Autologous Fibrin Glue as a Novel Platelet Concentration in Palatal Wound Healing
Article Information
Mustafa Özay Uslu*, Mehmet K?z?ltoprak, Ömer Alperen K?rm?z?gül
Department of Periodontology, Faculty of Dentistry, Inonu University, Malatya, Turkey
*Corresponding Author: Mustafa Özay USLU, Department of Periodontology, Faculty of Dentistry, Inonu University, 44280, Malatya, Turkey
Received: 06 September 2019; Accepted: 26 September 2019; Published: 30 September 2019
Citation: Mustafa Özay Uslu, Mehmet Kızıltoprak, Ömer Alperen Kırmızıgül. Autologous Fibrin Glue as a Novel Platelet Concentration in Palatal Wound Healing. Dental Research and Oral Health 2 (2019): 040-046.
Share at FacebookAbstract
Aim: Free gingival grafts (FGG) are one of the most commonly used autogenous soft tissue grafts to treat mucogingival problems. Postoperative complications can be seen in the FGG as the donor area which heals by secondary wound healing. In this study, it was aimed to minimize the complications that can be seen in the donor area after free gingival graft operation with ‘autologue fibrin glue’(AFG) application.
Methods: Systemically healthy, 28 year-old female patient applied to Department of Periodontology with complaints of bleeding, pain and esthetics due to recession of gingiva in the lower central teeth. As a result of the clinical examination, inadequate attached gingiva was detected in the area related. FGG operation was planned to increase the amount of gingival tissue.
In addition, AFG application to the palatal region was planned in order to minimize the complications that may be seen in the donor area after the operation. AFG, which were obtained from the patient's blood, was applied to the open wound surface. Wound healing was assessed with clinical parameters at 3, 7, 14 days and 1 month postoperatively.
Results: Wound healing was achieved without any complications. It was seen that the use of AFG in palatal wound healing, reduced post-operative complications and accelerated the healing process.
Conclusion: The adhesive structure of AFG may be preferred to another platelet concentrates with ease of use in the palatal region.
Keywords
Autologous Fibrin Glue, Free Gingival Graft, Palatal Wound Healing
Autologous Fibrin Glue articles, Free Gingival Graft articles, Palatal Wound Healing articles
Autologous Fibrin Glue articles Autologous Fibrin Glue Research articles Autologous Fibrin Glue review articles Autologous Fibrin Glue PubMed articles Autologous Fibrin Glue PubMed Central articles Autologous Fibrin Glue 2023 articles Autologous Fibrin Glue 2024 articles Autologous Fibrin Glue Scopus articles Autologous Fibrin Glue impact factor journals Autologous Fibrin Glue Scopus journals Autologous Fibrin Glue PubMed journals Autologous Fibrin Glue medical journals Autologous Fibrin Glue free journals Autologous Fibrin Glue best journals Autologous Fibrin Glue top journals Autologous Fibrin Glue free medical journals Autologous Fibrin Glue famous journals Autologous Fibrin Glue Google Scholar indexed journals Free Gingival Graft articles Free Gingival Graft Research articles Free Gingival Graft review articles Free Gingival Graft PubMed articles Free Gingival Graft PubMed Central articles Free Gingival Graft 2023 articles Free Gingival Graft 2024 articles Free Gingival Graft Scopus articles Free Gingival Graft impact factor journals Free Gingival Graft Scopus journals Free Gingival Graft PubMed journals Free Gingival Graft medical journals Free Gingival Graft free journals Free Gingival Graft best journals Free Gingival Graft top journals Free Gingival Graft free medical journals Free Gingival Graft famous journals Free Gingival Graft Google Scholar indexed journals Palatal Wound Healing articles Palatal Wound Healing Research articles Palatal Wound Healing review articles Palatal Wound Healing PubMed articles Palatal Wound Healing PubMed Central articles Palatal Wound Healing 2023 articles Palatal Wound Healing 2024 articles Palatal Wound Healing Scopus articles Palatal Wound Healing impact factor journals Palatal Wound Healing Scopus journals Palatal Wound Healing PubMed journals Palatal Wound Healing medical journals Palatal Wound Healing free journals Palatal Wound Healing best journals Palatal Wound Healing top journals Palatal Wound Healing free medical journals Palatal Wound Healing famous journals Palatal Wound Healing Google Scholar indexed journals gingival tissue articles gingival tissue Research articles gingival tissue review articles gingival tissue PubMed articles gingival tissue PubMed Central articles gingival tissue 2023 articles gingival tissue 2024 articles gingival tissue Scopus articles gingival tissue impact factor journals gingival tissue Scopus journals gingival tissue PubMed journals gingival tissue medical journals gingival tissue free journals gingival tissue best journals gingival tissue top journals gingival tissue free medical journals gingival tissue famous journals gingival tissue Google Scholar indexed journals Periodontology articles Periodontology Research articles Periodontology review articles Periodontology PubMed articles Periodontology PubMed Central articles Periodontology 2023 articles Periodontology 2024 articles Periodontology Scopus articles Periodontology impact factor journals Periodontology Scopus journals Periodontology PubMed journals Periodontology medical journals Periodontology free journals Periodontology best journals Periodontology top journals Periodontology free medical journals Periodontology famous journals Periodontology Google Scholar indexed journals mouthwash articles mouthwash Research articles mouthwash review articles mouthwash PubMed articles mouthwash PubMed Central articles mouthwash 2023 articles mouthwash 2024 articles mouthwash Scopus articles mouthwash impact factor journals mouthwash Scopus journals mouthwash PubMed journals mouthwash medical journals mouthwash free journals mouthwash best journals mouthwash top journals mouthwash free medical journals mouthwash famous journals mouthwash Google Scholar indexed journals palatal wound healing articles palatal wound healing Research articles palatal wound healing review articles palatal wound healing PubMed articles palatal wound healing PubMed Central articles palatal wound healing 2023 articles palatal wound healing 2024 articles palatal wound healing Scopus articles palatal wound healing impact factor journals palatal wound healing Scopus journals palatal wound healing PubMed journals palatal wound healing medical journals palatal wound healing free journals palatal wound healing best journals palatal wound healing top journals palatal wound healing free medical journals palatal wound healing famous journals palatal wound healing Google Scholar indexed journals
Article Details
1. Introduction
The adequate attached gingiva is necessary to maintain oral hygiene and periodontal health [1]. Free gingival graft (FGG) is the most commonly used autogenous soft tissue graft to treat mucogingival problems such as inadequate gingival and gingival recessions around the teeth and implants [2]. In this technique, a soft-tissue graft is taken from the palate and open wound heals by secondary healing [3]. However, postoperative complications such as excessive bleeding, delayed wound healing, pain, sensory loss and necrosis can be seen at the donor site [4]. Many studies had used hemostatic agents, ozonated oil, herbal products, antibacterial agents, bioactive substances, low level laser therapy and platelet concentrate to reduce postoperative complications [5, 6]. Recently, platelet concentrates have become popular in periodontal regenerative therapy due to autologous nature. The regenerative potentials of platelet concentrate were associated with the presence of various growth factors released in a-granules when platelets were activated. This event supports wound healing after surgical periodontal treatment, reduces postoperative pain and swelling with anti-inflammatory properties [7]. In additional, there were also studies reporting anti-bacterial potentials [8]. The researchers obtained different platelet concentrations when centrifuging whole blood at different times for different periods. The different platelet concentrations, such as ‘Platelet Rich Plasma’ (PRP), ‘Platelet Rich Fibrin’(PRF), ‘Titanium-prepared Platelet Rich Fibrin’ (T-PRF) obtained were used in various regenerative therapies including palatal wound healing [5, 6, 9]. 'Autologous fibrin glue' (AFG) was an injectable platelet concentration that presented in 2010 by Sohn [10, 11]. AFG had been used in the construction of 'sticky bone', which is mixed with bone grafts, allowing the graft to remain more stable in the bone defect [10]. The aim of this case report was to evaluate the effect of AFG on secondary wound healing after FGG. This case report was the first to assess the effect of AFG on wound healing.
2. Case Report
Systemically healthy, 28 year-old female patient applied to ?nönü University Faculty of Dentistry Department of Periodontology with complaints of bleeding, pain and esthetics due to recession of gingiva in the lower central teeth. As a result of the clinical examination, inadequate attached gingiva was detected in the area related. FGG operation was planned to increase the amount of gingival tissue. In addition, AFG application to the palatal region was planned in order to minimize the complications that may be seen in the donor area after the operation.
2.1 Surgical procedures
Local anesthesia (Maxicaine Fort, Articaine hydrochloride 80 mg + epinephrine 0.020 mg / ampule) was applied to the recipient and donor area. A 1.74 mm thick split-thickness graft (15 mm × 6 mm size) was obtained from the palatal region. The palatal graft was stabilized with resorbable suture (7/0 (PGA Pegesorb, Do?san, Turkey) to cover the recipient area. 9 ml venous blood collected from the patient was transferred to the tube without any anticoagulants. The protocol recommended by Sohn et al., venous blood collected in the tube was centrifuged at 2700 RPM (approximately 692 g) for 2 min in a special centrifuge (Medifuge, Silfradent, Italy) to obtain two layers: the lower layer of red blood cells and the upper layer of AFG. The AFG at the top of the tube was collected by means of an injector and transferred to the metal bowl. The polymerization was allowed to stand for 15-20 minutes. After polymerization occurred, the membrane form of AFG in the metal bowl was applied to the wound surface in the donor site (Figure 1). Then the wound surface was covered with the adhesive gel part which was obtained from the same blood. The donor area was covered with aluminum foil and periodontal pat (Coe-Pak, GC America Inc, IL, USA). The patient was told about the postoperative recommendations, analgesics and mouthwash were prescribed. Pain, epithelialization and wound healing were evaluated at postoperative 3rd, 7th, 14th days and 1st month. In addition, the palatal tissue thickness was evaluated at postoperative 1st month. No postoperative complications were observed.
2.2 Pain assessment
Pain assessment was performed with the Visual analogue scale (VAS) (0-10). In this method, patients were asked to question the pain as a score between 0 (no pain) and 10 (the most severe pain in my life).
2.3 H2O2-bubbling test
Wound epithelialization was assessed by the H2O2 - bubble test. In this test, 3% H2O2 was applied with a syringe after the wound surface was dry. The appearance of bubbles indicates that epithelization was not complete (Figure 2). Bubble formation did not observe if the epithelium was completed.
2.4 Wound healing
The wound healing process was assessed by a modification of the Manchester Scar Scale which including color, contour, and distortion categories (Table 1). In these categories, where the range of the score ranges from 1 to 4, a low score indicates that healing is good [12].
2.5 Palatal tissue thickness
After the graft boundaries were determined at the time of operation, tissue thickness was measured with electronic calipers via spreaders from four spots (2 mm distance between the mesial and distal sides of the graft) and the midpoint of the graft, and averaged. The same procedure was repeated 1 months after the surgery. The clinical appearance of the healing in the palatal area was shown in (Figure 3).
The evaluation of VAS, H2O2 and Manchester scales was shown in (Table 2). The VAS score was 1 on the postoperative 3rd day and no pain was shown at any other controls. The epithelialization test with H2O2, 80% of the total wound surface were epithelialized on the postoperative 14th day. There was no area without epithelization a postoperative 1st month. In the evaluation of wound healing with the Manchester scar scale, it was seen that wound healing was at a good level for the postoperative 14th day. Initial and 1st month palatal tissue thickness was shown in (Table 3). At the end of the 1st month, a difference of 0.22 mm of palatal tissue thickness was detected when compared to the initial measurements.
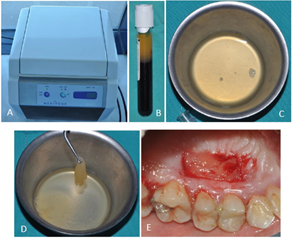
Figure 1: (A) Centrifuge device Medifuge, Silfradent, Italy; (B) Components of centrifuged blood in the tube. Top to bottom layers respectively AFG layer, buffy coat and red blood cells; (C) Liquid form of AFG; (D) Membrane form of AFG; (E) Application of AFG membrane to the surgical area.
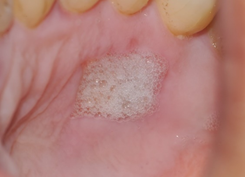
Figure 2: H2O2-bubbling test.
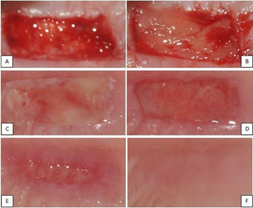
Figure 3: (A) The appearance after graft obtained; (B) The clinical appearance when AFG is applied; (C) Postoperative 3rd day clinical appearance; (D) Postoperative 7th day clinical appearance; (E) Postoperative 14th day clinical appearance; (F) Postoperative 1st month clinical appearance.
|
Manchester Scar Scala (MSS) |
||
|
Visual Analog Scala Excellent Poor |
||
|
Color |
Perfect |
1 |
|
Slight mismatch |
2 |
|
|
Obvious mismatch |
3 |
|
|
Gross mismatch |
4 |
|
|
Contour |
Flush with surrounding skin |
1 |
|
Slightly proud/Indented |
2 |
|
|
Hypertrophic |
3 |
|
|
Keloid |
4 |
|
|
Distortion |
None |
1 |
|
Mild |
2 |
|
|
Moderate |
3 |
|
|
Severe |
4 |
|
Table 1: Manchester Scar Scala for evaluating wound healing.
|
Parameters |
3 Day |
7 Day |
14 day |
1 Month |
|
H202 |
+ |
+ |
+* |
- |
|
VAS (0-10) |
1 |
0 |
0 |
0 |
|
MSS (Color) |
2 |
2 |
1 |
1 |
|
MSS (Contour) |
2 |
2 |
1 |
1 |
|
MSS (Distortion) |
2 |
2 |
1 |
1 |
*non-epithelialized area of %20
Table 2: The evaluation of VAS, H2O2 and Manchester scales at different observation periods.
|
Palatal tissue thickness |
Baseline |
1 Month |
|
4,658 |
4,465 |
Table 3: The comparison of palatal tissue thickness at baseline and first month.
3. Discussion
In this case report, the effect of using AFG in palatal wound healing was presented. Some complications such as excessive bleeding, delayed wound healing, pain, loss of sensation and necrosis can be seen in autogenous soft tissue grafts taken from the palatal area like FGG in the postoperative period [4]. In many studies; hemostatic agents, ozonated oil, herbal products, antibacterial agents, bioactive substances, low level laser therapy and platelet concentrates have been tested to minimize these complications and improve patient comfort [6]. In this case, we used AFG, a platelet concentrate, to reduce postoperative complications and to accelerate the healing process.
There were studies in which platelet concentrates such as PRP, PRF, T-PRF are used for palatal wound healing. Ustao?lu et al. [5] studied the effects of T-PRF on wound healing, patient complaints and palatal tissue thickness in the donor area after FGG; It was observed that the healing process was faster in the T-PRF group and the initial tissue thickness at the palatal region was reached at the end of 6 months. Samanni et al. [9] investigated the effect of PRP, a platelet concentrate, on palatal wound healing. In the split-mouth study, there was a significant difference in all parameters except tissue thickness in the group using PRP, and it was found that it accelerated the healing process. In a study in which PRF was used by Özcan et al. [6], palatal wound healing parameters in the PRF group was found to be better. In this case report, the clinical parameters showed that the healing process was rapid. In addition, the palatal tissue thickness was satisfactory at 1st month. Platelet concentrates (PRP, PRF, T-PRF) used in these studies were applied to the wound surface with suture or adhesive agents. AFG through an adhesive structure, it does not need such application. This also eliminates the risk of adversely affecting the healing that the suture or adhesive agent can cause. The ease of use of the adhesive structure of AFG and the time saved during application make AFG more advantageous than other platelet concentrates.
4. Conclusion
From the literature view, many platelets concentrates have been used for palatal wound healing and AFG was mostly used for making 'sticky bony' by mixing with bone grafts. This case report was the first study to assess the effect of AFG on wound healing, and showed satisfactory clinical results.
Conflict of Interest
The authors declare that they have no conflict of interest.
Acknowledgements
The authors have no declared financial interests.
Written consent was obtained for the publication of photographs from the patient.
References
- Takei HH, Klokkevold PR, Caranza FA. Caranza's Clinical Periodontology. 12th Edn. St Louis, Missouri,Elsevier Saunders (2012).
- Park J-B. Widening keratinized tissue using modified free gingival graft. J Oral Implantol 42 (2016): 114-116.
- Sullivan HC. Free autogenous gingival grafts. I. Principles of successful grafting. Periodontics 6 (1968): 121-129.
- Zucchelli G, Mele M, Stefanini M, Mazzotti C, Marzadori M, et al. Patient morbidity and root coverage outcome after subepithelial connective tissue and de?epithelialized grafts: a comparative randomized?controlled clinical trial. J Clin Periodontol 37 (2010): 728-738.
- Ustao?lu G, Ercan E,Tunali M. The role of titanium-prepared platelet-rich fibrin in palatal mucosal wound healing and histoconduction. Acta Odontol Scand 74 (2016): 5585-5564.
- Ozcan M, Ucak O, Alkaya B, Keceli S, Seydaoglu G, et al. Effects of Platelet-Rich Fibrin on Palatal Wound Healing After Free Gingival Graft Harvesting: A Comparative Randomized Controlled Clinical Trial. Int J Periodontics Restorative Dent 37 (2017): e270-e278.
- El-Sharkawy H, Kantarci A, Deady J, Hasturk H, Liu H, et al. Platelet-rich plasma: growth factors and pro-and anti-inflammatory properties. J Periodontol 78 (2007): 661-669.
- Yeaman MR. The role of platelets in antimicrobial host defense. Clin Infect Dis 25 (1997): 951-968.
- Samani MK, Saberi BV, Tabatabaei SA, Moghadam MG. The clinical evaluation of platelet-rich plasma on free gingival graft's donor site wound healing. Eur J Dent 11 (2017): 447.
- Sohn D. Lecture titled with sinus and ridge augmentation with CGF and AFG. In Symposium on CGF and AFG Tokyo (2010).
- K?z?ltoprak M,Uslu MÖ. Historical Development and Properties of Platelet Concentrates in Dentistry. Cumhuriyet Dent J 21 (2018): 152-166.
- Beausang E, Floyd H, Dunn K, Orton C, Ferguson M. A new quantitative scale for clinical scar assessment. Plast Reconstr Surg 102 (1998): 1954-1961.

