techniques PubMed articles analytical techniques PubMed Central articles analytical techniques 2023 articles analytical techniques 2024 articles analytical techniques Scopus articles analytical techniques impact factor journals analytical techniques Scopus journals analytical techniques PubMed journals analytical techniques medical journals analytical techniques free journals analytical techniques best journals analytical techniques top journals analytical techniques free medical journals analytical techniques famous journals analytical techniques Google Scholar indexed journals medicinal plants articles medicinal plants Research articles medicinal plants review articles medicinal plants PubMed articles medicinal plants PubMed Central articles medicinal plants 2023 articles medicinal plants 2024 articles medicinal plants Scopus articles medicinal plants impact factor journals medicinal plants Scopus journals medicinal plants PubMed journals medicinal plants medical journals medicinal plants free journals medicinal plants best journals medicinal plants top journals medicinal plants free medical journals medicinal plants famous journals medicinal plants Google Scholar indexed journals Pharmacognosy articles Pharmacognosy Research articles Pharmacognosy review articles Pharmacognosy PubMed articles Pharmacognosy PubMed Central articles Pharmacognosy 2023 articles Pharmacognosy 2024 articles Pharmacognosy Scopus articles Pharmacognosy impact factor journals Pharmacognosy Scopus journals Pharmacognosy PubMed journals Pharmacognosy medical journals Pharmacognosy free journals Pharmacognosy best journals Pharmacognosy top journals Pharmacognosy free medical journals Pharmacognosy famous journals Pharmacognosy Google Scholar indexed journals Pharmaceutical Sciences articles Pharmaceutical Sciences Research articles Pharmaceutical Sciences review articles Pharmaceutical Sciences PubMed articles Pharmaceutical Sciences PubMed Central articles Pharmaceutical Sciences 2023 articles Pharmaceutical Sciences 2024 articles Pharmaceutical Sciences Scopus articles Pharmaceutical Sciences impact factor journals Pharmaceutical Sciences Scopus journals Pharmaceutical Sciences PubMed journals Pharmaceutical Sciences medical journals Pharmaceutical Sciences free journals Pharmaceutical Sciences best journals Pharmaceutical Sciences top journals Pharmaceutical Sciences free medical journals Pharmaceutical Sciences famous journals Pharmaceutical Sciences Google Scholar indexed journals Biochemistry articles Biochemistry Research articles Biochemistry review articles Biochemistry PubMed articles Biochemistry PubMed Central articles Biochemistry 2023 articles Biochemistry 2024 articles Biochemistry Scopus articles Biochemistry impact factor journals Biochemistry Scopus journals Biochemistry PubMed journals Biochemistry medical journals Biochemistry free journals Biochemistry best journals Biochemistry top journals Biochemistry free medical journals Biochemistry famous journals Biochemistry Google Scholar indexed journals Pharmacology articles Pharmacology Research articles Pharmacology review articles Pharmacology PubMed articles Pharmacology PubMed Central articles Pharmacology 2023 articles Pharmacology 2024 articles Pharmacology Scopus articles Pharmacology impact factor journals Pharmacology Scopus journals Pharmacology PubMed journals Pharmacology medical journals Pharmacology free journals Pharmacology best journals Pharmacology top journals Pharmacology free medical journals Pharmacology famous journals Pharmacology Google Scholar indexed journals
Article Details
1. Introduction:
A variety of classical and advanced analytical techniques are available for the determination of heavy metals in medicinal plants [1]. Among all available techniques Atomic Absorption Spectroscopy (AAS) is one of the most advanced, sophisticated, reliable, and sensitive technique for the elemental analysis of medicinal plants. Flame atomic absorption spectroscopy (FAAS), Graphite furnace atomic absorption spectroscopy (GF-AAS) also known as Electro thermal AAS, Mercury Hydride system atomic absorption spectroscopy (MHS-AAS) are the various types of AAS to carry out both qualitative as well as quantitative analysis [2]. Prior to atomic absorption spectroscopy, matrix degradation of plant samples is very important step towards the accurate elemental analysis. Purpose of sample digestion and matrix degradation is to remove all organic matter leaving behind the metallic contents. Dry Ashing and wet digestion methods are the two important sample digestion protocols used for elemental analysis [3]. It is a common misperception that being natural in origin herbs and related herbal formulations is safe and can be taken as self-medication without consulting herbal specialist. But the idea is dangerously false because the green pharmacy has been originated from nature, so any disturbance in environment will ultimately affect the quality of phytopharmaceuticals made from it. Heavy metal contamination of herbal drugs is one of the most burning issues as far as the quality and safety of herbal medicaments is concerned [4-6]. It is very important to check the heavy metal contamination especially Pb, Cd, As, and Hg in crude drugs as well as in medicinal plants that are going to be used as raw material for herbal formulations [7]. Justicia adhatoda is very useful plant from Acanthaceae family that is widely utilized as sedative, expectorant, and antispasmodic. It is commonly known as Aroosa in Pakistan and its leaves are used to treat various respiratory tract diseases including cold, cough, bronchitis, chest diseases, pneumonia, asthma, tuberculosis, chronic bronchitis. Leaves are also effective in diarrhea, dysentery, and malaria fever. Major phytochemicals found in this medicinal plants are alkaloid vasicine, vasicol, adhatonine,vasicinone, and vasicinol responsible for its therapeutic activities [8-10]. Purpose of this study was to compare the effect of wet and dry digestion protocols for sample preparation/matrix degradation of J. adathoda leaves for its atomic absorption spectroscopy.
2. Material and Methods
2.1 Chemicals and reagents:
Certified Atomic Absorption stock standard solutions 1000 ± 5 ppm were procured from May and Baker Rhone. All chemicals utilized in research were analytical grade purchased from Sigma Aldrich. De ionized water was used for making serial dilutions.
2.2 Collectionand Treatment of medicinal plants:
Samples of Justicia adhatoda (100 g) were collected from local herbal market of Karachi city coded as region 1, region 2, and region 3. Sample was identified and authenticated by Prof. Dr. Ghazal H Rizwani. After collection samples were washed, dried, and grinded, labeled and stored in air tight amber colored jars till further use.
2.3 Digestion of plant samples for matrix degradation:
Samples of J. adhatoda were prepared for atomic absorption spectrophotometric analysis using two different protocols i.e. Dry ashing and Wet digestion protocols following the procedure mentioned in [3].
2.4 Wet digestion:
1 g sample was weighted and reflex heated with 10 ml of concentrated HNO3 until the formation of red nitrous oxide fumes has been ceased. After cooling at room temperature 4 ml 70% HCLO4 has been added and heated again till evaporation to a small volume. Solution was filtered and volume was made 50 ml with deionized water.
2.5 DryAshing
Ash of 1 g sample was prepared in a porcelain crucible by heating the sample at 500 °c overnight in a muffle furnace. After cooling ash was dissolved in 5 ml 20% HCL, warming the solution, then filtered and Volume was made up 50 ml with deionized water.
2.6 Instrumentation:
Atomic absorption spectrophotometer [3] with flame atomization (FAAS) was used for elemental analysis. All recommended operating parameters for the successful execution of analysis were followed summarized in Table 1.
All measurements were run in triplicate for the samples and standard solutions and the average of these three absorption signals (ppm) were used for subsequent calculations. Calibration curves for all tested metals were obtained after appropriate serial dilution of “certified Atomic Absorption stock standard solutions 1000 ± 5 ppm”. In order to maintain quality control and avoidance of any kind of degradation fresh Standard solutions for each metal was prepared.
Table 1: Instrument setting of atomic absorption spectrophotometer (Perkin Emmer 3030 B)
2.7 Data Analysis
Results observed from atomic absorption spectroscopy were recorded as ppm. This data was used to calculate the concentrations of heavy metals in mg present in per Kg sample of J. adhatoda. Statistical evaluation was performed using t-test at significance level of p<0.05 in order to find differences between wet digestion and Dry ashing protocols. While to observe statistically significant differences in heavy metals content among samples obtained from regions 1, 2, and 3 ANOVA test was applied following post hoc test.
3. Results
Concentration of toxic heavy metals (Pb, and Cd) as well as trace elements (Cu, Co, Ni, Zn, Fe, and Cr) in J. adhatoda samples was determined by two methods i.e. dry ashing and wet digestion. Graphical representation of results obtained from both protocols are mentioned in Figure1a, 1b, 1c, 1d, 1e, 1f, 1g, and 1h.
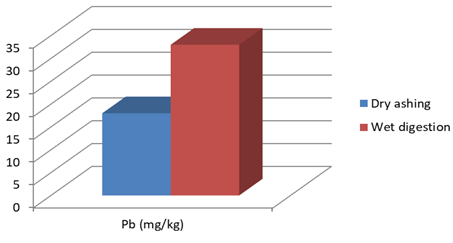
Figure 1a: Concentration of Pb in J.adhatoda after wet digestion and dry ashing
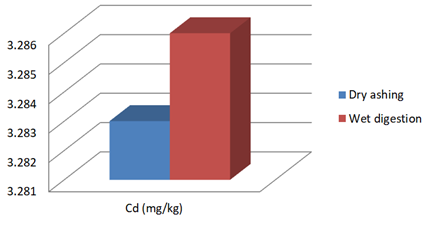
Figure 1b: Concentration of Cd in J.adhatoda after wet digestion and dry ashing
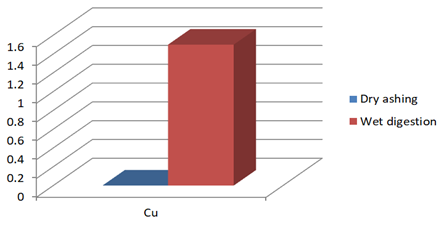
Figure 1c: Concentration of Cu (mg/Kg) in J.adhatoda after wet digestion and dry ashing
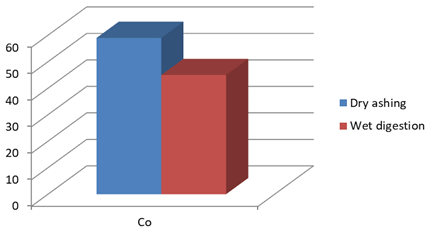
Figure 1d: Concentration of Co (mg/Kg) in J.adhatoda after wet digestion and dry ashing
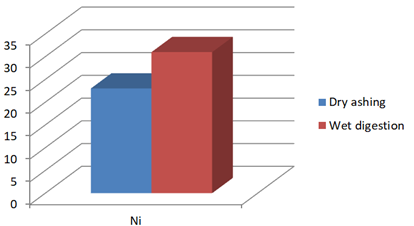
Figure 1e: Concentration of Ni (mg/Kg) in J.adhatoda after wet digestion and dry ashing
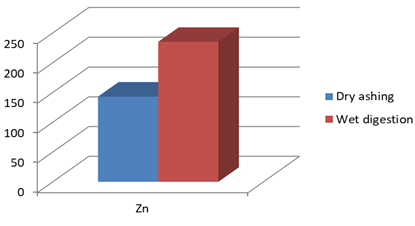
Figure 1f: Concentration of Zn (mg/Kg) in J.adhatoda after wet digestion and dry ashing
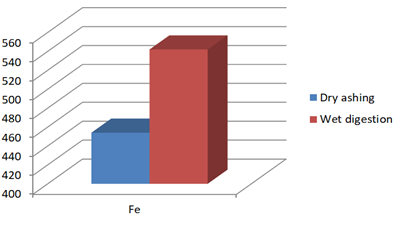
Figure 1g: Concentration of Fe (mg/Kg) in J.adhatoda after wet digestion and dry ashing
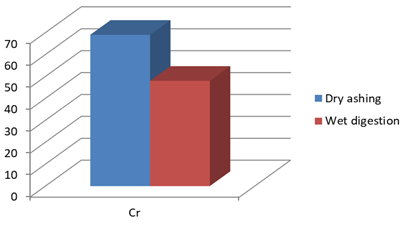
Figure 1h: Concentration of Cr (mg/Kg) in J.adhatoda after wet digestion and dry ashing
In order to find significant differences between the two digestion protocols opted for atomic absorption Spectrophotometric analysis, t test was used. Results of statistically significant comparison are given in Table 2.
Table 02: comparison of Heavy Metal contents (mg/kg) in Justica adhatoda followed by two different sample preparation methods opted for AAS
|
Heavy metals |
Concentration after Dry Ashing (mg/Kg) |
Concentration after Wet Digestion (mg/Kg) |
|
|
Toxic metals |
Pb |
18 |
33* |
|
Cd |
3.283** |
3.286 |
|
|
Essential Metals |
Cu |
0 |
1.5* |
|
Co |
59** |
45 |
|
|
Ni |
23 |
31* |
|
|
Zn |
143 |
235* |
|
|
Fe |
454 |
542* |
|
|
Cr** |
69 |
48 |
|
* Each value represents mean of three replicates. Results are significant at p< 0.05 using t-test.
** No significant difference is noted using t- test at p< 0.05
Table 03 represents the statistical comparison of heavy metal contents of J.adhatoda collected from three different regions of Karachi city using One Way ANOVA followed by Duncan test at p< 0.05.
Table 03: comparison of heavy metal contents (mg/kg) of J.adhatoda collected from different zones of Karachi city
|
Collection Zones |
Heavy Metals* (mg/kg) |
Trace elements* |
||||||
|
(mg/kg) |
||||||||
|
Lead |
Cadmium |
Copper |
Zinc |
Chromium |
Nickel |
Cobalt |
Iron |
|
|
Region 1 |
32 |
3.28 |
1.14b |
235 |
47.65b |
31.22b |
45.33b |
542.34a |
|
Region 2 |
17 |
0 |
0.00a |
228.3 |
0.00a |
57.09c |
53.66b |
1508b |
|
Region 3 |
29 |
0.003 |
1.98c |
234.7 |
.003a |
2.81a |
0.00a |
529.09a |
*each value represents the mean of three observations. Mean followed by same letter in a column represent no significant difference One Way ANOVA following Duncan test (p<0.05)
The average concentrations of toxic metals Pb and Cd detected were compared by different allowable limits provided by regulatory authorities and scientific contributors mentioned in Fig 2a and 2b. On the other hand fig 3a-3f represent the theoretical intakes of essential metals (Cu, Zn, Cr, Ni, Co, and Fe) if taken as 1-50 g sample.
4. Discussion
Justicia adhatoda is widely used both as crude drug in complementary and alternative system of medicines and as raw material for herbal formulations by herbal industries especially in respiratory tract diseases. Being natural in origin there are chances of contamination of toxic heavy metals like Pd, Cd, As, and Hg and other environmental pollutants. Moreover medicinal plants contain some essential metals also that are not only necessary for plant life and metabolism but also beneficial for human consumption provided that their concentrations remain in safety limit [11, 12]. So it is very important to detect any kind of toxic metal contamination as well as concentration of essential metals for the safety of consumers. Technique of AAS is successfully utilized in the qualitative and quantitative analysis of heavy metals in phytopharmaceuticals. However accuracy of analytical results is mainly dependent on complete matrix degradation of organic matter of plant samples. This paper describes the comparison of two different sample preparation techniques i.e. wet digestion and dry ashing protocols for the atomic absorption spectroscopy. Results indicates the presence of all trace elements in the samples treated with wet and dry digestion except Cu that was not detected using dry ashing procedure (Figures1a-1g). Although both methods can be successfully utilized for the detection of elemental composition but the wet digestion was found statistically significant for the analysis of lead, copper, zinc, nickel, and iron (Table 02). Karachi is counted as one of the most polluted city in the world. Due to this reasons all specimens of J adhatoda sampled from regions 1, 2, and 3 was not only found contaminated with Pb and Cd, but trace element levels were also towards higher side (Table 3, Figures 2a and 2b). Pb and Cd are the two major toxic metals that are not required by the body and can produce deleterious effects on various body organs and systems. The average contents of Pb and Cd were compared with different allowable limits given by different regulatory and scientific contributors [13, 14] that are mentioned in Figs. 2a and 2b. Unfortunately mean concentrations of Pb and Cd were found beyond the allowable limits.
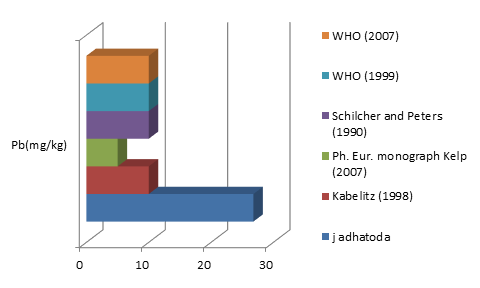
Figure2a: Comparison of Pb (mg/kg) content of J. adhatoda with allowable limits
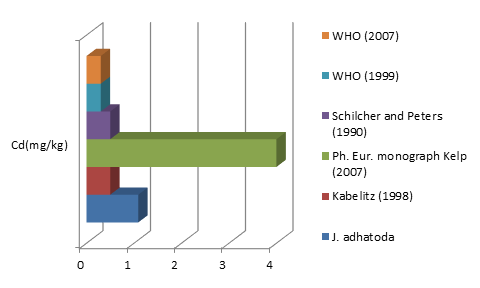
Figure2b: Comparison of Cd (mg/kg) content of J. adhatoda with allowable limits
Essential metals (Cu, Zn, Cr, Ni, Co, and Fe) are required in trace quantities to regulate general metabolism and physiological activities of human beings. However reference daily intakes are recommended for the safe consumption of theses trace elements 15-18]. Figures 3a-3f presents graphical comparisons of theoretical elemental intake upon consumption of J. adhatoda in the dose range of 1-50 g. I t can be concluded that higher the dose of medicinal plant, higher will be the amount of essential metals reach to the body. The herb is found as a good source of essential metals but quantities should not exceed the safety limits.
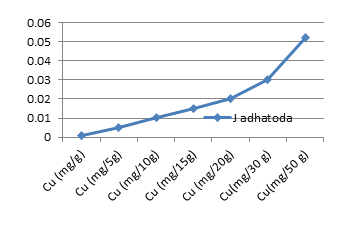
Figure 3a: Theoretical intake of Cu from 1-50g J.adhatoda
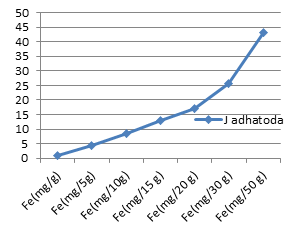
Figure 3b: Theoretical intake of Fe from 1-50g J.adhatoda
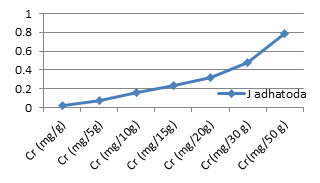
Figure 3c: Theoretical intake of Cr from 1-50g J.adhatoda
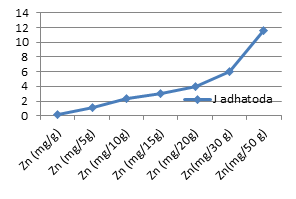
Figure 3d: Theoretical intake of Zn from 1-50 g J. adhatoda
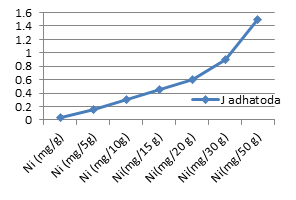
Figure 3e: Theoretical intake of Ni from 1-50 g J. adhatoda
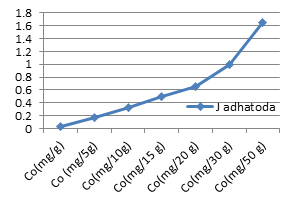
Figure 3f: Theoretical intake of Co from1-50g J.adhatoda
Conclusion:
Atomic absorption spectroscopy can be successfully employed in toxic heavy metal analysis as well as elemental profile using both wet digestion and dry ashing protocols in medicinal plants. However wet digestion method was found more superior then the dry ashing method in this regard. This analytical technique not only helps in maintaining the quality and purity of medicinal plants but also to monitor contaminants. AAS can be utilized in herbal industries quality control and research organizations working on the efficacy and safety of herbal medicines.
Study limitations:
Since the heavy metal uptake depends on various factors as well as environmental pollution, so their concentration may vary from batch to batch.
Acknowledgements:
Authors are very thankful to Fuel Research Centre (FRC, PCSIR) Karachi, Pakistan to provide technical support for this study.
References
- Dasgupta A, and Stableer C.H. Herbal Supplements, Efficacy, Toxicity, Interactions with Western Drugs and Effects on Clinical Laboratory Tests. John Wiley and Sons, Inc; Hoboken, New Jersey 17(2012): 16-19.
- Yuan X, Chapman R. Land ZhiqianWu.Analytical Methods for Heavy Metals in Herbal Medicines Phytochem. Anal 22 (2011): 189-198.
- Hina B, Rizwani G-H, Naseem S. Determination of toxic metals in some Herbal Drugs through Atomic Absorption Spectroscopy. Pak.J. Pharm.Sci 24 (2011): 353-358.
- Perkin Elmer. Analytical Methods for Atomic Absorption Spectroscopy. The Perkin-Elmer Cooperation, USA (1996): 138-143.
- Calixto J.B. Efficacy, Safety, Quality Control, Marketing, and Regulatory Guidelines for Herbal Medicines (Phytotherapeutic Agents). Brazilian Journal of Medical and Biological Research 33 (2000):179-189.
- Hina B, Rizwani G. H, Shareef H, et al. Trace Metal Detection in Some Medicinal Plants Used in Respiratory Tract Diseases through Atomic Absorption Spectroscopy in Karachi. The Journal of Phytochemistry. Photon 115 (2014): 276-285.
- Hina B, Rizwani G.H, Zahid H. Hematological screening of heavy metals among patients of asthma using medicinal herbs in Karachi, Pakistan. Pak. J. Pharm. Sci 27 (2014): 1899-1904.
- WHO Guidelines for assessing quality of Herbal medicines with reference to contaminants and residues (2007): 1, 13 and 19.
- Flora of Pakistan. http://www.efloras.org/florataxon.aspx?flora_id=5&taxon_id=220006995
- Bown D. New Encyclopedia of herbs and their uses. London: Dorling Kindersley Limited 248 (1995-2002).
- Said M H. Hamdard Pharmacopoeia of Eastern Medicine. Hamdard National Foundation, Pakistan by the Hamdard Academy 355 (1969).
- Hina B, Rizwani GH, Shareef H, et al. Atomic absorption spectroscopic analysis of some pakistani herbal medicinal products used in respiratory tract infections. Pak.J. Pharm.Sci 25 (2012):247-253.
- Quality Control Methods for Medicinal Plant Materials (1998): 62-63.
- Gasser U, Klier B, Kuhn A.V, et al. Current Findings on the Heavy Metal Content in Herbal Drugs. Pharmeuropa Scientific 1 (2009): 37-50.
- Divrikli U, Horzum N, Soylak M, et al. Trace heavy metal contents of some spices and herbal plants from western Anatolia, Turkey. International Journal of Food, Science, and Technology 41 (2006): 712-716.
- Council of Europe’s policy statements concerning materials and articles intended to come in contact with food stuffs Technical document, Guidelines on metals and alloys used as food contact materials (2001).
- Haider S, Naithani V, Barthwal J,et al. Heavy metal contents in some therapeutically important medicinal plants. Bull. Environ. Contam. Toxicol72 (2004): 119-127.
- Franceschini MA, Fantini S.Hemodynamic evoked response of the sensorimotor cortex measured noninvasively with near-infrared optical imaging. Psychophysiology 40 (2003): 548-560.
