Altered Neurotransmitter Metabolites in Vitamin B12 Deficiency
Article Information
Gregory Russell-Jones,
B12 Oils Pty Ltd, Sydney, Australia
*Corresponding Author: Gregory Russell-Jones, B12 Oils Pty Ltd, Sydney, Australia.
Received: 01 December 2023; Accepted: 13 December 2023; Published: 19 December 2023
Citation: Gregory Russell-Jones, Karl J. New. Altered Neurotransmitter Metabolites in Vitamin B12 Deficiency. Journal of Psychiatry and Psychiatric Disorders. 7 (2023): 223-227.
Share at FacebookAbstract
Vitamin B12 deficiency is often associated with neuronal symptoms such as difficulty sleeping, anxiety and depression. Using the Organic Acid Test as a tool it has been possible to examine various neurotransmitter markers to determine how they are altered in functional vitamin B12 deficiency, and therefor try to correlate these alterations with the symptoms. Depending upon the extent and type of functional vitamin B12 deficiency it has been found that lack of methyl B12 activity causes a dramatic increase in the levels of serotonin, dopamine and nor-epinephrine. Potentially the excess dopamine is causative for the increase in anxiety, whilst excess serotonin would eventually cause depression, and the lack of melatonin production, sleep issues. The findings have significant implications in the treatment of anxiety, depression and sleep disorders.
Keywords
Vitamin B2; Vitamin B12; Neurotransmitters; Anxiety; Depression; Autism; Developmental Delay; Organic acids test.
Vitamin B2 articles; Vitamin B12 articles; Neurotransmitters articles; Anxiety articles; Depression articles; Autism articles; Developmental Delay articles; Organic acids test articles.
Vitamin B2 articles Vitamin B2 Research articles Vitamin B2 review articles Vitamin B2 PubMed articles Vitamin B2 PubMed Central articles Vitamin B2 2023 articles Vitamin B2 2024 articles Vitamin B2 Scopus articles Vitamin B2 impact factor journals Vitamin B2 Scopus journals Vitamin B2 PubMed journals Vitamin B2 medical journals Vitamin B2 free journals Vitamin B2 best journals Vitamin B2 top journals Vitamin B2 free medical journals Vitamin B2 famous journals Vitamin B2 Google Scholar indexed journals Vitamin B12 articles Vitamin B12 Research articles Vitamin B12 review articles Vitamin B12 PubMed articles Vitamin B12 PubMed Central articles Vitamin B12 2023 articles Vitamin B12 2024 articles Vitamin B12 Scopus articles Vitamin B12 impact factor journals Vitamin B12 Scopus journals Vitamin B12 PubMed journals Vitamin B12 medical journals Vitamin B12 free journals Vitamin B12 best journals Vitamin B12 top journals Vitamin B12 free medical journals Vitamin B12 famous journals Vitamin B12 Google Scholar indexed journals Neurotransmitters articles Neurotransmitters Research articles Neurotransmitters review articles Neurotransmitters PubMed articles Neurotransmitters PubMed Central articles Neurotransmitters 2023 articles Neurotransmitters 2024 articles Neurotransmitters Scopus articles Neurotransmitters impact factor journals Neurotransmitters Scopus journals Neurotransmitters PubMed journals Neurotransmitters medical journals Neurotransmitters free journals Neurotransmitters best journals Neurotransmitters top journals Neurotransmitters free medical journals Neurotransmitters famous journals Neurotransmitters Google Scholar indexed journals Anxiety articles Anxiety Research articles Anxiety review articles Anxiety PubMed articles Anxiety PubMed Central articles Anxiety 2023 articles Anxiety 2024 articles Anxiety Scopus articles Anxiety impact factor journals Anxiety Scopus journals Anxiety PubMed journals Anxiety medical journals Anxiety free journals Anxiety best journals Anxiety top journals Anxiety free medical journals Anxiety famous journals Anxiety Google Scholar indexed journals Depression articles Depression Research articles Depression review articles Depression PubMed articles Depression PubMed Central articles Depression 2023 articles Depression 2024 articles Depression Scopus articles Depression impact factor journals Depression Scopus journals Depression PubMed journals Depression medical journals Depression free journals Depression best journals Depression top journals Depression free medical journals Depression famous journals Depression Google Scholar indexed journals Autism articles Autism Research articles Autism review articles Autism PubMed articles Autism PubMed Central articles Autism 2023 articles Autism 2024 articles Autism Scopus articles Autism impact factor journals Autism Scopus journals Autism PubMed journals Autism medical journals Autism free journals Autism best journals Autism top journals Autism free medical journals Autism famous journals Autism Google Scholar indexed journals Developmental Delay articles Developmental Delay Research articles Developmental Delay review articles Developmental Delay PubMed articles Developmental Delay PubMed Central articles Developmental Delay 2023 articles Developmental Delay 2024 articles Developmental Delay Scopus articles Developmental Delay impact factor journals Developmental Delay Scopus journals Developmental Delay PubMed journals Developmental Delay medical journals Developmental Delay free journals Developmental Delay best journals Developmental Delay top journals Developmental Delay free medical journals Developmental Delay famous journals Developmental Delay Google Scholar indexed journals Organic acids test articles Organic acids test Research articles Organic acids test review articles Organic acids test PubMed articles Organic acids test PubMed Central articles Organic acids test 2023 articles Organic acids test 2024 articles Organic acids test Scopus articles Organic acids test impact factor journals Organic acids test Scopus journals Organic acids test PubMed journals Organic acids test medical journals Organic acids test free journals Organic acids test best journals Organic acids test top journals Organic acids test free medical journals Organic acids test famous journals Organic acids test Google Scholar indexed journals S-Adenosylmethionine articles S-Adenosylmethionine Research articles S-Adenosylmethionine review articles S-Adenosylmethionine PubMed articles S-Adenosylmethionine PubMed Central articles S-Adenosylmethionine 2023 articles S-Adenosylmethionine 2024 articles S-Adenosylmethionine Scopus articles S-Adenosylmethionine impact factor journals S-Adenosylmethionine Scopus journals S-Adenosylmethionine PubMed journals S-Adenosylmethionine medical journals S-Adenosylmethionine free journals S-Adenosylmethionine best journals S-Adenosylmethionine top journals S-Adenosylmethionine free medical journals S-Adenosylmethionine famous journals S-Adenosylmethionine Google Scholar indexed journals National Health and Medical Research Council guidelines articles National Health and Medical Research Council guidelines Research articles National Health and Medical Research Council guidelines review articles National Health and Medical Research Council guidelines PubMed articles National Health and Medical Research Council guidelines PubMed Central articles National Health and Medical Research Council guidelines 2023 articles National Health and Medical Research Council guidelines 2024 articles National Health and Medical Research Council guidelines Scopus articles National Health and Medical Research Council guidelines impact factor journals National Health and Medical Research Council guidelines Scopus journals National Health and Medical Research Council guidelines PubMed journals National Health and Medical Research Council guidelines medical journals National Health and Medical Research Council guidelines free journals National Health and Medical Research Council guidelines best journals National Health and Medical Research Council guidelines top journals National Health and Medical Research Council guidelines free medical journals National Health and Medical Research Council guidelines famous journals National Health and Medical Research Council guidelines Google Scholar indexed journals
Article Details
1. Introduction
Vitamin B12 deficiency has been associated with many symptoms including weakness, fatigue, loss of vision, loss of sensation and dementia. Accompanying these symptoms many persons report other symptoms, such as difficulty sleeping, intestinal issues, depression, anxiety and mood changes [1, 2, 3, 4, 5, 6]. Measurement of vitamin B12 deficiency has generally been performed by simple analysis of levels in serum, however, recently it has become apparent that functional vitamin B12 deficiency may occur with normal or elevated serum vitamin B12, which may present as Paradoxical B12 deficiency, in which case additional measurements of biochemical markers, such as Methylmalonic Acid and homocysteine may be required to confirm the diagnosis of deficiency. More recently, in preliminary studies, it was established that there are other markers of functional vitamin B12 which can be identified through the use of an Organic Acids Test of urine [6]. In those preliminary studies, correlative data has shown that functional B12 deficiency is most often caused by functional vitamin B2 deficiency. These studies have been extended to examining the synthetic pathways for melatonin and epinephrine, both of which require a terminal methylation step during synthesis, and as such are dependent upon functional MethylB12 for activity.
2. Methods
Study sample Data analysis was carried out under the Australian National Health and Medical Research Council guidelines (NHMRC). Under these guidelines, all data was deidentified and steps were taken to ensure the anonymity and confidentiality of the data. Deidentification has consisted of absolute anonymity and confidentiality of the data, such that no specifics such as gender, ethnicity, Country of Origin, etc., is associated with any data point in the study. As such per the NHMRC guidelines: [7] 1. The research does not carry any risk to the participants 2. The benefits of the research are many and will be of considerable benefit to any past, current or future participants, and as such represent no harm. 3. The data is from over 1600 participants collected over 6 years and as such it would be impracticable to obtain consent from the participants. Further the participants had been notified at the time of analysis that data presented for analysis might potentially be used in research – but would be totally de-identified (which it has been). 4. There is no known reason why any of the participants would not have consented if they had been asked 5. Given the total de-identification of the data, there is absolute protection of their privacy 6. Data is only housed in one location and has only been assessed by one person, and as such the confidentiality of the data can be assured. 7. No financial benefits from the data are anticipated, rather the data will be used to help prevent and treat those to whom the data applies. 8. The waiver is not prohibited by State, Federal or International Law.
A retrospective analysis was performed upon data submitted to us for analysis from a cohort of individuals with various conditions, including autism spectrum disorder, chronic fatigue syndrome, healthy controls, and hypothyroidism. The data was submitted from countries including USA, Canada, United Kingdom, Ireland, Germany, Spain, France, Italy, Bulgaria, India, Sweden, Bulgaria, Serbia, Dubai, Croatia and Australia. No selection was made in the acceptance of data, with no data being rejected. Data is presented regardless of sex, or age. Ages varied from 1 year old to seventy-two years old. Metabolic analysis was performed on Organic Acid Test Data (1620 sets, Great Plains Laboratories, Lenexa, KS, USA), which had been submitted to us for interpretation. Data was tabulated in an Excel spreadsheet, and processed using the standard plotting functions in the program.
3. Results
Individual data is plotted as Scattergrams (Figures 1-6). Data comparisons were made between Methyl Malonic Acid, a standard marker of Adenosylcobalamin deficiency, glutaric acid, a marker of functional B2 deficiency, and the neurotransmitter metabolites homovanillic acid (HVA), Vannillyl mandelic acid (VMA), 5-hydroxyindole acetic acid (5HIAA), Quinolinic Acid (QA), and Kynurenic Acid (KA), as well as the ratio of QA:KA.
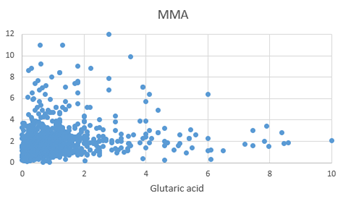
Figure 1: Scattergram of MMA values plotted against Glutaric acid.
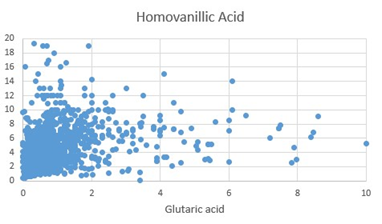
Figure 2: Scattergram of Homovanillic Acid (HVA) values plotted against Glutaric acid.
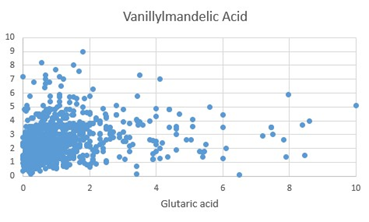
Figure 3: Scattergram of Vanillylmandelic Acid (VMA) values plotted against Glutaric acid.
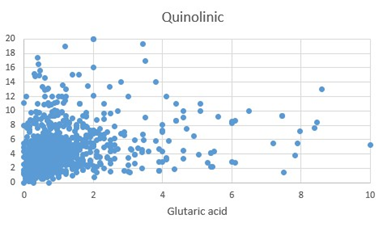
Figure 4: Scattergram of Quinolinic Acid (QA) values plotted against Glutaric acid
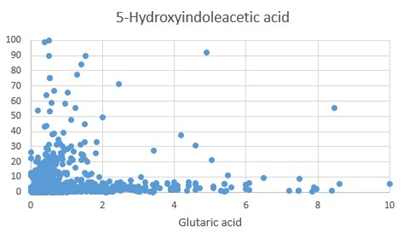
Figure 5: Scatter gram of 5-Hydroxyindoleacetic acid (5HIAA) values plotted against Glutaric acid.
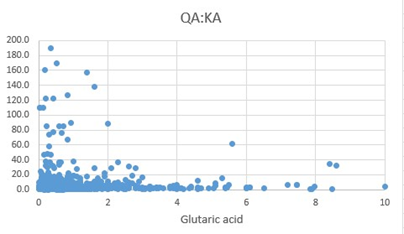
Figure 6: Scattergram of QA:KA ratio values plotted against Glutaric acid.
4. Discussion
It is apparent from the data that as functional vitamin B2 deficiency increases, as measured by urinary glutaric acid, so too does the standard vitamin B12 deficiency marker, MMA, supporting the role for functional vitamin B2 in the maintenance of vitamin B12 activity. Hence, during methylation of homocysteine to regenerate methionine, the enzyme methionine synthase donates the methyl group to homocysteine and becomes the inactive Co(I)B12. Incoming 5-methyltetrahydrofolate (5MTHF), then is used to regenerate MethylCo(III)B12. 5MTHF is “supplied” either from dietary sources or from the reduction of 5,10,methylene-THF by the FAD-dependent enzyme 5-methylenetetrahydroflate reductase. In the absence of FAD, there is reduced available 5MTHF within the cell. If levels of 5MTHF are low, Co(I)B12 is rapidly converted to inactive Co(II)B12, which requires the FMN/FAD dependent enzyme Methionine Synthase Reductase (MTRR) plus S-Adenosylmethionine (SAM), to regenerate MethylCo(III)B12.
{MTRR-FMN/FAD/NADPH}Co(II)B12-MTR + SAM => {MTRR-FMN/FAD/NAD}Co(III)B12-MTR + SAH
In the absence of FMN/FAD there is an accumulation of inactive Co(II)B12, and the reduced production of SAM, with a concomitant reduction in methylation reactions within the cell. Thus, as functional vitamin B2 decreases, vitamin B12 is gradually inactivated to Co(II)B12, and hence markers such as MMA and homocysteine will rise. Potentially this is why there is an association between symptoms of vitamin B12 deficiency in those with hypothyroidism [8].
4.1 Role of Methylcobalamin in the production of Adrenalin
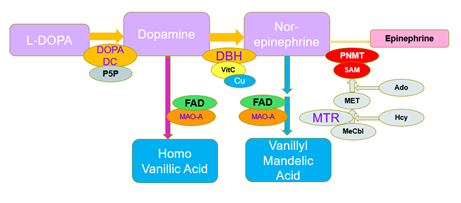
Figure 7:Synthetic pathway for Epinephrine synthesis with degradation products
The final step in the production of Epinephrine (adrenalin), is the methylation of Nor-epinephrine by phenylalanine-N-Methyl Transferase using S-Adenosylmethionine as the methyl donor. In functional B12 deficiency, the efficiency of this step is reduced, and so the two precursors accumulate inside the cell. In the OAT, increased levels of Homovanillic acid (HVA) and Vanillyl Mandelic acid (VMA) were measured (Figures 1, 2), representing decreased methylation. The levels of HVA and VMA may be somewhat compromised in functional B2 deficiency, as the reactivity of MAO decreases. Linkage of FAD to MAO, occurs via a covalent bond, during synthesis of MAO, whereas linkage of FAD and FMN to MTHFR and MTRR is ionic, and the activity of these two enzymes are greatly compromised as levels of FAD decrease, particularly in the non-wild-type phenotypes. Hence, theoretically there should be a greater reduction in the production and regeneration of methylCo(III)cobalamin as the levels of FMN and FAD decrease. Lack of methylation due to B12 insufficiency would explain the adrenal fatigue and POTS associated with vitamin B12 deficiency [9, 10].
4.2 Role of vitamin B12 in the production of Melatonin
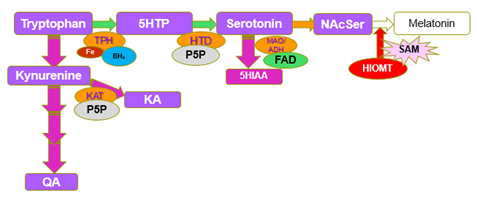
Synthesis of Melatonin, is a multi-step process, culminating in the methylation of NAcetyl-Serotonin, by the enzyme Hydroxy-Indole-O-Methyl Transferase, which uses S-Adenosylmethionine as the methyl donor. As methylation rate drops, the cell responds by increasing the uptake of tryptophan and producing more 5HTP => Serotonin => N-Acetylserotonin. This is accompanied by an increase in the rate of the Kynurenine pathway. The degradation products 5-hydroxyindole acetic acid (5HIAA), Kynurenic Acid (KA) and Quinolinic Acid (QA), become elevated as the rate of melatonin production decreases and the cell tries to compensate. Of note in the production of these degradation products is the reliance of FAD, for the activity of MAO, but more importantly the reliance of the enzymes, hydroxytryptophan decarboxylase (HTD), and Kynurenine Amino transferase on the active form of vitamin B6, Pyridoxal-5-phosphate (P5P). P5P must first be synthesized by the FMN-dependent enzyme pyridoxal kinase. Hence in a deficiency of FMN, the synthesis of both serotonin, and kynurenic acid is compromised. Lack of activity of HTD would in turn further increase the production of KA and QA, however the lack of P5P would then reduce the formation of KA and hence the ratio of QA:KA should increase. Lack of methylation due to B12 insufficiency would explain the lack of sleep that has been associated with vitamin B12 deficiency [11, 12, 13, 14, 15]. In addition, the upregulation of serotonin production in B12 insufficiency would also explain the depression, which is the second most common symptom associated with vitamin B12 deficiency [16, 17, 18, 19, 20, 21, 22].
5. Conclusion
Functional vitamin B12 deficiency has a dramatic effect on the efficacy of the synthesis of Melatonin and epinephrine, which is dependent upon the mechanism by-which the functional VB 12 deficiency occurs. In overt B12 deficiency, there is a compensatory increase in the levels of serotonin, and dopamine and nor-epinephrine, which in turn leads to build up the metabolites, 5HIAA, QA and KA (in the melatonin-related pathway), and VMA and HVA in the epinephrine pathway. If, the functional B12 deficiency, is due to a deficiency in FMN and FAD, as the deficiency becomes overt, the levels of serotonin may be reduced dramatically as too KA, 5HIAA and an increase in the QA:KA ratio. Eventually, if the B2 deficiency is great enough levels of VMA and HVA tend to plateau out. It would thus be expected that the symptoms of absolute B12 deficiency and functional B12 deficiency, mediated by functional B2 deficiency would be somewhat different in nature, and so may explain the wide range of symptoms associated with functional B12 deficiency. Arguably, the most important outcome of these findings will be in their application to the treatment of conditions such as depression and anxiety, which are currently treated with SSRIs. Clearly, in functional B12 deficiency, there is actually a huge over-production of serotonin, which would ultimately lead to down-regulation of serotonin receptors. In that case, rather than treat with SSRIs, these conditions should be treated by correcting the functional vitamin B12 deficiency, which is the cause of the conditions. Potentially this would be of considerable economic benefit to both the patients and to the health budgets of many countries.
Compliance with Ethical Standards
We declare that there are no potential conflicts of interest. Research did no involve human participants and/or animals. No Informed consent is required, all data is “blinded” and as such is anonymous.
References
- Sahu P, Thippeswamy H, Chaturvedi SK. Neuropsychiatric manifestations in vitamin B12 deficiency. Vitamins and hormones 119 (2022): 457-470.
- Lachner C, Steinl, NI, Regenold, WT. The neuropsychiatry of vitamin B12 deficiency in elderly patients. The Journal of neuropsychiatry and clinical neurosciences 24 (2012): 5-15.
- Clementz GL, Schade SG. The spectrum of vitamin B12 deficiency. American family physician 41(1990): 150-162.
- Hvas AM, Nexo E. Diagnosis and treatment of vitamin B12 deficiency--an update. Haematologica, 91 (2006): 1506-1512.
- Iqtidar N, Chaudary MN. Misdiagnosed vitamin B12 deficiency a challenge to be confronted by use of modern screening markers. JPMA. The Journal of the Pakistan Medical Association 62 (2012): 1223-1229.
- Russell-Jones, G. Paradoxical Vitamin B12 Deficiency: Normal to Elevated Serum B12, With Metabolic Vitamin B12 Deficiency. J Biol Today's World, 2022A, 11 (2022): 001-004
- Russell-Jones G. Functional Vitamin B12 Deficiency in Autism. J Psychia 25 (2022): 495
- Collins AB, Pawlak R. Prevalence of vitamin B-12 deficiency among patients with thyroid dysfunction. Asia Pacific journal of clinical nutrition 25 (2016): 221-226.
- Öner T, Guven B, Tavli V, et al. Postural orthostatic tachycardia syndrome (POTS) and vitamin B12 deficiency in adolescents. Pediatrics 133 (2014): 138-142.
- Pektas A, Koken R, Koca HB. Serum vitamin B-12 in children presenting with vasovagal syncope. Asia Pacific journal of clinical nutrition 27 (2018): 176–181.
- Al-Musharaf S, Alabdulaaly A, Bin Mujalli H, et al. Sleep Quality Is Associated with Vitamin B12 Status in Female Arab Students. International journal of environmental research and public health 18 (2021):
- Djokic G, Vojvodic P, Korcok D, et al. The Effects of Magnesium - Melatonin - Vit B Complex Supplementation in Treatment of Insomnia. Open access Macedonian journal of medical sciences, 7 (2019): 3101-3105.
- Ji X, Grandner MA, Liu J. The relationship between micronutrient status and sleep patterns: a systematic review. Public health nutrition 20 (2017): 687-701.
- Ekinci RMK, Balci S, Serbes M, et al. Decreased serum vitamin B12 and vitamin D levels affect sleep quality in children with familial Mediterranean fever. Rheumatology international 38 (2018): 83-87.
- Jahrami H, Alekri E, BaHammam AS, et al. The association between micronutrient status and sleep quality in patients with depression: a case-control study. Sleep & breathing = Schlaf & Atmung 25 (2021): 1571-1579.
- Bar-Shai M, Gott D, Marmor S. Acute psychotic depression as a sole manifestation of vitamin B12 deficiency. Psychosomatics 52 (2011): 384-386.
- Huijts M, Duits A, Staals J. Association of vitamin B12 deficiency with fatigue and depression after lacunar stroke. PloS one 7 (2012):
- Hanna S, Lachover L, Rajarethinam RP. Vitamin b12 deficiency and depression in the elderly: review and case report. Primary care companion to the Journal of clinical psychiatry 11 (2009): 269-270.
- Coppen A, Bolander-Gouaille C. Treatment of depression: time to consider folic acid and vitamin B12. Journal of psychopharmacology (Oxford, England) 19 (2005): 59-65.
- Berkins S, Schiöth HB, Rukh G. Depression and Vegetarians: Association between Dietary Vitamin B6, B12 and Folate Intake and Global and Subcortical Brain Volumes. Nutrients 13 (2021): 1790.
- Esnafoglu E, Ozturan, DD. The relationship of severity of depression with homocysteine, folate, vitamin B12, and vitamin D levels in children and adolescents. Child and adolescent mental health, 25 (2020): 249-255.
- Bharti A, Tevatia MS, Prakash J, et al. Are vitamin D, B12, and folate deficiency associated with depressive disorder? A case-control study. Industrial psychiatry journal, 32 (2023): 100-105.
