A Single Dose of Intravenous Human Amniotic Membrane-Derived Mesenchymal Stem Cells Limits Transmural Infarction, Reduces Fibrosis Size, and Improves Left Ventricular Systolic Function In The Myocardial Ischemic/ Reperfusion Model of Rats
Article Information
Jia-Wei Sung1†, Yen-Ting Yeh1,2†, Fu-Shiang Peng3, Yi-Chen Chen1, Ai-Hsien Li2, Shinn-Chih Wu1
1Department of Animal Science and Technology, National Taiwan University, Taipei, Taiwan
2Cardiology Division of Cardiovascular Medical Center, Far Eastern Memorial Hospital, New Taipei City, Taiwan
3Department of Obstetrics and Gynecology, Far Eastern Memorial Hospital, New Taipei City, Taiwan
*Corresponding Author: Shinn-Chih Wu, Department of Animal Science and Technology, National Taiwan University, Taipei, Taiwan.
†Two authors contributed equally to this article.
Received: 21 February 2023; Accepted: 01 March 2023; Published: 15 March 2023
Citation:
Jia-Wei Sung, Yen-Ting Yeh, Fu-Shiang Peng, Yi-Chen Chen, Ai-Hsien Li, Shinn-Chih Wu. A Single Dose of Intravenous Human Amniotic Membrane-Derived Mesenchymal Stem Cells Limits Transmural Infarction, Reduces Fibrosis Size, and Improves Left Ventricular Systolic Function In The Myocardial/Reperfusion Model of Rats. Archives of Nephrology and Urology. 6 (2023): 07-13.
Share at FacebookAbstract
Introduction: Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) have been shown with properties of immunomodulation, anti-inflammation, and low immunogenicity, which make them good candidates for cell therapies. In this study, we evaluated the therapeutic effects of Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) in a rat model of myocardial ischemia/reperfusion (I/R).
Methods: Myocardial ischemic/reperfusion was generated in Sprague Dawley rats. hAMSCs were injected into rats tail vein during coronary reperfusion. Dulbecco's phosphate-buffered saline (DPBS) were injected as negative control. Cardiac function was evaluated by echocardiography. Infarct size was analyzed by histochemical staining.
Results: Compared with negative control (DPBS group), hAMSCs group resulted in improvement of cardiac function and smaller infarct size at 28 days post-I/R.
Conclusion: A single dose of intravenous hAMSCs administered at the time of coronary reperfusion prevented transmural infarction, alleviated myocardial loss, and improved left ventricular systolic function in the rat I/R model.
Keywords
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs); Cell therapies; Myocardial ischemia/reperfusion (I/R); Echocardiography
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) articles; Cell therapies articles; Myocardial ischemia/reperfusion (I/R) articles; Echocardiography articles
Human amniotic membrane-derived mesenchymal stem cells articles Human amniotic membrane-derived mesenchymal stem cells Research articles Human amniotic membrane-derived mesenchymal stem cells review articles Human amniotic membrane-derived mesenchymal stem cells PubMed articles Human amniotic membrane-derived mesenchymal stem cells PubMed Central articles Human amniotic membrane-derived mesenchymal stem cells 2023 articles Human amniotic membrane-derived mesenchymal stem cells 2024 articles Human amniotic membrane-derived mesenchymal stem cells Scopus articles Human amniotic membrane-derived mesenchymal stem cells impact factor journals Human amniotic membrane-derived mesenchymal stem cells Scopus journals Human amniotic membrane-derived mesenchymal stem cells PubMed journals Human amniotic membrane-derived mesenchymal stem cells medical journals Human amniotic membrane-derived mesenchymal stem cells free journals Human amniotic membrane-derived mesenchymal stem cells best journals Human amniotic membrane-derived mesenchymal stem cells top journals Human amniotic membrane-derived mesenchymal stem cells free medical journals Human amniotic membrane-derived mesenchymal stem cells famous journals Human amniotic membrane-derived mesenchymal stem cells Google Scholar indexed journals Cell therapies; Myocardial ischemia/reperfusion articles Cell therapies; Myocardial ischemia/reperfusion Research articles Cell therapies; Myocardial ischemia/reperfusion review articles Cell therapies; Myocardial ischemia/reperfusion PubMed articles Cell therapies; Myocardial ischemia/reperfusion PubMed Central articles Cell therapies; Myocardial ischemia/reperfusion 2023 articles Cell therapies; Myocardial ischemia/reperfusion 2024 articles Cell therapies; Myocardial ischemia/reperfusion Scopus articles Cell therapies; Myocardial ischemia/reperfusion impact factor journals Cell therapies; Myocardial ischemia/reperfusion Scopus journals Cell therapies; Myocardial ischemia/reperfusion PubMed journals Cell therapies; Myocardial ischemia/reperfusion medical journals Cell therapies; Myocardial ischemia/reperfusion free journals Cell therapies; Myocardial ischemia/reperfusion best journals Cell therapies; Myocardial ischemia/reperfusion top journals Cell therapies; Myocardial ischemia/reperfusion free medical journals Cell therapies; Myocardial ischemia/reperfusion famous journals Cell therapies; Myocardial ischemia/reperfusion Google Scholar indexed journals Echocardiography articles Echocardiography Research articles Echocardiography review articles Echocardiography PubMed articles Echocardiography PubMed Central articles Echocardiography 2023 articles Echocardiography 2024 articles Echocardiography Scopus articles Echocardiography impact factor journals Echocardiography Scopus journals Echocardiography PubMed journals Echocardiography medical journals Echocardiography free journals Echocardiography best journals Echocardiography top journals Echocardiography free medical journals Echocardiography famous journals Echocardiography Google Scholar indexed journals Mesenchymal stem cells articles Mesenchymal stem cells Research articles Mesenchymal stem cells review articles Mesenchymal stem cells PubMed articles Mesenchymal stem cells PubMed Central articles Mesenchymal stem cells 2023 articles Mesenchymal stem cells 2024 articles Mesenchymal stem cells Scopus articles Mesenchymal stem cells impact factor journals Mesenchymal stem cells Scopus journals Mesenchymal stem cells PubMed journals Mesenchymal stem cells medical journals Mesenchymal stem cells free journals Mesenchymal stem cells best journals Mesenchymal stem cells top journals Mesenchymal stem cells free medical journals Mesenchymal stem cells famous journals Mesenchymal stem cells Google Scholar indexed journals ischemia/reperfusion articles ischemia/reperfusion Research articles ischemia/reperfusion review articles ischemia/reperfusion PubMed articles ischemia/reperfusion PubMed Central articles ischemia/reperfusion 2023 articles ischemia/reperfusion 2024 articles ischemia/reperfusion Scopus articles ischemia/reperfusion impact factor journals ischemia/reperfusion Scopus journals ischemia/reperfusion PubMed journals ischemia/reperfusion medical journals ischemia/reperfusion free journals ischemia/reperfusion best journals ischemia/reperfusion top journals ischemia/reperfusion free medical journals ischemia/reperfusion famous journals ischemia/reperfusion Google Scholar indexed journals coronary artery bypass grafting articles coronary artery bypass grafting Research articles coronary artery bypass grafting review articles coronary artery bypass grafting PubMed articles coronary artery bypass grafting PubMed Central articles coronary artery bypass grafting 2023 articles coronary artery bypass grafting 2024 articles coronary artery bypass grafting Scopus articles coronary artery bypass grafting impact factor journals coronary artery bypass grafting Scopus journals coronary artery bypass grafting PubMed journals coronary artery bypass grafting medical journals coronary artery bypass grafting free journals coronary artery bypass grafting best journals coronary artery bypass grafting top journals coronary artery bypass grafting free medical journals coronary artery bypass grafting famous journals coronary artery bypass grafting Google Scholar indexed journals Human amniotic membrane-derived mesenchymal stem cells articles Human amniotic membrane-derived mesenchymal stem cells Research articles Human amniotic membrane-derived mesenchymal stem cells review articles Human amniotic membrane-derived mesenchymal stem cells PubMed articles Human amniotic membrane-derived mesenchymal stem cells PubMed Central articles Human amniotic membrane-derived mesenchymal stem cells 2023 articles Human amniotic membrane-derived mesenchymal stem cells 2024 articles Human amniotic membrane-derived mesenchymal stem cells Scopus articles Human amniotic membrane-derived mesenchymal stem cells impact factor journals Human amniotic membrane-derived mesenchymal stem cells Scopus journals Human amniotic membrane-derived mesenchymal stem cells PubMed journals Human amniotic membrane-derived mesenchymal stem cells medical journals Human amniotic membrane-derived mesenchymal stem cells free journals Human amniotic membrane-derived mesenchymal stem cells best journals Human amniotic membrane-derived mesenchymal stem cells top journals Human amniotic membrane-derived mesenchymal stem cells free medical journals Human amniotic membrane-derived mesenchymal stem cells famous journals Human amniotic membrane-derived mesenchymal stem cells Google Scholar indexed journals
Article Details
1. Introduction
Myocardial infarction (MI) is a major cause of cardiovascular death around the world [1]. Despite the advances in mechanical reperfusion strategies such as percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG), there are still unmet needs of cardioprotection to alleviate myocardial necrosis and ischemia/reperfusion (I/R) injury during MI, which could result in post-MI cardiac remodeling and eventually lead to heart failure. Loss of myocardium and extended fibrosis play critical roles in the pathogenesis of post-MI cardiac remodeling [2], and therefore we need cardioprotective strategies in addition to opening the occluded coronary arteries.
Mesenchymal stem cells (MSCs) are multipotent adult stem cells with immunomodulatory and anti-inflammatory properties carrying minimal tumorigenesis potential [3], which make them good candidates for cell therapies. Recent studies demonstrated that MSCs reduced infarct size, alleviated cardiac fibrosis, and improved post-MI heart function in animal models [4, 5]. Therefore, MSC therapy has been one of the potential cardioprotective strategies that seems promising to attenuate LV remodeling after MI. MSCs derive from a variety of different tissues, and human amniotic membrane, which is discarded postpartum as medical waste, is one of good sources of MSCs. Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) are provided with large amounts, easy to harvest, with low immunogenicity, and without ethical issues [6-8]. Moreover, transplantation of hAMSCs has been demonstrated with encouraging results in prevention of fibrosis and functional improvement in different animal models [9, 10]. The aim of this study is to investigate the cardioprotective effect of intravenously administered hAMSCs to reduce infarct size and improve cardiac function in the cardiac I/R model of rats.
2. Methodology
2.1 Isolation and culture of hAMSCs
The samples of amnion membrane were obtained from caesarean sections, and all samples were collected from donors with informed consent. The Ethical Committee of the Far Eastern Memorial Hospital approved the study, which was conducted according to principles of the Helsinki Declaration. Fragments of human amnion membrane were washed in Hanks’ balanced salt solution (HBSS, calcium- and magnesium-free) and digested in 0.05% trypsin-EDTA for 1 hour at 37 °C. Then the membrane was washed with cold HBSS to discard the trypsin digest, transferred to new tubes, and added with digesting solution for 1 hour at 37 °C. The supernatant was collected, added with equal volume of HBSS, and centrifuged at 200 g for 4 minutes.
Harvested cells were cultured in polystyrene culture dishes at 37 °C with an atmosphere of 5 % CO2 and in low-glucose Dulbecco’s Modified Eagle Medium (Gibco Co.) supplemented with 10 % fetal bovine serum (Hyclone Co.), 1% sodium pyruvate (Gibco Co.), 1% penicillin-streptomycin (Gibco Co.), 1 mM Non Essential Amino Acids (Gibco Co.), 2 mM GlutaMAX (Gibco Co.), 0.1 mM beta-mercaptoethanol (Gibco Co.), and hEGF 10 ng/mL (Gibco Co.). The cultured medium was refreshed every 3 days. At confluence, the cells were harvested for passage with 1X trypLE (Gibco Co.).
2.2 Animals
24 Male Sprague Dawley rats at 8 weeks old were purchased from BioLASCO (Taiwan).
All experimental protocols were approved by the Institutional Animal Care and Use Committees of National Taiwan University, Taipei, Taiwan.
2.3 The cardiac I/R model of rats
The I/R model was performed as previously described [11]. In brief, the rats were anesthetized with sodium pentobarbital (80mg/kg, i.p.) and endotracheally ventilated with 2% isoflurane (in pure oxygen). Left-sided thoracotomy was performed by a small incision in the fourth intercostal space, and the left anterior descending coronary artery (LAD) was found and tied for 30 minutes before reperfusion. Ischemia was confirmed by electrocardiography and paleness of myocardium dominated by LAD. The tie on the LAD was released after 30 minutes of ligation to allow reperfusion.
2.4 Echocardiographic measurements
24 Rats were randomly divided into 4 groups (n = 6), including control group (rats with no surgical operation; positive control), sham group (only underwent thoracotomy without IR), negative control (after IR, injected 500μL DPBS via tail vein), hAMSCs group (after IR, injected 5*106 hAMSCs dissolving in 500μL DPBS). Echocardiography was performed before the I/R procedure and 1, 2, 3, and 4 weeks after the procedure (Figure 1) in anesthetized rats using a PROSPECT ULTRASOUND IMAGING SYSTEM. Left ventricular end-diastolic diameter (LVEDD), left ventricular end-systolic diameter (LVESD), and left ventricular fraction shortening (LVFS) were recorded from parasternal long-axis M-mode images using averaged measurements in 3 to 5 consecutive cardiac cycles in accordance with the American Society of Echocardiography guidelines. Left ventricular end-diastolic and end-systolic volumes (LVEDV and LVESV) were calculated from bidimensional long-axis parasternal views taken through the infarcted area by means of the single-plane area-length method [12]. Left ventricular ejection fraction (LVEF) was calculated as following: LVEF = (LVEDV-LVESV)/LVEDV) × 100%.
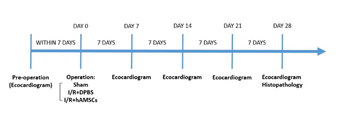
Figure 1: Study flowchart.
2.5 Pathological assessment
Four weeks after surgery, the rats were euthanized to remove the heart. The heart was simply trimmed to eliminate the atrium after immersion in 10% Neutral buffered formalin (NBF) for 72 hours. The remainder of the heart was cut into four pieces with equal width. These pieces were embedded in paraffin and cut into 5-μm slices, which were stained with either hematoxylin and eosin to detect cellular infiltration or with Masson’s trichrome stain (Sigma) to detect fibrosis and assess its area. LV anterior wall thickness was measured and averaged. The ratio of fibrotic area to entire LV cross-sectional area was calculated with Image J software (Version 1.51j8, NIH).
2.6 Mesenchymal Trilineage Differentiation Tests of hAMSCs
The hAMSCs were cultured using MesenCult™ Adipogenic Differentiation Kit (STEMCELL TECHNOLOGIES, Inc, Canada) and MesenCult™ Osteogenic Differentiation Kit (STEMCELL TECHNOLOGIES, Inc., Canada) to induce differentiation. Cell pellets (containing 2 × 106) were cultured for 21 days in MesenCult™-ACF Chondrogenic Differentiation Kit (STEMCELL TECHNOLOGIES, Inc, Canada) to induce differentiation. Control cells for all treatments were cultured under normal conditions as previously described. After the induced differentiation stage, the cells were stained with alizarin red to assess osteogenic differentiation, Oil Red O to assess adipogenic differentiation, and Alcian blue for chondrogenic differentiation. Chondrogenic pellets were paraffin-embedded and 3 μm sections were taken, which were then rehydrated and further stained with 1% Alcian blue solution.
2.7 Flow cytometry
hAMSCs were identified via flow cytometry with FITC-conjugated antibodies against FITC-anti CD90 (BD Bioscience, Franklin Lakes, NJ, USA, 1:100), FITC-anti CD45 (eBioscience, San Diego, CA, USA, 1:100), FITC-anti CD34 (eBioscience, 1:100), FITC-anti CD11b (eBioscience, 1:100), FITC-anti CD105 (eBioscience, 1:100), FITC-anti CD73 (eBioscience, 1:100), FITC-anti CD14 (eBioscience, 1:100), and FITC-anti mouse IgG as negative control.
2.7 Statistical analysis
All data were expressed as mean ± standard error. In echocardiographic measurement, the paired t-test and unpaired t-test were used for difference comparison. One-way analysis of variance (ANOVA) followed by Scheffé's post-hoc tests of the least significant difference was used in the comparison of LV anterior wall thickness. P < 0.05 was considered statistically significant.
3. Results
3.1 Characterization of hAMSCs
In appearance, hAMSCs displayed properties of MSCs including compact colonies, adhesion, and spindle-shaped morphology (Figure 2). Characterization was done by surface marker analysis using flow cytometry. They were negative for CD34, CD11b, CD45, and HLA-DR and positive for CD105, CD90, CD73, and CD44 mesenchymal markers (Figure 3). They were capable of tri-lineage differentiation into adipocytes, chondrocytes, and osteoblasts in vitro (Figure 4).
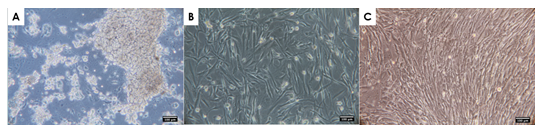
Figure 2:Cell morphology of hAMSCs
(A) hAMSCs on day 1 of primary culture. (B) Fibroblastoid morphology observed at passage 4. (C) Fibroblastoid morphology observed at passage 10. Scale bar = 100 μm.
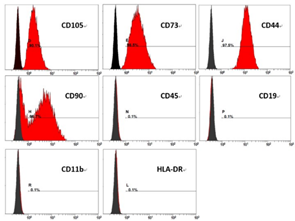
Figure 3:FACS analysis of surface marker expression on hAMSCs at passage 10.
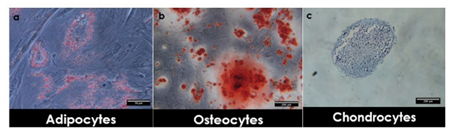
Figure 4: In vitro differentiation of hAMSCs into mesodermal lineages. Cells were cultivated in adipogenic, osteogenic, or chondrogenic differentiation medium.
After culture, adipogenic differentiation of cells was shown by intracellular accumulation of neutral lipids stained with Oil Red O solution. Scale bar = 50 μm. (b) Formation of a mineralized matrix after osteogenic differentiation was observed by Alizarin-Red staining. Scale bar = 200 μm. (c) Chondrogenic differentiation was shown by staining with Alcian-Blue. Scale bar = 200 μm.
3.2 Intravenous administration of hAMSCs improved
cardiac function in the I/R model
The rats were randomized into 4 groups. The control group (n=6) was the group with healthy rats that did not undergo any procedure. The sham group (n=6) underwent thoracotomy but the LAD was not ligated. The negative control (n=6) underwent the whole I/R procedure, and 500 μL DPBS without hAMSCs was injected via the tail vein after reperfusion. The hAMSC group (n=6) underwent the whole I/R procedure and was injected with 5 x 106 hAMSCs (passage 10) suspended in 500 μL DPBS via the tail vein. In 4 weeks after the I/R procedure, no rat died in each group, implying that intravenous treatment of hAMSCs was safe for rats. There was no significant difference in LVEF and LVFS between the sham group and the control group. In the hAMSC group, the LVEF and LVFS were significantly higher than those of the negative control that received DPBS without hAMSCs throughout the whole 4-week duration (Figure 5 & 6).
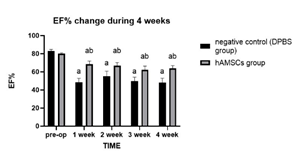
Figure 5: Comparison of LVEF between the hAMSC group and the negative control during the 4-week period after the procedure
“a” indicates significant difference (P < 0.05) from pre-op, and “b” indicates significant difference (P < 0.05) from the negative control.
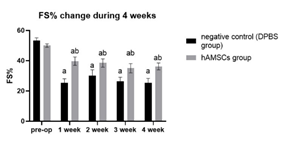
Figure 6: Comparison of LVFS between the hAMSC group and the negative control during the 4-week period after the procedure.
“a” indicates significant difference (P < 0.05) from pre-op. “b” indicates significant difference (P < 0.05) from the negative control
3.3 Intravenously administered hAMSCs prevented
transmural infarction and reduced LV fibrosis
Cross sections of the harvested hearts were examined with H & E staining and Masson trichrome staining 28 days after the I/R procedure. The overall structure of the LV remained unchanged in the sham group compared to that in the control group. However, an overtly dilated LV and thin LV anterior wall were observed in the negative control (Figure 7). Quantitative analysis showed that there was no significant difference in LV anterior wall thickness among the control group, the sham group, and the hAMSC group, but the LV anterior wall in the negative control was significantly thinner than that of the other groups (Figure 8). Fibrotic tissue in LV was marked by blue color with Masson trichrome staining. There was barely blue-stained tissue in the control group and the sham group. In the negative control, extensive transmural fibrosis of LV wall could be observed. Notably, there was very little transmural fibrosis in the hAMSC group, and most of the fibrotic tissue was limited in subendocardial regions (Figure 9). Quantitative analysis showed the ratio of fibrotic area to total cross sectional area of LV was significantly reduced in the hAMSC group compared to that in the negative control (Figure 10).
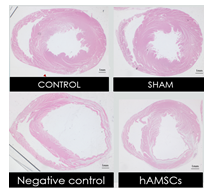
Figure 7:Representative LV sections of the harvested hearts at midventricular level with H & E staining.
Scale bar = 1 mm.
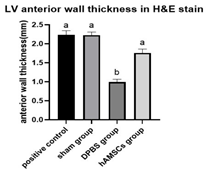
Figure 8:Averaged LV anterior wall thickness of the harvested hearts.
”a” indicates significant difference (P < 0.05) from the DPBS group (negative control). “b” indicates significant difference (P < 0.05) from the control group.
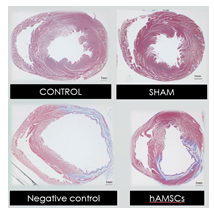
Figure 9: Representative LV sections of the harvested hearts at midventricular level with Masson trichrome staining.
Scale bar = 1 mm.
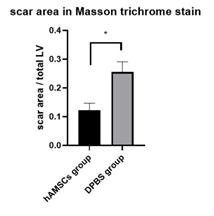
Figure 10: The ratio of fibrotic area to total cross sectional area of LV was significantly reduced in hAMSCs group.
* P < 0.05
4. Discussion
In clinical practice, LV transmural infarction is commonly seen when a coronary artery is totally obstructed in the setting of acute myocardial infarction (AMI) even if the culprit vessel is timely opened with primary PCI. Unfortunately, transmural infarction often leads to consequences including infarct expansion, excessive fibrosis, LV aneurysms, and eventually heart failure [13]. In this study, we successfully established a rat I/R model that mimics the clinical setting of AMI undergoing timely coronary reperfusion. In the negative control, extensive LV transmural fibrosis and marked loss of LV myocardium developed despite rapid restoration of blood flow of the ligated LAD in the I/R procedure. On the other hand, histological examination in the hAMSC group showed very little transmural fibrosis, and the LV myocardium was largely preserved since there was no significant thinning of the anterior wall. The data of serial echocardiographic measurement were consistent with histological findings. The hAMSC group had significantly better LV systolic function compared to the negative control. These results demonstrated a single dose of intravenous hAMSCs at the moment of coronary reperfusion could prevent transmural infarction and alleviate LV remodeling, which translated into better post-MI LV systolic function.
Amniotic membrane (AM), traditionally a medical waste, has been employed as scaffolding material in tissue engineering applications such as skin dressing or the graft for corneal treatment [14, 15], which have shown the safety of allogeneic use of AM tissue. In recent years, hAMSCs, isolated from human AM, have been demonstrated with good differentiation potentials, immunomodulatory properties, and low immunogenicity [6-8]. Studies demonstrated that xenotransplantation of hAMSCs resulted in minimal acute graft rejection reaction, making them excellent candidates of cell therapy [16-19].
Clinical studies have shown that autologous bone marrow cells or MSCs exhibited some benefits in AMI patients [20-22]. However, the therapeutic effect of autologous cell therapy could be largely limited by several factors. The timing of cell administration in clinical studies was likely suboptimal since procedures of harvest, culture, and expansion of autologous cells take at least several days, which might have already passed the “golden time” of cardioprotection. The dosage of administered cells was limited to the donor factor and short expansion time, which also made quality control barely possible. Allogeneic hAMSCs could be the solution of above problems since their property of low immunogenicity and proven safety of allogeneic use of human AM. Given the abundant sources of hAMSCs, which could be collected from daily caesarean section procedures and made into a prestored product, the dosage and quality of cells would be problems no more. Furthermore, intracoronary or intravenous hAMSCs could be given right at the time of coronary reperfusion to achieve best cardioprotective effect, which was demonstrated in this animal study to prevent transmural infarction, alleviate myocardial loss, improve LV systolic function, and possibly reduce heart failure. Future studies are warranted.
5. Conclusion
A single dose of intravenous hAMSCs administered at the time of coronary reperfusion prevented transmural infarction, alleviated myocardial loss, and improved left ventricular systolic function in the rat I/R model.
Funding acknowledgments
This research was funded by the Ministry of Science and Technology, R.O.C. (111-2313-B-002-012).
6. References
- Zaman S, Kovoor P. Sudden cardiac death early after myocardial infarction: pathogenesis, risk stratification, and primary prevention. Circulation 129 (23) (2014): 2426-2435.
- Hinderer S, Schenke-Layland K. Cardiac fibrosis - A short review of causes and therapeutic strategies. Adv Drug Deliv Rev 146 (2019): 77-82.
- Saeedi P, Halabian R, Imani Fooladi AA. A revealing review of mesenchymal stem cells therapy, clinical perspectives and Modification strategies. Stem Cell Investig 6 (2019): 34.
- Amado LC, Saliaris AP, Schuleri KH, St John M, Xie JS, et al. Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA 102 (32) (2005): 11474-11479.
- Lee RH, Pulin AA, Seo MJ, Kota DJ, Ylostalo J, et al. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell 5 (1) (2009): 54-63.
- Kang JW, Koo HC, Hwang SY, Kang SK, Ra JC, et al. Immunomodulatory effects of human amniotic membrane-derived mesenchymal stem cells. J Vet Sci 13 (1) (2012): 23-31.
- Farhadihosseinabadi B, Farahani M, Tayebi T, Jafari A, Biniazan F, et al. Amniotic membrane and its epithelial and mesenchymal stem cells as an appropriate source for skin tissue engineering and regenerative medicine. Artif Cells Nanomed Biotechnol 46 (2018): 431-440.
- Ragni E, Papait A, Perucca Orfei C, Silini AR, Colombini A, et al. Amniotic membrane-mesenchymal stromal cells secreted factors and extracellular vesicle-miRNAs: Anti-inflammatory and regenerative features for musculoskeletal tissues. Stem Cells Transl Med 10 (7) (2021): 1044-1062.
- Kubo K, Ohnishi S, Hosono H, Fukai M, Kameya A, et al. Human Amnion-Derived Mesenchymal Stem Cell Transplantation Ameliorates Liver Fibrosis in Rats. Transplant Direct 1(4) (2015).
- Kotton CN, Kumar D, Caliendo AM, Huprikar S, Chou S, et al. The Transplantation Society International CMV Consensus Group. The Third International Consensus Guidelines on the Management of Cytomegalovirus in Solid-organ Transplantation. Transplantation 102 (6) (2018): 900-931.
- Cetinkaya B, Unek G, Kipmen-Korgun D, Koksoy S, Korgun ET. Effects of Human Placental Amnion Derived Mesenchymal Stem Cells on Proliferation and Apoptosis Mechanisms in Chronic Kidney Disease in the Rat. Int J Stem Cells 12 (1) (2019):151-161.
- Chen J, Ceholski DK, Turnbull IC, Liang L, Hajjar RJ. Ischemic Model of Heart Failure in Rats and Mice. Methods Mol Biol 1816 (2018):175-182.
- Moon C, Krawczyk M, Ahn D, Ahmet I, Paik D, et al. Erythropoietin reduces myocardial infarction and left ventricular functional decline after coronary artery ligation in rats. Proc Natl Acad Sci USA 100 (20) (2003): 11612-11617.
- Jenca D, Melenovsky V, Stehlik J, Stanek V, Kettner J, et al. Heart failure after myocardial infarction: incidence and predictors. ESC Heart Fail 8 (1) (2021): 222-237.
- Meller D, Pauklin M, Thomasen H, Westekemper H, Steuhl KP. Amniotic membrane transplantation in the human eye. Dtsch Arztebl Int 108 (14) (2011): 243-248.
- Salehi SH, As'adi K, Mousavi SJ, Shoar S. Evaluation of Amniotic Membrane Effectiveness in Skin Graft Donor Site Dressing in Burn Patients. Indian J Surg 77 (2015): 427-431.
- Qiang Y, Liang G, Yu L. Human amniotic mesenchymal stem cells alleviate lung injury induced by ischemia and reperfusion after cardiopulmonary bypass in dogs. Lab Invest 96 (5) (2016):537-546
- Cui P, Xin H, Yao Y, Xiao S, Zhu F, et al. Human amnion-derived mesenchymal stem cells alleviate lung injury induced by white smoke inhalation in rats. Stem Cell Res Ther 9(1) (2018): 101.
- Hua D, Ju Z, Gan X, Wang Q, Luo C, et al. Human amniotic mesenchymal stromal cells alleviate acute liver injury by inhibiting the pro-inflammatory response of liver resident macrophage through autophagy. Ann Transl Med 7(16) (2019): 392.
- Yin L, Zhou ZX, Shen M, Chen N, Jiang F, et al. The Human Amniotic Mesenchymal Stem Cells (hAMSCs) Improve the Implant Osseointegration and Bone Regeneration in Maxillary Sinus Floor Elevation in Rabbits. Stem Cells Int 2019: 9845497.
- Martin-Rendon E, Brunskill SJ, Hyde CJ, Stanworth SJ, Mathur A, Watt SM. Autologous bone marrow stem cells to treat acute myocardial infarction: a systematic review. European Heart Journal 29 (15) (2008): 1807-1818.
- Narita T, Suzuki K. Bone marrow-derived mesenchymal stem cells for the treatment of heart failure. Heart Fail Rev 20 (1) (2015): 53-68.
- Kim SH, Cho JH, Lee YH, Lee JH, Kim SS, et al. Improvement in Left Ventricular Function with Intracoronary Mesenchymal Stem Cell Therapy in a Patient with Anterior Wall ST-Segment Elan. 32(4)(2018): 329-338.
