Immunomodulatory Effect of Electromagnetic Field in the Treatment of Traumatic Brain Injury
Article Information
Tye Patchana1, Devendra K Agrawal2, David Connett2, David Baron2, Dan Miulli1,2,3*
1Riverside University Health System, Department of Neurological Surgery, USA
2College of Osteopathic Medicine of the Pacific, Western University of the Health Sciences, Pomona, California, USA
3Arrowhead Regional Medical Center, Department of Neurological Surgery, USA
*Corresponding Author: Dan Miulli, Riverside University Health System, Department of Neurological Surgery, USA.
Received: 03 January 2023; Accepted: 13 January 2023; Published: 01 February 2023
Citation: Tye Patchana, Devendra K Agrawal, David Connett, David Baron, Dan Miulli. Immunomodulatory Effect of Electromagnetic Field in the Treatment of Traumatic Brain Injury. Journal of Biotechnology and Biomedicine 6 (2023): 32-41.
Share at FacebookAbstract
A review was performed utilizing PubMed and GoogleScholar to highlight the future directions of EMF research in the setting of brain pathology, specifically in ischemic and traumatic brain injury. Additionally, a critical review of the current state-of-the art of EMF use in treating brain pathology has been conducted. The authors have added to this large body of research their own experimental studies, including a description of the on-going studies. The field of EMF utilization in the diagnosis and treatment of brain injury is highly promising and warrant careful studies in clinically relevant experimental models followed by human trials in TBI.
Keywords
Electromagnetic Field; Immunomodulation; Traumatic Brain Injury (TBI); Neuroinflammation; Excitotoxicity
Electromagnetic Field articles Electromagnetic Field Research articles Electromagnetic Field review articles Electromagnetic Field PubMed articles Electromagnetic Field PubMed Central articles Electromagnetic Field 2023 articles Electromagnetic Field 2024 articles Electromagnetic Field Scopus articles Electromagnetic Field impact factor journals Electromagnetic Field Scopus journals Electromagnetic Field PubMed journals Electromagnetic Field medical journals Electromagnetic Field free journals Electromagnetic Field best journals Electromagnetic Field top journals Electromagnetic Field free medical journals Electromagnetic Field famous journals Electromagnetic Field Google Scholar indexed journals Immunomodulation articles Immunomodulation Research articles Immunomodulation review articles Immunomodulation PubMed articles Immunomodulation PubMed Central articles Immunomodulation 2023 articles Immunomodulation 2024 articles Immunomodulation Scopus articles Immunomodulation impact factor journals Immunomodulation Scopus journals Immunomodulation PubMed journals Immunomodulation medical journals Immunomodulation free journals Immunomodulation best journals Immunomodulation top journals Immunomodulation free medical journals Immunomodulation famous journals Immunomodulation Google Scholar indexed journals Traumatic Brain Injury (TBI) articles Traumatic Brain Injury (TBI) Research articles Traumatic Brain Injury (TBI) review articles Traumatic Brain Injury (TBI) PubMed articles Traumatic Brain Injury (TBI) PubMed Central articles Traumatic Brain Injury (TBI) 2023 articles Traumatic Brain Injury (TBI) 2024 articles Traumatic Brain Injury (TBI) Scopus articles Traumatic Brain Injury (TBI) impact factor journals Traumatic Brain Injury (TBI) Scopus journals Traumatic Brain Injury (TBI) PubMed journals Traumatic Brain Injury (TBI) medical journals Traumatic Brain Injury (TBI) free journals Traumatic Brain Injury (TBI) best journals Traumatic Brain Injury (TBI) top journals Traumatic Brain Injury (TBI) free medical journals Traumatic Brain Injury (TBI) famous journals Traumatic Brain Injury (TBI) Google Scholar indexed journals Neuroinflammation articles Neuroinflammation Research articles Neuroinflammation review articles Neuroinflammation PubMed articles Neuroinflammation PubMed Central articles Neuroinflammation 2023 articles Neuroinflammation 2024 articles Neuroinflammation Scopus articles Neuroinflammation impact factor journals Neuroinflammation Scopus journals Neuroinflammation PubMed journals Neuroinflammation medical journals Neuroinflammation free journals Neuroinflammation best journals Neuroinflammation top journals Neuroinflammation free medical journals Neuroinflammation famous journals Neuroinflammation Google Scholar indexed journals Excitotoxicity articles Excitotoxicity Research articles Excitotoxicity review articles Excitotoxicity PubMed articles Excitotoxicity PubMed Central articles Excitotoxicity 2023 articles Excitotoxicity 2024 articles Excitotoxicity Scopus articles Excitotoxicity impact factor journals Excitotoxicity Scopus journals Excitotoxicity PubMed journals Excitotoxicity medical journals Excitotoxicity free journals Excitotoxicity best journals Excitotoxicity top journals Excitotoxicity free medical journals Excitotoxicity famous journals Excitotoxicity Google Scholar indexed journals Medicine articles Medicine Research articles Medicine review articles Medicine PubMed articles Medicine PubMed Central articles Medicine 2023 articles Medicine 2024 articles Medicine Scopus articles Medicine impact factor journals Medicine Scopus journals Medicine PubMed journals Medicine medical journals Medicine free journals Medicine best journals Medicine top journals Medicine free medical journals Medicine famous journals Medicine Google Scholar indexed journals mechanisms articles mechanisms Research articles mechanisms review articles mechanisms PubMed articles mechanisms PubMed Central articles mechanisms 2023 articles mechanisms 2024 articles mechanisms Scopus articles mechanisms impact factor journals mechanisms Scopus journals mechanisms PubMed journals mechanisms medical journals mechanisms free journals mechanisms best journals mechanisms top journals mechanisms free medical journals mechanisms famous journals mechanisms Google Scholar indexed journals brain injury articles brain injury Research articles brain injury review articles brain injury PubMed articles brain injury PubMed Central articles brain injury 2023 articles brain injury 2024 articles brain injury Scopus articles brain injury impact factor journals brain injury Scopus journals brain injury PubMed journals brain injury medical journals brain injury free journals brain injury best journals brain injury top journals brain injury free medical journals brain injury famous journals brain injury Google Scholar indexed journals Neuroinflammatory articles Neuroinflammatory Research articles Neuroinflammatory review articles Neuroinflammatory PubMed articles Neuroinflammatory PubMed Central articles Neuroinflammatory 2023 articles Neuroinflammatory 2024 articles Neuroinflammatory Scopus articles Neuroinflammatory impact factor journals Neuroinflammatory Scopus journals Neuroinflammatory PubMed journals Neuroinflammatory medical journals Neuroinflammatory free journals Neuroinflammatory best journals Neuroinflammatory top journals Neuroinflammatory free medical journals Neuroinflammatory famous journals Neuroinflammatory Google Scholar indexed journals nutrition articles nutrition Research articles nutrition review articles nutrition PubMed articles nutrition PubMed Central articles nutrition 2023 articles nutrition 2024 articles nutrition Scopus articles nutrition impact factor journals nutrition Scopus journals nutrition PubMed journals nutrition medical journals nutrition free journals nutrition best journals nutrition top journals nutrition free medical journals nutrition famous journals nutrition Google Scholar indexed journals
Article Details
Introduction
Medicine has a history of several thousand years. During this time, patients have been continuously treated people with drugs and surgery. Surgery has been and is currently performed with a knife, usually cutting away tissue, rarely adding material. Lesions can be grossly removed but not on a cellular level, and therefore many cancers remain incurable. Chemotherapy affects the whole body, and many times with adverse effects. Conventional methods are limited in targeting a specific organ, tissue, or cell. However, recent advancements in science allowed the development of novel techniques and tools to treat some diseases with lasers, ultrasonic dissectors, and even focused beam radiation. The next era of medicine will begin when we can target specific sites in a pathological lesion using the new technology. However, it is critical to understand the underlying cellular, molecular, genetic, and epigenetic mechanisms of the disease process for precision and accuracy, and then control the direction of the treatment. However, this may not change the immediate chemistry of the cell and the tissue. It is critical to understand life on a molecular layer or smaller, and then use this information to control the organelles, the cells, the tissues, the organs, and the entire body, both in physiological and pathological conditions. It is now well established that electrons spin, protons vibrate, and molecules vibrate. Harnessing this vibration information may allow us to control almost all aspects of medicine. This is not an unusual concept. Indeed, ultrasound and radiation, with the help of electrical energy are used in the diagnosis, treatment, control of a disease process, and possibly cure. We even depend upon the natural vibrational signal of hydrogen when we use an MRI scanner. There has already been precedence exploring the resonance of targets and influencing those targets with the same vibration. Just as two opposite vibrational sound waves make silence together, a specific frequency may silence a deadly killer or bring back the sweet sound of life. Electrical energy using Pulsed Electrical Magnet Field (PEMF) is used in spinal cord stimulators to relieve pain, in bone stimulators to heal non unions, in tremors to calm the hands, and in the heart to capture a beat. Indeed, such techniques require a multidisciplinary approach with the integration of expertise and tools in chemistry, physics, biology, anatomy, physiology, pathology, and clinical medicine. The purpose of this review is to critically analyze the concept and the scientific foundation underlying PEMF-induced immunomodulation of traumatic injury in the brain. Furthermore, a discussion is provided on potential modification of the currently designed and tested equipment for experimental models to understand the underlying cellular and molecular mechanisms and examine the effect of PEMF in the management of traumatic brain injury.
Methods
A comprehensive literature search utilizing PubMed and Google Scholar was conducted between January 2001 through December 2022. Articles published in English were reviewed and included in this article. The following key words were utilized: EMF stroke, electromagnetic field, cerebrovascular accident, pulsed electro-magnetic field (PEMF), EMF, and traumatic brain injury, brain trauma, neuroprotection and EMF, immunomodulation, and various iterations. The findings were critically reviewed, and the information was synthesized into this comprehensive review article highlighting the clinical application and future directions.
Types and characteristics of traumatic brain injury
The inclusion of the myriad ways in which traumatic brain injury occurs under one umbrella term “Traumatic Brain Injury (TBI)” continues to make it difficult to identify therapeutic targets. TBI occurs in the setting of Motor Vehicle Accidents (MVA), falls, blast injuries, and penetrating brain trauma. The primary selection criteria for inclusion into TBI trials have been the GCS scoring system [1]. According to the current paradigm in TBI, there is an initial precipitating event (primary injury) followed by a clinicopathological cascade of events resulting in secondary injury [2]. Epidemiological studies have identified that mild, moderate, and severe TBI is a widespread pathology, and the milder forms of TBI may escape inclusion in the database [3]. Recent studies investigating the dynamics of TBI and MVA have demonstrated that the majority of TBIs involve skull fracture, Subarachnoid Hemorrhage (SAH), focal injury, and Subdural Hematoma (SDH) with the moderate-to-severe pathology in majority of the cases [4]. Penetrating brain injuries are a considerable type of TBI in both civilian and military personnel with mortality rates that can approach 90% in some studies [5]. Management of the patients who survive the primary injury hinges on the attenuation of the secondary injury resulting from cascading cellular events. Blast type injuries are also prominent in law and military personnel, leading to cognitive deficit, attention, and sleep impairments. Interesting, it was recently found that repeated low-to- moderate intensity blasts demonstrate similar neuropathological effects as one large blast [6].
Pathophysiology of Traumatic Brain Injury
Seminal to the definition of traumatic brain injury are temporary or permanent dysfunction or dysregulation of neural tissue resulting from external forces. These impairments can manifest as an altered state of consciousness, physical, psychological, and cognitive impairments [7]. As discussed, a primary inciting event occurs in TBI, followed by a series of events in which clinicians may intervene to improve outcomes. This secondary injury includes excitotoxicity, disruption of mitochondrial function, and accumulation of oxidative stress, lipid peroxidation, inciting apoptosis, and neuroinflammation (Figure 1).
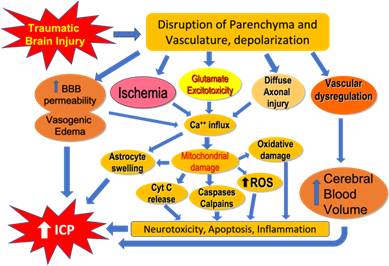
Figure 1: Schematic diagram showing the pathophysiology of traumatic brain injury resulting in increased Blood Brain Barrier (BBB) permeability, vasogenic edema, diffuse axonal injury, ischemia, mitochondrial dysfunction damage and increased Reactive Oxygen Species (ROS), neurotoxicity, apoptosis, inflammation in the brain. Vascular dysregulation results in increased cerebral blood volume and cerebral swelling. All these cellular events result in Increased Intracranial Pressure (ICP) and neurological deficits following traumatic brain injury.
Neuroinflammatory changes following TBI have similarities to brain injury following ischemic or reperfusion events [8]. Prostaglandins, cytokines (interleukins, chemokines, growth factors, etc.) that are pro-inflammatory, Reactive Oxygen Species (ROS) including free radicals comprise mediators are activated [9 -10]. The overall result of these mediators is a disruption of the normal permeability of the Blood Brain Barrier (BBB), which in turns causes edema and resultant neurological deficits (Figure 1). This neuroinflammation may even be prolonged over the course of years, as has been shown by Bye et al. with activation of macrophages, that in turn activate microglial cells, which increase release of astrocytes [11]. Failure of the normal homeostatic mechanisms of the BBB cause the release of excess neurotransmitters that have been implicated in TBI. Of these, glutamate is notable as failure of glutamate transporters has been implicated in the TBI cascade of events [12-13]. With release, glutamate and its associated metabolites bind to glutamate receptors causing activation and inducing excitotoxicity [8]. Among these, N-methyl-D-aspartate (NMDA) and Alpha-Amino-3-Hydroxy-5-Methyl-4-Isoxazoleproprionic Acid Receptor (AMPA) receptors allow influx of sodium, potassium, and calcium with resultant depolarization [14]. Excess calcium is implicated in the apoptotic cascade-including calpain, calcineurin, and caspases [15]. Recent evidence has correlated calpain to GCS in patients with TBI [16]. Mitochondrial dysregulation damage and oxidative stress are also implicated in the TBI cascade. Indeed, the influx of calcium itself into mitochondria may result in Reactive Oxygen Species (ROS), causing membrane depolarization that hinders ATP synthesis and further dysregulates the system [17]. Lastly, at a brain neuronal circuit level, shear stress on the longitudinally oriented neural structures leads to Diffuse Axonal Injury (DAI) (Figure 1). The resultant shear stress causes destruction to the axonal cytoskeleton, microtubules and neurofilaments [18].
Current Treatment and management strategies in TBI
The current paradigm in TBI management and treatment hinges on protection of the neural elements from the secondary injury discussed above. Initial goals remain protection of the airway and the circulatory system (Table 1). Given that hypotension doubles mortality, and hypoxia and hypotension triples mortality, support of these systems is paramount [19]. The Brain Trauma Foundation guidelines further recommend Intracranial Pressure (ICP) monitoring in patients with GCS3-8 with abnormal CT of the head or a normal CT scan with age greater than 40, motor posturing, or systolic blood pressure less than 90mmHg (http://braintrauma.org/coma/guidelines). At our institution, following hemodynamic stabilization and ICP control, we aim for initiation of early nutrition, physical occupational speech therapies, and if necessary, tracheostomy and percutaneous gastronomy tube placement. Additionally, our institution incorporates osteopathic manipulative treatment, which has been shown to decrease ICP [20] (Table 1). In Marino, et al [21], the authors review the connection between cerebral concussion, brain edema, and other diseases and how the glymphatic’s neurophysiology changes with different conditions. The glymphatic flow and drainage can be enhanced with treatments that affect the peri-arterial CSF influx route, the perivenous efflux route, and the transparenchymal route. These are currently facilitated with medications, procedures, and osteopathic manipulative treatment and may be further enhanced by manipulating the brain neuronal circuit with EMF.
|
Intervention |
Effect |
Notes |
|
AIRWAY |
Secure airway for oxygenation |
Hypoxia and hypotension Triples mortality |
|
CIRCULATION |
Supports cerebral perfusion pressure |
Hypotension doubles mortality |
|
ICP MONITORING |
Monitoring ICP, divert CSF If needed |
Maintain cerebral perfusion pressure, prevent ischemia |
|
SUPPORTIVE MEASURES |
Early therapy, Rehabilitation, nutrition |
Early rehabilitation to improve neural function |
Table 1: Current treatment strategies in traumatic brain injury.
Overview on Different Types of EMF
In addition to following the Brain Trauma Foundation Guidelines for the Management of Severe Head Injury, which depends on best medical and surgical care, TBI may be further diagnosed and treated using a new technology based on the brain’s electromagnetic field. 92% of patients with severe head injury suffer five minutes of ischemia and infarction is found at autopsy in 80% of patients dying of severe head injury [22-25]. Clearly, ischemia and infarction greatly affect patient outcomes in TBI and can be modulated with recognition and treatment. Current treatment includes, as stated above best medical and surgical care. Measuring and modulating the brain EMF may change outcomes as well. Figure 2 demonstrates the different types of EMF that have been used in stroke treatment as outlined by Moya Gomez et al., 2021 [26]. Several types of EMF may be utilized. Stationary Magnetic Field (SMF) requires an electromagnetic field with the same direction and magnitude due to a suppressed electrical component or 0 Hz frequency [26]. Low energy, time varying magnetic fields typify Pulsed Electro-Magnetic Field (PEMF). These low energy electric fields are comprised of specific shapes and amplitudes (Markov 2007) and may be found in a variety of different patterns including asymmetric, biphasic, quasi-rectangular or triangular [27]. Sinusoidal electromagnetic fields are non-pulsed (SEMF), in contrast to the on and off nature of PEMF, follows a sinusoidal waveform [26]. Non-pulsed SEMF has been shown to have a modulation effect on reactive oxygen species, on nitrous oxide species (NOS), and have neuroprotective effects in ischemia of the rat model [28].
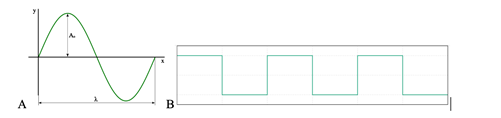
Figure 2: Different types of EMF used for stroke treatment, including (A) EMF based on sinusoidal waveform and frequency, and (B) Pulsed waveform, indicative of PEMF.
Electromagnetic Field and Radio Frequency in Ischemic Stroke
Low energy, time varying magnetic fields are Pulsed Electro-Magnetic Field (PEMF). Several studies have shown beneficial effects of PEMF on ischemic models [29-32]. The fields can be generated under various conditions from the purely electrical, to the magnetic, to the Radio Frequency (RF). This is athermal energy used since 1970s in the healing of bony non-unions. Each time that there is an electrical field there is a magnetic field. A varying electrical field does cause a varying magnetic field. Two methods for activating cells in the motor cortex utilizing PEMF, have been developed. This technique does not require direct contact with the surface of the brain. Merton and Morton introduced the first method, Transcranial Electrical Stimulation (TES), in 1980. It utilizes a high-voltage electrical pulse (up to 2 kV) of a short duration (less than 10 us) delivered through electrodes placed on the scalp with the anode over the cortical region to be stimulated (anode stimulation). This produces a magnetic field that passes largely unimpeded through the tissues of the head, in contrast to electric fields, which are diffused by head tissue. Oscillations in the magnetic field, in turn, induce an electrical current in the brain. The strength of the induced current is, in part, a function of the rate of change of the magnetic field, which varies with the current in the coil. Barker and CO-workers first reported the other form of noninvasive cortical cell activation, Transcranial Magnetic Stimulation (TMS), in 1985. They used a high-current (4 kA) and short-duration (110 us) pulse width. This was discharged through a coil placed on the scalp over the motor strip to produce a brief magnetic field, resulting in secondary currents inside the brain, which then depolarized cells within the motor cortex. For a given magnetic pulse, the direction of current flow through the coil, as well as the shape, size, and windings of the coil, play important roles in determining the effectiveness of TMS [33]. Both TMS and TES have limitations in depth of penetration. RF does not have these draw backs and in fact can and does penetrate all tissue levels. The distance traveled is proportional to the frequency.
Like the electrical stimulation of the exposed motor cortex in cats, monkeys and humans, spinal cord recordings during human spinal surgery have confirmed that single transcranial stimuli, either electrical or magnetic, evoke multiple descending volleys in humans. At above-threshold intensities, the shortest latency direct wave results from depolarization of the neuronal circuit of the pyramidal cell axons. Subsequently occurring indirect waves likely originate from nearby cortical neurons, which then excite pyramidal cells via synaptic connections. Due to their transynaptic origin, the number and amplitude of indirect waves are influenced by the excitability level of the motor cortex. Multiple descending volleys travel through the fast-conducting portion of the corticospinal tract and summate at the segmental motor centers to produce firing of alpha-motor neurons, which ultimately results in fairly synchronized short-latency Motor Evoked Potential (MEPs). This is what occurs daily as the neurophysiologists monitors patients during surgery. The current human stimulation of specific neuronal circuits during surgery is based upon years of research detailing the neuronal circuit and brain electrical activity. Individual cellular electrical action potentials have also been studied using an invasive probe. The action potential occurs over 1 millisecond (ms) (1000Hz); it is hyperpolarized for 2-3 ms (300 Hz) at +15 mV with a relative refractory period of 15 ms, yielding a relative Action Potential (AP) generation rate of 66.6 Hz. Post-synaptic potentials arrive at a frequency of 1000 Hz. Cell maintenance occurs at 30-5000 Hz. Additionally, inserting clinical micro-recording devices the frequencies of groups of cells have been discovered. Nordhausen developed a 3D silicon contact electrode array which records 100 separate channels of neuronal activity. Rousche and Normann reported that this same type of contact array called the Utah Intracortical Electrode Array stimulates and records in a single layer up to 1.5 mm beneath the surface. Cochlear cells have a frequency of 1000-4000 Hz at –20 to –80 mV, subthalamic nuclei 37 Hz, globus pallidal external cells 40 Hz, substantial nucleus reticulata 75 Hz, and globus pallidal interna cells 80-90 Hz. Individual atoms are known to vibrate at individual Larmor frequencies. The frequencies at 1 Tesla are H+ 42.6 MHz, H2 6.5 MHz, P31 17.2 MHz, C13 10.7 MHz, N14 3.1 MHz, N15 4.3 MHz, S33 3.3 MHz, F19 40.1 MHz. These frequencies are detected and “tuned in” by non-contact magnetic resonance imaging.
There is a wide range of varying electrical fields in the neuronal population. Rappaport [34] developed some of the initial investigations into this field with a PEMF utilizing a 27.1 MHz carrier that was modulated by 400 Hz pulse on rats [34]. The most studied electrical potentials are those obtained from electrodes inserted into the scalp. This is the coordination of brain activity. The activity occurs over a frequency from 1 to 50 Hz and from 2 to 100 mV. Specifically, EEG activity is classified as Beta (14-30 Hz), alpha (8-13 Hz), Theta (4-7 Hz), and delta activity (0.5-4 Hz). EEG activity is the summation of the cortical thickness of 2.5 mm, comprising a surface area of 2300 cm2, with a density of 10,000 neurons per mm3, yielding approximately 1010 neurons. Apart from the EEG, visual evoked potentials have frequencies of 1-100 Hz, brain stem auditory evoked responses range from 100 to 3000 Hz, and cognitive functions (P300) are measured at 3.3 Hz and +4 mV extending to P900 1.1 Hz. Memory generally occurs at 2.5 Hz and 10 mV. In a rabbit model of ischemic stroke, PEMF reduced edema as assessed by MRI and histological studies [35]. These EMF signals have been measured in detail in a continuous non-contact fashion at a distance and as a result, in a similar fashion the EMF signal can be transmitted to affect the brain neuronal circuit. At the cellular level, EMF has been found to have affect a large range of biological properties [36]. These include but are not limited to nitrous oxide modulation [37] superoxide modulation [38], apoptosis modulation [39] modulation of microcirculation [40], and modulation of inflammation and edema [41]. Pena-philippides et al., 2016 demonstrated cytokine/chemokine gene expression modulation by PEMF in mice studies with PEMFs demonstrating a suppression of inflammation in stroke, albeit in the later period [42]. Additionally, apoptosis indicators have been shown to be decreased by PEMF [43]. However, Bates et al. did find no improvement in functional or histological outcomes in a rat model [44]. It is known that both brain trauma and acute ischemic injury have similar mechanisms, involving cascades of cellular events with blood brain barrier disruption and influx of immune cells that further cause damage [45]. From the authors’ perspectives, trauma is stroke and inflammation.
There are limited reports on the investigation of EMF effect in TBI. Nonetheless, PEMF has been shown to have both anti-inflammatory and pro-regenerative effects in both animal models and in human trials. PEMF has been implicated in the reduction of both neuroinflammation and infarct size, highlighting it as a potential post-stroke therapy [42]. Normal mitochondrial function is imperative for the normal production of ATP and associated cellular function. During cerebral ischemia, this process is negatively affected, with hypoxia resulting in a breakdown of the aerobic respiration pathway in mitochondria. This results in lack of ATP and breakdown of the Na+/K+ pump, cellular swelling, and eventual neuronal death. PEMF has been found to act as a modulator of the adenosine receptor. This increases the function of endogenous adenosine with upregulation of A2A and A3 receptors in neuronal cells, with resulting neuroprotection [46]. Though studies have shown modulation of lipid peroxidation in rat models, the overall results are unclear, though suggesting protection to various cell types [47]. In view of the limited reports, there are several outstanding questions that warrant detailed careful investigations. These include: the effect of EMF on mitochondrial function and lipid peroxidation in brain cells; the effect of EMF on neuroinflammation, the release of excitatory amino acids from presynaptic nerve terminals in the brain, infiltration of inflammatory immune cells following injury, release of inflammatory cytokines, axonal degeneration, apoptosis, and impaired autophagy; the effect of EMF on oxidative stress; and EMF induce neuronal regeneration. There is no conclusive data on the effect of EMF in large animals in traumatic brain injuries.
The future of neuromodulation by EMF
There is a great need for the treatment of severe head injury, ischemic, and hemorrhagic stroke. There may be a revolution in the treatment of neurological disease. We have treated this system with surgery to cut parts out and medicine to try to add a change to the whole system to achieve the desired isolated effect. We expect to influence a cellular process by bathing it with chemicals, but it may be more probable that we can affect the organs, cells, and organelles on their level. We can cause these changes by utilizing the intrinsic properties. Of course, medication will continue to be needed to provide the substrate for reactions to take place. Indeed, the electrical energy is necessary and can be manipulated on all levels by the neurological system. Neuromodulation utilizes and changes the innate system in fighting disease in a continuous non-contact manner from a distance. However, utilizing the current treatments of Transcranial Magnetic Stimulation (TMS) and Transcranial Electrical Stimulation (TES) physicians can stimulate a large area of the intact human brain. The technologies have been applied generally, to affect an outcome, similarly to the discovery of x-rays and its treatment of lice and many other conditions. Radiation treatment has advanced to include focusing numerous sources of gamma radiation into pinpoints, at certain dosages, to kill tumor cells or desensitize nerve pain. The current EMF technology can in the intact human brain, in real-time, continuously, at a distance, identify specific brain neuronal circuits, the specific signals from those circuits, and pinpoint a continuous treatment to that diseased neuronal circuit, with real-time feedback. Such treatments may consist of an externally applied helmet and wave guide. The helmet and wave guide have been constructed. The EMF sensors easily pick up EMF activity in a non-contact manner through the scalp from any part and depth of the brain. The brain neuronal circuit EMF characteristics may then be adjusted. The transmitter and receiver sensors operate from 0.01 Hz to GHz. Voltage sensitivity is from picovolts to millivolts. Pulse width range from nanoseconds to milliseconds. Patterns recognition is completed with artificial intelligence analyzing the EMF wave. The pattern orientation of the transmitted EMF is adjusted to account for the different directions of the signals obtained. The depth of penetration and area of transmission will initially be measured and adjusted in the pig model. Once the appropriate EMF measurement are obtained in the traumatic pig and human, and a modulated PEMF applied in the pig trauma model, a modulating PEMF may be able to be applied in the human traumatic brain injury in the hospital setting, continuously, in a non-contact manner. This new diagnosis and treatment could then be applied at the time of brain injury in the human at the scene until specific injuries can be ascertained and appropriate frequencies applied. These may be frequencies to inhibit some areas while stimulating other areas to survive. This helmet and waveguide can be shaped to allow the insertion of ICP monitors and measure the frequencies of the dysfunctional brain. After stabilization of damage the helmet and waveguides would be applied during rehabilitation.
This same type of modulation can also take place during regular learning. A 3.3 Hz, 10 mV pulse to re-enforce the education would follow a stimulus. Southworth reported on the Alpha Stim CES to enhance attention and the ability to learn new tasks. Other uses would be in psychological conditions. The cap could be applied in the acute setting of trauma and stroke and worn during the chronic phase of trauma and stroke to affect memory, concentration, impulsivity, and other behaviors. Cyberonics promotes the Vagus nerve stimulator in the treatment of treatment resistant major non-psychotic depression. Utilizing data obtained from the shark’s electrical signature, the helmet and waveguides could be modulated to that frequency to inhibit cancer growth. Psychological symptoms such as cognitive deficit, impulsivity, mania, depression, anxiety, and a host of neuropsychiatric conditions are unfortunately common following TBI. Additionally, the pharmacological treatments of these conditions have been studied in few controlled clinical trials [48]. Unfortunately, the chronic sequala of TBI usually does not garner as much attention in the literature as the acute manifestations, though a large number will go on to have persistent sequelae of neuropsychological problems [49]. Studies have shown that a reduction in oxidative stress may occur with low frequency EMF utilized in post-acute stroke rehabilitation patients [50]. In this setting and in trauma, neuromodulation may offer a treatment with fewer side effects than medications. Future studies will help to elucidate this.
Future studies
One of the primary goals of this article is to suggest possible future modulations of neuronal circuits in complex neurological diseases such as the physiological and psychological ramifications of head trauma and stroke. The project is divided into multiple parts. The first involves measuring the normal frequencies, electrical activity and magnetic field in the pig and human. Animal models will need to be used because the microenvironment is important negating single cell systems. The “signal” will be the electrical and magnetic signature, calibrated by amplitude, duration, type of wave, frequency, and flux. This will involve applying advanced technology as well as modifying the current technology. The information will have to be processed and refined. After the information is obtained in the normal state it will have to be obtained in the diseased state. Next, in the pig TBI model EMF in a continuous non-contact fashion will be applied to potentially reverse neuronal circuit damage. Finally, this type of information can be used to treat human problems of head trauma and stroke including its psychological components. A helmet and waveguide detector will be placed on the pig model to look for reproducible frequencies. Prior studies, as stated above, demonstrate information in the Hz range, which is the normal EEG, and encourage the investigation, quantification, and qualification for EMF signals in the kHz, MHz, THz, and GHz range. The second phase will be the recording after clinical lesioning, reproducing trauma or stroke in the pig model, once again looking for the differences in the information, from normal to abnormal. The lesion will be trauma, and secondary ischemia injury and anoxic injury. The third phase would be the neuromodulation of abnormal brain cell circuits, cellular, and psychological abnormalities: brain infarction injury, ischemia, trauma, and anoxic injury. This device can be applied easily and using telemetry would allow the treatment of patients at a distance. Some of the challenges to bringing PEMF modulation in the setting of brain injury to fruition involve the lack of a standardized setting for EMF use. Within the literature there is a large array of differences in the use of wave type, intensity, type of technology, frequency usage, and exposure time creating confounding variables [41]. Overall, there is convincing data that demonstrate that EMF is effective in the treatment of brain injury, whether that injury occur via acute ischemia or traumatic brain injury, which has been corroborated by animal studies.
In our initial testing, proprietary induction sensors (patented, Model BS-1000, Quasar Federal Systems, San Diego, CA) were used to measure the EMF of specific human brain activities in a non-contact and non-invasive fashion using a prototype lightweight helmet as previously described by Wiginton et al. and Brazdzionis et al. [51-53]. This helmet was constructed using two layers of Mu-metal (MuMETAL®, Magnetic Shield Corporation, Bensenville, IL) with inner and outer layers of interlaced copper mesh to absorb and reflect the external EMF signal. Both layers of Mu-metal were separated approximately 2.5 cm. Four holes were drilled within the helmet to accommodate four 18-inch induction sensors. These four sensors (Bx, By, Bz, and B319) were configured to bitemporal and bifrontal locations as previously described [51-53]. These induction sensors require a 9-volt power supply and consume 3.3 mA in order to measure non-direct current, low-magnetic fields in a single axis between 1 Hz and 30 kHz through the use of a solenoid coil around a 0.2 inch diameter high-permeability core. Each sensor was ensured to be positioned with the positive end (+) oriented toward the scalp. Furthermore, the Bz sensor was placed on the right side at approximately the right motor strip with the trajectory pointed toward the left motor strip, the By sensor was placed at the right temporal region with a trajectory oriented toward the left motor strip, the B319 sensor was placed on the left side at approximately the location of the left motor strip with a trajectory orienting toward the left motor strip, and the Bx sensor was placed in the left temporal region with a trajectory pointed toward the left motor strip. Each sensor was placed within plastic Polyvinyl Chloride (PVC) piping. The PVC piping containing the sensor was then wrapped in Mu-metal to form an EMF channel to function similarly to a waveguide to focus the generated EMF by targeted brain tissue and exclude external EMF signal. Sensor recordings were recorded on a laptop and post-processed as described by Wiginton et al. using Fast Fourier Transformation (FFT) [3]. Detection sensitivity for sensors was set at 1.5 pTrms/rtHz at 1 Hz, 0.15 pTrms/rtHz at 10 Hz, 0.025 pTrms/rtHz at 100 Hz, and 0.02 pTrms/rtHz at 1 kHz and above. Sensor responses were captured between 1 Hz and 2 kHz, and a gain filter module was used set with a 10x gain for amplifying signal with a 2-kHz low-pass filter for anti-aliasing. Data capture was completed at 5,000 samples per second. Each test was conducted with a non-ferromagnetic metronome placed outside the Faraday cage containing the human participant, helmet, and sensors. This metronome produced a sound at 120 bpm (2 Hz) to synchronize activities and to detect a reproducible recording of brain activity from each subject. Utilizing a helmet, electromagnetic field channels will be connected to the helmet construct with the sensors placed within each channel. Thin lines connecting the helmet to the amplifier and gain filter module and from the amplifier to the computer and recording equipment represent wires connecting the devices [52]. Volunteer human subjects performed each test by participating in designated reproduced activities. Subjects were placed supine and donned the helmet. Activities involving hand or foot tapping were completed at a rate of 2 Hz. Of note, within the Faraday cage, subjects were asked to keep their eyes closed to limit visual stimulus. Furthermore, the shielded room and Faraday cage were dark to limit visual stimulation. Mechanistically, hand tapping was completed through the protocol delineated by Brazdzionis et al. [52]. This tapping activity entailed lifting the hand as a singular unit while leaving the wrist fixed on the ground. The tips of the fingers were elevated 6 cm and then brought back to the ground. Each elevation and return to baseline consisted of a single tap. Foot tapping was completed with the knees and hips flexed. Each foot was dorsiflexed off the ground with the heel remaining fixed on the ground. Toes were elevated 6 inches through dorsiflexion and plantarflexed back to the ground. Each dorsiflexion and plantar flexion cycle was designated as a single tap. Each test started with 30 seconds of "rest" wherein the participant was asked to not tap and try to maintain a lack of higher cortical thoughts. This rest was followed by trials of 60 seconds of the designated activities with 30-second rest "breaks" between each 60-second activity bin. For analysis, summative FFT transformed data placed into 20-second bins were plotted graphically. Testing was completed to investigate the ability and the effects of brain attenuation of external magnetic fields on stereotyped motor activities. This was done to assess the effectiveness of measurement of the generated magnetic field in the brain and the effects of external magnetic field transmission of the generated field. Further investigation included evaluating sensors from a distance when using Mu-metal shielding as an EMF channel functioning like a waveguide to evaluate the decay of the magnetic field over distance.
Conclusion
Overall, this review has highlighted some of the future directions of this novel EMF research in the setting of brain pathology, specifically in ischemic and traumatic brain injury. Additionally, a critical review of the current state-of-the art of EMF continuous non-contact treatment of brain injury insults has been conducted. The authors have additionally added to this large body of research their own experimental studies, including a description of the on-going studies. Challenges include completing studies to streamline the process and evaluate therapeutic and diagnostic metrics. However, the novel field of EMF utilization in the diagnosis and treatment of brain injury is highly promising and warrant careful studies in clinically relevant experimental models followed by human trials in TBI.
Author Contributions
Concept and design: DM, TP, DKA; Literature Search: DM, TP; Critical review and interpretation of the findings: TP, DM; Drafting the article: TP, DKA, DM; Revising and editing the manuscript: TP, DKA, DM; Final approval of the article: TP, DKA, DC, DB, DM
Funding
The research work of DKA is supported by the grants R01 HL144125 and R01 HL 147662 from the National Institutes of Health, USA. The contents of this article are solely the responsibility of the authors and do not necessarily represent the official views of the Natinal Institutes of Health.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable
Data Availability Statement
Not applicable since the information is gathered from published articles.
Acknowledgments
None
Conflicts of Interest
The authors declare no conflict of interest.
References
- Saatman KE, Duhaime AC, Bullock R, et al. Classification of Traumatic Brain Injury for Targeted Therapies. Journal of Neurotrauma 25 (2008): 719-738.
- Prough DS, DeWitt DS, Jenkins LW. Secondary Injury after Clinical Traumatic Brain Injury. In J. O. Johnson R, J Sperry, TH Stanley (Eds.), Neuroanesthesia: Papers presented at the 42nd Annual Postgraduate Course in Anesthesiology (1997): 153-164.
- Summers CR, Ivins B, Schwab KA. Traumatic brain injury in the United States: an epidemiologic overview. Mt Sinai J Med 76 (2009): 105-110.
- Baker CE, Martin P, Wilson MH, et al. The relationship between road traffic collision dynamics and traumatic brain injury pathology. Brain Communications 4 (2022): fcac033.
- Hawryluk GWJ, Selph S, Lumba-Brown A, et al. Rationale and Methods for Updated Guidelines for the Management of Penetrating Traumatic Brain Injury. Neurotrauma Reports 3 (2022): 240-247.
- Ravula AR, Das T, Gosain A, et al. An update on repeated blast traumatic brain injury. Current Opinion in Biomedical Engineering 24 (2022): 100409.
- Maxwell WL. Traumatic brain injury in the neonate, child and adolescent human: An overview of pathology. International Journal of Developmental Neuroscience 30 (2012): 167-183.
- Thapa K, Khan H, Singh TG, et al. Traumatic Brain Injury: Mechanistic Insight on Pathophysiology and Potential Therapeutic Targets. J Mol Neurosci 71 (2021): 1725-1742.
- Plata-Salaman CR. Brain cytokines and disease. Acta Neuropsychiatrica 14 (2002): 262-278.
- Werner C, Engelhard K. Pathophysiology of traumatic brain injury. BJA: British Journal of Anaesthesia 99 (2007): 4-9.
- Bye N, Habgood MD, Callaway JK, et al. Transient neuroprotection by minocycline following traumatic brain injury is associated with attenuated microglial activation but no changes in cell apoptosis or neutrophil infiltration. Experimental Neurology 204 (2007): 220-233.
- Berry JAD, Wacker M, Menoni R, et al. Return-to-Play After Concussion: Clinical Guidelines for Young Athletes. J Am Osteopath Assoc 119 (2019): 833-838.
- Berry JAD, Elia C, Sweiss R, et al. Pathophysiologic mechanisms of concussion, development of chronic traumatic encephalopathy, and emerging diagnostics: A narrative review. J Am Osteopath Assoc (2020).
- Brustovetsky T, Bolshakov A, Brustovetsky N. Calpain activation and Na+/Ca2+ exchanger degradation occur downstream of calcium deregulation in hippocampal neurons exposed to excitotoxic glutamate. Journal of Neuroscience Research 88 (2010): 1317-1328.
- Weber J. Altered Calcium Signaling Following Traumatic Brain Injury. Frontiers in Pharmacology 3 (2012).
- Wang Y, Brazdzionis J, Dong F, et al. P13BP, a Calpain-2-Mediated Breakdown Product of PTPN13, Is a Novel Blood Biomarker for Traumatic Brain Injury. J Neurotrauma 38 (2021): 3077-3085.
- Xiong Y, Peterson Pl, Lee CP. Effect of N-Acetylcysteine on Mitochondrial Function Following Traumatic Brain Injury in Rats. Journal of Neurotrauma 16 (1999): 1067-1082.
- Andriessen TMJC, Jacobs B, Vos PE. Clinical characteristics and pathophysiological mechanisms of focal and diffuse traumatic brain injury. Journal of Cellular and Molecular Medicine 14 (2010): 2381-2392.
- Spaite DW, Hu C, Bobrow BJ, et al. The Impact of Combined Prehospital Hypotension and Hypoxia on Mortality in Major Traumatic Brain Injury. Ann Emerg Med 69 (2017): 62-72.
- Kashyap S, Brazdzionis J, Savla P, et al. Osteopathic Manipulative Treatment to Optimize the Glymphatic Environment in Severe Traumatic Brain Injury Measured With Optic Nerve Sheath Diameter, Intracranial Pressure Monitoring, and Neurological Pupil Index. Cureus. 13 (2021): e13823.
- Marino MA, Petrova S, Sweiss R, et al. A Review of Glymphatics and the Impact of Osteopathic Manipulative Treatment in Alzheimer's Disease, Concussions, and Beyond. Cureus 14 (2022): e23620.
- Andrews PJ, Piper IR, Dearden NM, et al. Secondary insults during intrahospital transport of head-injured patients. Lancet 335 (1990): 327-330.
- Chesnut RM; Marshall LF, Klauber MR, et al. The role of secondary brain injury in determining outcome from severe head injury. The Journal of Trauma: Injury, Infection, and Critical Care 34 (1993): 216-222.
- Graham DI, Ford I, Adams JH, et al. Ischaemic brain damage is still common in non-missile head injury. Journal of Neurology, Neurosurgery and Psychiatry 52 (1989): 346.
- Patricia AJ, Andrews PJ, Midgley S, et al. Measuring the burden of secondary insults in head injured patients during intensive care. J Neurosurg Anesthesiol 6 (1994): 4-14.
- Moya GA, Font LP, Brône B, et al. Electromagnetic Field as a Treatment for Cerebral Ischemic Stroke. Frontiers in Molecular Biosciences 8 (2021).
- Shupak NM, Prato FS, Thomas AW. Therapeutic uses of pulsed magnetic-field exposure: A review. URSI Radio Science Bulletin 307 (2003): 9-32.
- Font LP, Cardonne MM, Kemps H, et al. Non-pulsed Sinusoidal Electromagnetic Field Rescues Animals From Severe Ischemic Stroke via NO Activation. Frontiers in Neuroscience. 13 (2019): 561.
- Grant G, Cadossi R, Steinberg G. Protection against focal cerebral ischemia following exposure to a pulsed electromagnetic field. Bioelectromagnetics 15 (1994): 205-216.
- Balind SR, Selakovic V, Radenovic L, et al. Extremely Low Frequency Magnetic Field (50 Hz, 0.5 mT) Reduces Oxidative Stress in the Brain of Gerbils Submitted to Global Cerebral Ischemia. PLOS ONE 9 (2014): e88921.
- Extremely Low Frequency Magnetic Field (50 Hz, 0.5 mT) Reduces Oxidative Stress in the Brain of Gerbils Submitted to Global Cerebral Ischemia | PLOS ONE (2022).
- Rauš S, Selakovic V, Radenovic L, et al. Extremely low frequency magnetic field induced changes in motor behaviour of gerbils submitted to global cerebral ischemia. Behavioural Brain Research 228 (2012): 241-246.
- McKay W, Stokic D, Dimitrijevic M. Assessment of corticospinal function in spinal cord injury using transcranial motor cortex stimulation: a review. J Neurotr 14 (1997): 539-548.
- Rappaport Z, Young W. Effect of pemf on calcium tissue changes in focal ischaemia. Neurol Res 12 (1990): 95-98.
- Grant G, Cadossi R, Steinberg G. Protection against focal cerebral ischemia following exposure to a pulsed magnetic field. Bioelectromagnetics 15 (1994): 205-216.
- Pickard WF, Moros EG. Energy deposition processes in biological tissue: nonthermal biohazards seem unlikely in the ultra-high frequency range. Bioelectromagnetics 22 (2001): 97-105.
- Akdag MZ, Bilgin MH, Dasdag S, et al. Alteration of Nitric Oxide Production in Rats Exposed to a Prolonged, Extremely Low-Frequency Magnetic Field. Electromagnetic Biology and Medicine 26 (2007): 99-106.
- Naarala J, Kesari KK, McClure I, et al. Direction-Dependent Effects of Combined Static and ELF Magnetic Fields on Cell Proliferation and Superoxide Radical Production. BioMed Research International (2017): e5675086.
- Palumbo R, Capasso D, Brescia F, et al. Effects on apoptosis and reactive oxygen species formation by Jurkat cells exposed to 50 Hz electromagnetic fields. Bioelectromagnetics 27 (2006): 159-162.
- Li X, Xu H, Lei T, et al. A pulsed electromagnetic field protects against glutamate-induced excitotoxicity by modulating the endocannabinoid system in HT22 cells. Frontiers in Neuroscience 11 (2017): 42.
- Liu J, Chang K, Chen W, et al. Astroglial injury caused by extreme low frequency electromagnetic field. Abstract AANS (1998): 113.
- Pena-Philippides JC, Yang Y, Bragina O, et al. Effect of Pulsed Electromagnetic Field (PEMF) on Infarct Size and Inflammation After Cerebral Ischemia in Mice. Transl Stroke Res 5 (2014): 491-500.
- Urnukhsaikhan E, Mishig-Ochir T, Kim SC, et al. Neuroprotective Effect of Low Frequency-Pulsed Electromagnetic Fields in Ischemic Stroke. Appl Biochem Biotechnol 181 (2017): 1360-1371.
- Bates KA, Clark VW, Meloni BP, et al. Short-term low intensity PMF does not improve functional or histological outcomes in a rat model of transient focal cerebral ischemia. Brain Res 1458 (2012):76-85.
- Dugue R, Barone FC. Ischemic, traumatic and neurodegenerative brain inflammatory changes. Future Neurology 11 (2016): 77-96.
- Capone F, Salati S, Vincenzi F, et al. Pulsed Electromagnetic Fields: A Novel Attractive Therapeutic Opportunity for Neuroprotection After Acute Cerebral Ischemia. Neuromodulation: Technology at the Neural Interface 25 (2022): 1240-1247.
- Musaev AV, Ismailova LF, Gadzhiev AM. [Influence of (460 MHz) electromagnetic fields on the induced lipid peroxidation in the structures of visual analyzer and hypothalamus in experimental animals. Vopr Kurortol Fizioter Lech Fiz Kult 5 (2005): 17-20.
- Lee HB, Lyketsos CG, Rao V. Pharmacological management of the psychiatric aspects of traumatic brain injury. International Review of Psychiatry 15 (2003): 359-370.
- Sudarsanan S, Chaudhary S, Pawar A, et al. Psychiatric Effects of Traumatic Brain Injury. Medical Journal Armed Forces India 63 (2007): 259-263.
- Cichon N, Bijak M, Synowiec E, et al. Modulation of antioxidant enzyme gene expression by extremely low frequency electromagnetic field in post-stroke patients. Scand J Clin Lab Invest 78 (2018): 626-631.
- Wiginton J 4th, Brazdzionis J, Patchana T, et al. Novel method of electromagnetic field measurements of the human brain. Cureus 14 (2022): e21982.
- Brazdzionis J, Wiginton J, Patchana T, et al. Evaluating the intrinsic electromagnetic field generated by neurons from repetitive motor activities in humans with a non-contact non-invasive electromagnetic helmet. Cureus 14 (2022): e23006.
- Wiginton J, Brazdzionis J, Patchana T, et al. Measuring electromagnetic field activity generated by neurons in vivo by humans with thoughts of repetitive motor activities and emotional thoughts. Cureus 14 (2022): e23332.
