A Clinical Benefit of Training Asthmatic Patients on How to Use Metered Dose Inhalers by using the 2Tone Trainer In Libya
Article Information
Walid Y Tarsin1, Nabila A Hshad1, Ishraq Elshamli2, Fathi M Sherif1*
1Department of Pharmacology and Clinical Pharmacy, University of Tripoli, Tripoli, Libya
2Department of Respiratory, Tripoli Medical Centre, Tripoli, Libya
*Corresponding Author: Fathi Mohamed Sherif, Department of Pharmacology and Clinical Pharmacy, University of Tripoli, Tripoli, Libya
Received: 20 March 2019; Accepted: 18 April 2019; Published: 24 April 2019
Citation: Walid Y Tarsin, Nabila A Hshad, Ishraq Elshamli, Fathi M Sherif. A Clinical Benefit of Training Asthmatic Patients on How to Use Metered Dose Inhalers by using the 2Tone Trainer In Libya. J Pharm Pharmacol Res 3 (2019): 028-040.
Share at FacebookAbstract
Bronchial asthma is a serious chronic inflammatory disease of the respiratory system. Aerosol inhalation as a route of drug delivery has become well-known in therapy of asthma. This study was designed to evaluate if the use of 2Tone helps patients maintain the correct inhalation technique after training and can improve the clinical benefit. 125 Libyan adult asthmatic patients were recruited. At the first clinical visit; 38, 44 and 43 patients were included as C, VT and 2T groups, respectively. Their inhalation flow rate through an MDI was measured using an In-Check Dial. Patients in 2T group were trained on how to use the 2Tone Trainer according to its PIL and practiced inhaling through this training aid to familiarize themselves with the different sounds according to the inhalation rates. At the second clinic visit for all the patients was held six weeks later, each patient was assessed in the same manner as on the first visit. Results show no significant difference in the change of FEV1 and PEFR between all the groups. Lung function measurements, percent predicted FEV1 and PEFR showed significant low correlation with all AQLQ domains, except for AQLQ environment which found no correlation with lung function measurement. Significant correlations between percent predicted FEV1 and PEFR with all AQLQ domains. Patients in the 2T group showed reduced IFR of about double that in VT group, whereas in the C group, there was no difference in IFR. Comparison of IFR between VT vs. 2T groups at visit one showed no statistical significant difference. However, at visit two, comparison between all the groups showed a highly significant difference. Thus, this study shows that 100% and 29% of the patients in the C and VT groups were inhaling at a high IFR while the 2T group shows only one patient (3%) was inhaling at the high flow rate while the rest of the patients mana
Keywords
2Tone trainer; Asthma; COPD; Libya; Lung drug delivery; MDI
Article Details
1. Introduction
Aerosol inhalation as a method of drug delivery to the respiratory tract has become well established in treatment of lung diseases such as asthma and chronic obstructive pulmonary disease (COPD). This route has several distinct advantages. Medication is delivered directly to the site of action allowing a rapid and predictable onset of action, avoiding the first-pass metabolism and degradation within the gastrointestinal tract. It is also lowing dosages than those by other routes which, therefore, minimises the unwanted side effects [1]. However, the efficiency of lung deposition from inhalation therapy is not high and about 10% of the inhaled dose reaches the lungs [2]. On the other hand, it is possible to increase the fraction of dose deposited in the lungs by training the patients the correct inhalation techniques [3], but, other study has, however, shown that patients forget their trained technique within one month [4]. Several important factors affecting drug therapy given by inhalation, including flow rate, period of breath holding, the volume of air inhaled and the volume of lung at the initiation of inhalation and the position of inhaler in relation to the mouth [5]. Inefficient inhaler technique is a common patient problem resulting in poor drug delivery, decreased disease control and increased inhaler use [6]. To ensure effective and consistent management of asthma. It is, therefore, essential that patients are compliant and that they can use the inhaled medication according to the instructions provided by the manufacturers. Achievement of these will ensure that the same dose is emitted with each inhalation. It may, thus, be necessary to train the patient with the required inhalation technique [2].
Metered dose inhalers (MDI) technique assessment have heavily focused on coordinating the dose release with the start of an inhalation. However, some studies have shown that co-ordination of dose actuation and inhalation is not important as long as the patient is inhaling when a dose is released [7, 8]. It has emphasized the importance of a slow inhalation flow rate (IFR) when using an MDI for optimal therapeutic effect. IFR is the most common mistake made by patients when using an MDI. Broeders and others [9] found that 80% of patients were inhaling too fast (>90 L/min) when using their MDI compared to 40% of patients with coordination problems. A slow and deep IFR when using an MDI was the highest mistake made by patients [10]. Pauwels and his collaborators [11] showed that the inhalation rate through MDIs should be <90 L/min. Therefore, patients prescribed MDIs should use a slow IFR but, this rate is difficult to define. A training aid; the 2Tone Trainer (Canday Medical Ltd) has been introduced to help patients obtain the most desirable inhalation rate when using an MDI. The patient information leaflet (PIF) provided with 2Tone encourages patients to practice using the device in the same way that they would use their MDI. During this use, the training device provides them with audible feedback according to the inhalation rate they have used. It makes a two-tone sound when inhaling faster than 60 L/min, one tone between 30-60 L/min and no sound at <30 L/min. Patients are advised to obtain the one-tone sound and thus become customized with the degree of inspiratory effort they need to use to achieve this rate through an MDI. Continued use of the 2Tone Trainer, at home, after a training session may be a solution to the problem of using a slow inhalation rate and repeated inhalation technique training. Thus, this study is designed to evaluate if the use of 2Tone helps patients maintain the correct inhalation flow rate after training the asthmatic patients.
2. Methods and Subjects
2.1 Patients
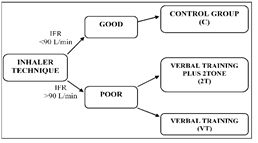
This study was designed to be parallel clinical study to assess the effect of inhaler technique on the clinical benefit in patients with asthma. Study patients were obtained from the Tripoli Medical Center (TMC), respiratory department, outpatient unit (2016). This study was of two groups; intervention and control. Allocation of patients to either control or intervention groups was according to their inhaler technique regarding their IFR, measured by using an In-Check dial™. A schematic design of the study is given in Figure 1. Approval for the study was obtained from the local ethical committee of the center to carry out this study (TMC, 8/2016). All patients, giving signed informed consent were asked to agree and sign. According to a previous study [12], the mean (SD) inhalation rate of 398 outpatients at Leeds Teaching Hospital, NHS Trust, when they inhaled through an MDI was 107(20) L/min. Using the table provided by Altman [13] to reduce the inhalation flow rate by 20% with 90% power at the 5% significance level, would require 30 subjects. Therefore, about 40 asthmatic patients were recruited to be included in each group to allow for any dropout.
The inclusion criteria were; asthma patients using at least one MDI without a spacer device, prescribed a preventer MDI (with or without a spacer) and signed written consent. While the patient exclusion criteria were; patients experiencing an acute exacerbation of asthma or receiving oral prednisolone in the four weeks prior to the study, patients with other diseases adversely affecting the respiratory system or with evidence of fixed respiratory obstruction, deaf or unable to distinguish between one and two tones with the 2Tone trainer and COPD.
2.2 Methods
Inhalation flow rate of the patients through MDI was measured using In-Check Dial (Clement Clarke International, UK). Patients were classified to poor and good inhalation technique according to IFR. Those with good IFR of <90L/min were considered a control group (C), whilst those with a poor IFR of >90L/min were intervention group. Patients in the intervention group were randomly allocated into the verbal training group (VT) or the verbal training plus 2Tone group (2T). C group was not told what the correct flow was nor directed on how to use their inhalers. VT patients were trained on the most desirable IFR. Patients in the 2T group received the same verbal training as the VT group. Patients in 2T group were also trained how to use the 2Tone Trainer according to its PIL and practiced inhaling through this training aid to familiarize themselves with the different sounds according to the inhalation rates. All inhaled consistently making one sound (equivalent to 30-60 L/min) before leaving the outpatient unit. Patients were advised to use the training device every morning and night to obtain one tone note and to use the same inhalation procedure when they used their active MDI. All patients in VT and 2Tone groups had their IFR through In-Check Dial measured at the end of the training session to ensure that they were inhaling at a rate <90L/min through MDI.
The second visit for all patients was held six weeks later and each patient was assessed in the same manner as on the first visit and data collection repeated. Patients were asked to demonstrate their inhaler technique with a placebo pMDI device. The inhalation technique was marked for 1 to 13 steps, according to the most desirable inhalation technique, and a score out of 13 was given for each patient. The same person carried out the tests on each occasion to provide consistency in the measurements.
3. Statistical Analysis
Data were collected from groups of patients to allow a baseline to be established with which to compare subsequent data. A statistical analysis was carried out using an SPSS version 18 database package by MINITAB, USA. For parametric data, one-way ANOVA followed by paired t test to compare responses of pre and post counselling in each group (visit one vs. two) for inhalation flow rates and peak flow measurements. Independent t test was used for responses between the groups (2Tone vs. VT, 2Tone vs. C and VT vs. C). For nonparametric data, comparison among groups was made by Wilcoxon test followed by Mann-Whitney U test between individual groups. *P <0.05 was considered significant, **P <0.01 highly significant and ***P <0.001 very highly significant.
4. Results
In this study, at first visit; 38, 44 and 43 patients were considered for three different groups (C, VT and 2T, respectively) but at the second visit, there were 36, 35, and 36 patients in the C, VT and 2T groups attended. The mean (SD) age of patients was 57.16 (15.1) years. The youngest patient was 22 and the oldest patient was 87 years old. The normality test revealed that IFR data was not normally distributed. However, data from patients AQLQ and lung function tests (FEV1 and PEFR) showed normal distribution.
4.1 Inhalation Flow Rate (IFR)
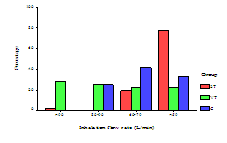
The results showed that 71 patients (66.4%) inhaled at a rate greater than 90 L/min and thus formed the intervention group. 11 patients (15.5%) in the intervention group (VT and 2T) returned for the follow-up (visit 2) at flow rate greater than 90 L/min after counselling (Table 1). 10 patients (28.6%) were from the VT group and one patient (2.8%) from 2T group (Figure 2). All patients in C group at visits one and two were inhaling at a flow rate <90 L/min with mean IFR of 66 L/min. Patients mean IFR in VT and 2T groups were less than 90 L/min at visit two (Figure 3). Mean (SD) of the patients IFR for all groups at visits one and two are given in Table 2.
|
Groups |
IFR (L/min) |
Visit one n (%) |
Visit two n (%) |
|
Control |
|||
|
C (n=36) |
>90 |
0 |
0 |
|
80-90 |
14 (38.9) |
9 (25) |
|
|
60-79 |
10 (27.8) |
15 (41.7) |
|
|
<59 |
12 (33.3) |
12 (33.3) |
|
|
Intervention |
|||
|
VT (n=35) |
>90 |
35 (100) |
10 (28.6) |
|
80-90 |
0 |
9 (25.7) |
|
|
60-79 |
0 |
8 (22.9) |
|
|
<59 |
0 |
8 (22.9) |
|
|
2T (n=36) |
>90 |
36 (100) |
1 (2.8) |
|
60-79 |
0 |
7 (19.4) |
|
|
<59 |
0 |
28 (77.8) |
|
Table 1: Inhalation flow rate (L/min) at visits one and two for all groups.
|
Parameters |
Control group |
Verbal group |
2Tone group |
|
IFR (1) |
66 (16) |
118 (5) |
117 (5) |
|
IFR (2) |
66 (14) |
81 (23) |
52 (13) |
|
Δ IFR |
0.28 (11.95) |
- 37.11 (24.24) |
- 64.64 (14.74) |
Table 2: Mean (SD) for patients IFR between visits one and two for all groups.
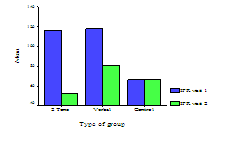
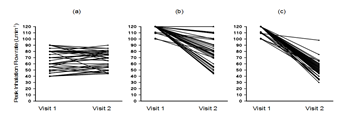
Comparisons of the IFR for patients in each group at visits one and two are shown in Figure 4. Patients in 2T group showed reduced IFR of about double that in VT group whereas in C group, there was no difference in IFR (Table 2). Comparison using Wilcoxon test of IFR using the median difference (95% confidence interval) between visits one and two in C group revealed no statistical significant difference. In contrast, VT and 2T groups showed highly significant difference (p <0.001) in IFR between visits one and two (Table 3). Furthermore, comparison of IFR between the individual groups by Mann-Whitney test, at visit one showed a highly significant difference (p <0.001) between 2T and VT groups against C group. However, comparison of IFR between VT vs. 2T groups at visit one showed no statistical significant difference. However, at visit two, comparison between all the groups showed highly significant difference (p <0.001) in IFR (Table 4).
|
Parameter |
Control group |
Verbal group |
2T group |
|
IFR |
0 (- 5, 2.5)*** |
37.5 (27.5, 46)*** |
65 (62.5, 70)*** |
***Statistically significant by P <0.001
Table 3: Median difference (95% confidence interval) of IFR between visits one and two for each group.
|
Groups |
2T against VT |
VT against C |
2T against C |
|
IFR (1) |
0.0 |
50 (60, 40)*** |
50 (60, 40)*** |
|
IFR (2) |
26.5 (20, 38)*** |
13 (25, 5)** |
15 (10, 20)*** |
|
Δ IFR |
28 (40, 18)*** |
40 (49.99, 25)*** |
65 (70, 60)*** |
Statistically significant by ** P <0.01 and *** P <0.001; Δ-denotes the overall change between visit one and two
Table 4: Median difference (95% confidence interval) for IFR at visits one and two together with the change between visits one and two (Δ IFR) between all groups.
4.2 Inhaler technique
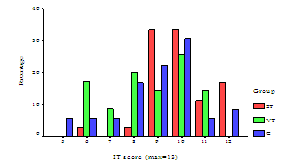
All 107 patients had their inhaler technique checked at visit two using placebo MDI. Inhalation technique was assessed according to the most desirable inhaler technique. A summary of patient’s inhalation technique is given in Table 5. The highest incorrect steps for C group were (step 11) not holding the breath for 10 seconds and (step 9) the breathing in not taking 5 seconds. For VT group, (step 12) the breathing out slowly and (step 9) breathing in takes 5 seconds were the most incorrect steps. 2T group showed almost equal percentage of incorrect steps of (step 7) coordination problems and (step 12) breathing out slowly. For all groups, none of the patients achieved of total number of scores which was 13. Figure 5 summaries the percentage the total number of inhaler technique score achieved by patients in each group. Using Mann-Whitney U-test to compare total inhaler technique score between the groups revealed significant difference between 2T group with VT and C groups (p <0.01and p <0.05, respectively).
|
Inhalation technique n (%) |
2 Tone n=36 |
Verbal n=35 |
Control n=36 |
|
(1) Remove cap (IT1) |
0 |
0 |
0 |
|
(2) Shake inhaler (IT2) |
11 (30.6) |
9 (25.7) |
15 (41.7) |
|
(3) Breathe out slowly (IT3) |
6 (16.7) |
9 (25.7) |
7 (19.4) |
|
(4) As far as comfortable to empty lungs (IT4) |
17 (47.2) |
14 (40) |
19 (52.8) |
|
(5) Place mouthpiece of inhaler between your lips (IT5) |
0 |
0 |
0 |
|
(6) Close your lips around mouthpiece, creating a seal (IT6) |
1 (2.8) |
3 (8.6) |
2 (5.6) |
|
(7) Start to breathe in slowly and press aerosol canister (IT7) * |
21 (58.3) |
19 (54.3) |
21 (58.3) |
|
(8) Breathe in slowly (IT8) |
1 (2.8) |
10 (28.6) |
0 |
|
(9) Breathe in should take 5 seconds (IT9) |
7 (19.4) |
25 (71.4) |
26 (72.2) |
|
(10) Remove inhaler and keep your mouth close (IT10) |
6 (16.7) |
6 (17.1) |
3 (8.3) |
|
(11) Hold breath for 10 seconds (IT11) |
17 (47.2) |
24 (68.6) |
28 (77.8) |
|
(12) Breathe out slowly (IT12) |
20 (55.6) |
28 (80) |
20 (55.6) |
|
(13) Repeat steps 1 to 12 after 30 seconds, if necessary (IT13) |
3 (8.3) |
5 (14.3) |
3 (8.3) |
Table 5: Patients incorrect steps for inhalation technique.
4.3 Peak Expiratory Flow Rate (PEFR) and Forced Expiratory Volume in one second (FEV1)
The mean (SD) percent predicted FEV1 and PEFR together with the actual values for visits one and two and changes between visits one and two for all groups are given in Table 6. Patients in both groups were classified according to their percent predicted FEV1 and PEFR in Table 7. Thus, comparison of percent predicted of FEV1 and PEFR with the actual values measured between visit one and visit two for the three groups using paired t test is shown in Table 8. Thus, it shows that there was no difference between visit one and visit two for C, 2T and VT groups. Comparison of actual values FEV1 and PEFR measured at visit one, two and change of visits one and two between the three groups using one-way ANOVA with Bonferroni correction is given in Table 9. A comparison of percent predicted FEV1 and PEFR at visit one, two and change of visits one and two between groups is shown in Table 10. No significant difference between 2T and VT in percent predicted and actual value of FEV1 and PEFR between 2T and VT groups at visits one and two was found. However, VT group shows a significant difference (p <0.01) with C group in percent predicted and actual value of FEV1 at visits one and two. In addition, VT shows significant difference (p <0.01) in actual value and percent predicted of PEFR with C group. 2T group shows significant difference (p <0.05 and p <0.01) in only PEFR actual value and percent predicted with C group, respectively. Furthermore, comparison was made for the change of visits one and two for actual and percent predicted between all the groups (Table 11). It shows no significant difference in the change of FEV1 and PEFR between all the groups. Lung function measurements, percent predicted FEV1 and PEFR showed significant low correlation with all AQLQ domains (p <0.05) except for the AQLQ environment which found no correlation with lung function measurement. Table 12 describes the correlations between percent predicted FEV1 and PEFR with all AQLQ domains.
|
Visit one |
Control |
Verbal |
2Tone |
|
FEV1 (L) |
1.6 (0.82) |
2.1 (0.75) |
1.95 (0.76) |
|
FEV1 (% pre.) |
62.2 (25.4) |
76.9 (24.1) |
71.7 (22) |
|
PEFR (L.min-1) |
251.5 (107.1) |
321.8 (98.7) |
312.9 (101.6) |
|
PEFR (% pre.) |
65.2 (26.1) |
77.3 (22) |
75.1 (23.6) |
|
Visit two |
|||
|
FEV1 (L) |
1.57 (0.73) |
2.16 (0.74) |
1.93 (0.63) |
|
FEV1 (% pre.) |
61.3 (22.2) |
78.3 (22.3) |
71.4 (16.9) |
|
PEFR (L.min-1) |
244.5 (93.4) |
320.9 (88.3) |
321.8 (86.8) |
|
PEFR (% pre.) |
62 (21.9) |
77.8 (19.8) |
76.2 (19.9) |
|
Δ FEV1 (L) |
-0.3 (0.27) |
0.06 (0.33) |
-0.01 (0.29) |
|
Δ FEV1 (% pre.) |
-0.9 (10.3) |
1.4 (9.4) |
-0.4 (10.6) |
|
Δ PEFR (L.min-1) |
-7 (40) |
-1 (62) |
9 (44) |
|
Δ PEFR (% pre.) |
-3.2 (11.8) |
0.5 (14.3) |
1.1 (13.3) |
Table 6: Mean (SD) of patient’s lung function measurements for two visits.
|
Groups |
Visit |
Severe n (%) |
Moderate n (%) |
Mild n (%) |
Total |
|
2Tone |
one |
11 (30.6) [8 (22.2)] |
14 (38.9) [15 (41.7)] |
11 (30.6) [13 (36.1)] |
36 (100) |
|
two |
9 (25) [5 (13.9)] |
17 (47.2) [19 (52.8)] |
10 (27.8) [12 (33.3)] |
||
|
Verbal |
one |
10 (28.6) [9 (25.7)] |
6 (17.1) [9 (25.7)] |
19 (54.3) [17 (48.6)] |
35 (100) |
|
two |
8 (22.9) [6 (17.1)] |
11 (31.4) [12 (34.3)] |
16 (45.7) [17 (48.6)] |
||
|
Control |
one |
20 (55.6) [14 (38.9)] |
8 (22.2) [12 (33.3)] |
8 (22.2) [10 (27.8)] |
36 (100) |
|
two |
19 (52.8) [14 (38.9)] |
8 (22.2) [14 (38.9)] |
9 (25) [8 (22.2)] |
Table 7: Morbidity associated with percent predicted FEV1 [PEFR] at visits one and two (all groups).
|
Lung function test |
Control |
Verbal |
2Tone |
|
FEV1 (L) |
0.03 (-0.06, 0.12) |
-0.06 (-0.18, 0.05) |
0.015 (-0.08, 0.11) |
|
FEV1 (% pre.) |
0.9 (-2.58, 4.37) |
-1.43 (-4.67, 1.8) |
0.35 (-3.24, 3.95) |
|
PEFR (L.min-1) |
7.03 (-6.6, 20.6) |
0.91 (-20.2, 22.1) |
-8.89 (-23.8, 6.1) |
|
PEFR (% pre.) |
3.2 (-0.78, 7.19) |
-0.51 (-5.41, 4.4) |
-1.08 (-5.59, 3.43) |
Table 8: Mean difference (95% confidence interval) of % predicted FEV1 and PEFR with the actual values for all groups between visits one and two.
|
Lung function measurement (% pre.) |
2T against VT |
VT against C |
2T against C |
|
FEV1 (1) |
-5.18 (-18.97, 8.62) |
14.75 (0.95, 28.54)* |
9.57 (-4.12, 23.27) |
|
FEV1 (2) |
-6.96 (-18.86, 4.94) |
17.08 (5.17, 28.98)*** |
10.12 (-1.7, 21.93) |
|
PEFR (1) |
-2.26 (-16.11, 11.6) |
12.14 (-1.71, 26) |
9.88 (-3.87, 23.64) |
|
PEFR (2) |
-1.68 (-13.55, 10.19) |
15.85 (3.98, 27.72)** |
14.17 (2.39, 25.95)** |
* P <0.05, ** P <0.01 and ***P <0.001.
Table 9: Mean difference (95% confidence interval) of percent predicted FEV1 and PEFR between groups.
|
Lung function measurement |
2T against VT |
VT against C |
2T against C |
|
FEV1 (1) (L/min) |
-0.15 (-0.6, 0.3) |
0.49 (0.04, 0.94)* |
0.34 (-0.1, 0.79) |
|
FEV1 (2) (L) |
-0.23 (-0.63, 0.18) |
0.59 (0.18, 0.99)** |
0.36 (-0.04, 0.76) |
|
PEFR (1) (L/min) |
-8.94 (-68.2, 50.3) |
70.3 (11.03, 129.5)* |
61.3 (2.51, 120.2)* |
|
PEFR (2) (L) |
0.86 (-50.86, 52.59) |
76.4 (24.67, 128.1)** |
77.25 (25.9, 128.6)** |
Statistically significant by * P <0.05 and ** P <0.01.
Table 10: Mean difference (95% confidence interval) of actual values of FEV1 and PEFR between groups.
|
Mean difference |
2T against VT |
VT against C |
2T against C |
|
Δ FEV1 (L) |
-0.08 (-0.25, 0.095) |
0.1 (-0.08, 0.27) |
0.02 (-0.15, 0.19) |
|
Δ PEFR (L/min) |
9.8 (-18.7, 38.3) |
6.11 (-22.4, 34.6) |
15.9 (-12.4, 44.24) |
|
Δ FEV1 (% pre.) |
-1.79 (-7.63, 4.06) |
2.33 (-3.52, 8.18) |
0.54 (-5.26, 6.35) |
|
Δ PEFR (% pre.) |
0.58 (-7.03, 8.18) |
3.71 (-3.89, 11.31) |
4.29 (-3.26, 11.83) |
Table 11: Mean difference (95% confidence interval) for actual values and percent predicted of Δ FEV1 and Δ PEFR between groups.
|
Lung function measurement (% predicted) |
AQLQ total |
AQLQ symptom |
AQLQ emotion |
AQLQ environment |
AQLQ activity |
|
FEV1 (L) |
+0.23* |
+0.2* |
+0.26* |
+0.09 |
+0.24* |
|
PEFR (L/min) |
+0.19* |
+0.24* |
+0.21* |
+0.01 |
+0.23* |
|
JMI |
-0.51** |
-0.6** |
-0.29* |
-0.32* |
-0.38*** |
Statistically significant by *P <0.05 and *** P <0.001
Table 12: Correlations between percent predicted FEV1 and PEFR with AQLQ domains.
5. Discussion
The inhaled route of administration is the optimal method of drug delivery for the treatment of patients with obstructive airway diseases and the pMDI is presently the most commonly used inhaler [11]. Despite the availability of efficacious therapies, asthma control is often poor [14, 15] and the improper use of inhaler devices is one of the major cause of poor disease control [16, 17, 18]. It has been suggested that approximately 50% of patients do not obtain sufficient benefit from their inhalers because of poor inhaler technique [19]. A correct inhaler technique by the patient is crucial for the success of the therapy [20, 21, 22]. Thus, the benefits of inhaled therapy are accompanied by the drawbacks, particularly the challenges patients' face when using their inhaler devices. It has been acknowledged that the most commonly encountered pMDI technique problems by patients are; poor coordination of inhalation with inhaler actuation, stopping to inhale shortly after activating pMDI and inspiration through the nose [23]. The main findings of this study is that nearly 75% of the patients in the control group made at least one technique error or completely misused their inhaler.
Several studies emphasized the importance of slow inhalation flow rate (IFR) when using MDI for optimal therapeutic effect. Poor inhaler technique is linked to poor disease control [24] and increased hospitalisation [25]. Data have shown that IFR is the most common mistake made by patients when using MDI. Broeders et al. [9] found that 80% of patients were inhaling too fast (>90 L/min) and Al-Showair et al. [26] found that only 8% of patients use slow inhalation with good co-ordination. The present results indicate that all the patients in the control group were using a high IFR when they used their MDIs.
Frequent inhaler training technique and proper inhaler handling are recommended, particularly by asthma management guidelines and that inhaler technique should be checked regularly during follow-up [8, 9]. However, the provision of inhaler technique training remains irregular. The majority of the previous studies suggested that a large proportion of patients do have problems using their inhalers but they are subjective assessments. The design of this study has therefore been designed to provide objective assessments by the use of a 2Tone device to help the patient to maintain the optimum IFR needed for the MDIs. The results of this study show that 100% and 29% of the patients in the control and verbal counselling groups, respectively, were inhaling at a high IFR while the 2Tone group shows that only one patient (3%) was inhaling at the high flow rate while the rest of the patients managed to obtain the optimum IFR needed for the MDIs and this indicates the importance of the use of the 2Tone device to train the patients to slow their IFR. In this study also no effect of good MDI technique on the improvement of PEFR (Peak Expiratory Flow Rate) with no significant differences were found between first and second visits.
References
- Newman SP, Clarke SW. Therapeutic aerosols I-physical and practical considerations. Thorax 38 (1983): 881-886.
- Newman SP, Moren F, Pavia D, Sheahan NF, Clarke SW. Deposition of pressurised aerosols in the human respiratory tract. Thorax 36 (1981): 52-55.
- Power GM, Dash CH. A new modified form of inhaler (Rotahaler) for Patients with chronic obstructive lung diseases. Pharmacotherapeutica 4 (1985): 98-100.
- Crompton GK. Problems patients have using pressurised aerosol inhalers. European Journal of Respiratory Disesases 63 (1982): 101-104.
- Timsina MP, Martin GP, Marriott C, Ganderton D, Yianneskis M. Drug delivery to the respiratory tract using dry powder inhalers. International Journal of Pharmacology 101 (1994): 1-13.
- Lenney J, Innes JA, Crompton GK. Inappropriate inhaler use: assessment of use and patient preference of seven inhalation devices. EDICI. Respiratory Medicine 94 (2000): 496-500.
- Dolovich M, Ruffin RE, Roberts R, Newhouse MT. Optimal delivery of aerosols from metered dose inhalers. Chest 80 (1981): 911-915.
- Newman SP, Pavia D, Clarke SW. Simple instructions for using pressurised aerosol bronchodilator. Journal of Royal Society of Medince 73 (1980): 776-779.
- Broeders ME, Molema J, Hop WC, Folgering HT. Inhalation profiles in asthmatics and COPD patients: reproducibility and effect of instruction. Journal of Aerosol Medicine16 (2003): 131-141.
- Hesselink AE, Penninx BW, Wijnhoven HA, Kriegsman DM, Van Eijk JT. Determinants of an incorrect inhalation technique in patients with asthma or COPD. Scandanvian Journal of Primary Health Care 19 (2001): 255-260.
- Pauwels R. Inhalation device, pulmonary deposition and clinical effects of inhaled therapy. Journal of Aerosol Medicine 10 (1997): 17-21.
- Chrystyn H, Brownlee K, Chetcuti P, Corrado OJ, Kanthapillai P, Person S, et al. Inhalation rates of asthmatic children (child) and adults and of chronic obstructive pulmonary disease (COPD) patients through inhalers. American Journal of Respiratory Critical Care Medicine 105 (2002): 190.
- Altman P, Wehbe L, Dederichs J, Guerin T, Ament B, Moronta MC, et al. Comparison of peak inspiratory flow rate via the Breezhaler®, Ellipta® and HandiHaler® dry powder inhalers in patients with moderate to very severe COPD: a randomized cross-over trial. BMC Pulmonary Medicine 18 (2018): 100.
- Chapman KR, Boulet LP, Rea RM, Franssen E. Suboptimal asthma control: prevalence, detection and consequences in general practice. European Respiratory Journal 31 (2008): 320-325.
- Giraud V, Allaert FA, Roche N. Inhaler technique and asthma: feasability and acceptability of training by pharmacists. Respiratory Medicine 105 (2011): 1815-1822.
- Crompton GK, Barnes PJ, Broeders M, Corrigan C, Corbetta L, Dekhuijzen R, et. The need to improve inhalation technique in Europe: a report from the aerosol drug management improvement team. Respiratory Medicine 100 (2006): 1479-1494.
- Molimard M, Le Gros V. Impact of patient-related factors on asthma control. Journal of Asthma 45 (2008): 109-113.
- Virchow JC, Crompton GK, Dal Negro R, Pedersen S, Magnan A, Seidenberg J, et al. Importance of inhaler devices in the management of airway disease. Respiratory Medicine 102 (2008): 10-19.
- Crompton G, Duncan J. Clinical assessment of a new breath-actuated inhaler. Practitioner 233 (1989): 268-269.
- Horsley MG, Bailie GR. Risk factors for inadequate use of pressurized aerosol inhalers. Journal of Clinical Pharmacology and Therapeutics 13 (1988): 139-143.
- Larsen JS, Hahn M, Ekholm B, Wick KA. Evaluation of conventional press-and-breathe metered-dose inhaler technique in 501 patients. Journal of Asthma 31 (1994): 193-199.
- Virchow JC, Crompton GK, Dal Negro R, Pedersen S, Magnan A, Seidenberg J, et al. Importance of inhaler devices in the management of airway disease. Respiratory Medicine 102 (2008): 10-19.
- Giraud V, Roche N. Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability. European Respiratory Journal 19 (2002): 246-251.
- Crompton GK., Problems patients have using pressurized aerosol inhalers. European Journal of Respiratory Diseases 119 (1982): 101-104.
- Melani AS, Bonavia M, Cilenti V, Cinti C, Lodi M, Martucci P, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respiratory Medicine 105 (2011): 930-938.
- Al-showair RAM, Pearson SB, Chrystyn H. The potential of a 2Tone trainer to help patients use their metered-dose inhalers. Chest 131 (2007): 1776-1782.