Ventricular Hyper-Excitability Revealing Cardiac and Mediastino-Pulmonary Sarcoidosis
Article Information
Houda Mokhlis1*, Houda Bachri1, Rime lemouaden2, Mohamed Malki1, Sara Ahchouch1, Meryem Boumaaz1, Achraf Zaimi1, Nadia Loudiyi1, Najat Mouine1, Ilyasse Asfalou1, Aatif Benyass1
1Department of Cardiology, Mohammed V Military Hospital, Mohammed V University, Rabat, Morocco
2Department of internal medicine, Mohammed V Military Hospital, Mohammed V University, Rabat, Morocco
*Corresponding author: Houda Mokhlis, Department of Cardiology, Mohammed V Military Hospital, Mohammed V University, Rabat, Morocco.
Received: 27 October 2022; Accepted: 12 November 2022; Published: 15 November 2022
Citation: Houda Mokhlis, Houda Bachri, Rime lemouaden, Mohamed Malki, Sara Ahchouch, Meryem Boumaaz, Achraf Zaimi, Nadia Loudiyi, Najat Mouine, Ilyasse Asfalou, Aatif Benyass. Ventricular Hyper-Excitability Revealing Cardiac and Mediastino- Pulmonary Sarcoidosis. Cardiology and Cardiovascular Medicine 6 (2022): 512-514.
Share at FacebookKeywords
Cardiac Sarcoidosis; Case Report; Ventricular Hyperexcitability
Article Details
1. Introduction
Sarcoidosis, also known as Besnier-Boeck-Schaumann disease, first described in 1877 [1], is a systemic granulomatous disease that affects multiple organs (nervous system, heart, liver and kidneys.. ....), but mainly affects the lungs and lymph glands. Sarcoidosis has no known cause, and it can affect a wide range of people and present a real diagnostic challenge. Cardiac sarcoidosis can manifest as complete heart block, ventricular arrhythmias, congestive heart failure, pulmonary hypertension, and ventricular aneurysms.
2. Case Report
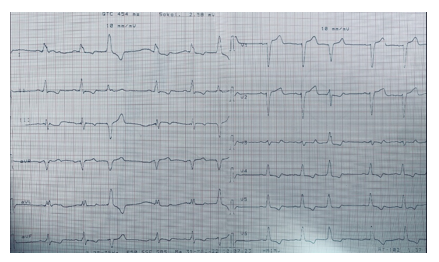
We report the case of a 65-year-old female patient; mother of seven children with no prior medical conditions, that was admitted to the cardiology department for paroxysmal palpitations and stage II NYHA Dyspnea. she also reported dysphonia evolving over several months. Physical examination was unremarkable except for an irregular pulse. EKG ( figure 1) showed sinus rhythm at 60 beats per minute, complete left bundle branch block, and multiple premature ventricular extrasystoles and a 24 h Holter ECG monitor was in favour of a ventricular hyperexcitability. Chest X-ray showed opacities with bilateral hilar projection and Echocardiography revealed segmental kinetic disorders with a reduced LVEF. A coronary angiography was performed showing non-significant lesions dismissing a coronary origin of the systolic left ventricular dysfunction.
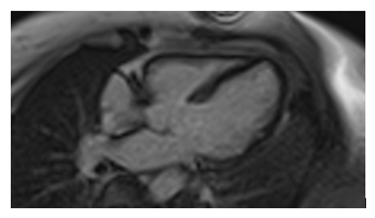
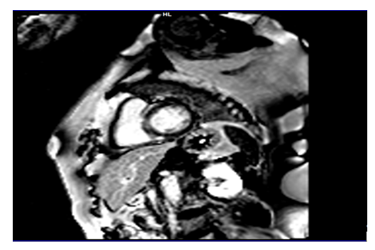
A Chest CT was performed showing the presence of several (sub- and supra-diaphragmatic) granulomatous calcified lymphadenopathies and an appearance suggestive of left vocal cord paralysis. Initial blood tests did not show abnormalities, in particular the hepatic, renal, urinary and blood phosphocalcic balance; Conversion enzyme levels were also normal. In the framework of the etiological assessment a TEP scan was performed showing hypermetabolic pathological focus on the left supraclavicular, hilar-pulmonary and abdominal areas as well as a hypermetabolic focus on the left posterolateral wall of the nasopharynx with no pathological focus on the heart. The supra clavicular lymph node micro- biopsy showed epithelioid and gigantocellular granulomatous inflammation without caseous necrosis in favor of a sarcoidosis. Because of the strong clinical suspicion of Cardiac sarcoidosis we performed an MRI that revealed -in addition to kinetic disorders a delayed enhancement of the septum confirming our doubts ( figure 2-3) .
3. Discussion
Cardiac sarcoidosis (CS)that is clinically manifest happens in only 5% of patients. Whereas asymptomatic CS is much more prevalent and estimated at 25% in patients with pulmonary or systemic sarcoidosis as shown by autopsy studies [2,3]. These findings were later found to be consistent with the results of more recent technology, namely the late gadolinium enhanced (LGE) cardiovascular resonance (CMR) technology [4]. Cardiac sarcoidosis (CS) is an important prognosis factor Considering the increased risk of sudden death .The myocardial process is comprised of granuloma related inflammation, first reversible but then progresses to irreversible fibrosis. Thus, early and accurate diagnosis of CS is capital to ensure an early and adequate treatment strategy [5]. The main manifestations of CS include conduction abnormalities, ventricular arrhythmias, and heart failure. These manifestations can be the first modality revealing sarcoidosis with very few or no extra cardiac symptoms [4]. The EKG is usually abnormal, showing atrio-ventricular block, Bundle branch block – as it is the case of our patient- , supraventricular or ventricular tachyarrhythmias [6]. Echocardiography is usually without anomalies in clinically silent CS. But it can reveal non-specific abnormalities such as LV and/or RV systolic and diastolic dysfunction, or regional wall abnormalities with no coronary distribution. Some features may be more specific to CS, like basal interventricular thinning [7]; LV global longitudinal strain is an emerging sensitive, simple and promising technique that can predict adverse outcomes and also reveal subclinical cardiac dysfunction in patients with sarcoidosis [8]. CMR imaging is commonly used to evaluate cardiac involvement in patients with sarcoidosis, especially in clinically asymptomatic CS as it can reveal myocardial damage even in the absence of systolic dysfunction [9]. Basal segments of the septal and lateral wall are the most frequent site of LGE. However, there is no specific pattern of LGE that confirms CS. Our case emphasizes the importance of the combined use of PET scans and cardiac MRI for better detection of cardiac sarcoidosis. Not only do they allow an early diagnosis, but they can also assess the extent of disease and monitor treatment response.
4. Conclusion
Given the increased risk of sudden death, early screening of cardiac sarcoidosis should be done as soon as the disease is diagnosed in order to detect subclinical forms ;although it remains a challenge ,early diagnosis and quick initiation of the appropriate treatment (corticosteroids or other immunosuppressive agents) may improve the outcome.
Conflicts of Interest
Authors do not declare any conflict of interest.
References
- Hutchinson J. Illustrations of Clinical Surgery; Churchill: London, UK (1828-1913): 42.
- Vuitch F, Perry A. Causes of death in patients with sarcoidosis. A morphologic study of 38 autopsies with clinicopathologic correlations. Arch Pathol Lab Med 119 (1995): 167-172.
- Iwai K, Tachibana T, Takemura T, et al. Pathological studies on sarcoidosis autopsy. I. Epidemiological features of 320 cases in Japan. Pathology International 43 (1993): 372-376.
- Birnie DH, Nery PB, Ha AC, et al. (2016) Cardiac Sarcoidosis. Journal of the American College of Cardiology 68 (2016): 411-421.
- Greulich S, Gatidis S, Gräni C, et al. Hybrid Cardiac Magnetic Resonance/Fluorodeoxyglucose Positron Emission Tomography to Differentiate Active From Chronic Cardiac Sarcoidosis. JACC: Cardiovascular Imaging 15 (2022): 445-456.
- Mehta D, Lubitz SA, Franke Z, et al. Cardiac Involvement in Patients with Sarcoidosis: diagnostic and prognostic value of outpatient testing. Chest 133 (2008): 1426-1435.
- Ayyala US, Nair AP, Padilla ML. Cardiac Sarcoidosis, Clinics in Chest Medicine 29 (2008): 493-508.
- Joyce E, Ninaber MK, Katsanos S, et al. (2015) Subclinical left ventricular dysfunction by echocardiographic speckle-tracking strain analysis relates to outcome in sarcoidosis: Strain analysis and outcome in sarcoidosis. European Journal of Heart Failure 17 (2015): 51-62.
- Nagai T, Kohsaka S, Okuda S, et al. Incidence and Prognostic Significance of Myocardial Late Gadolinium Enhancement in Patients With Sarcoidosis Without Cardiac Manifestation. Chest 146 (2014): 1064-1072.